Advanced Resolution of Axially Chiral Oxindole-Substituted Styrenes for Catalytic Applications
The landscape of asymmetric synthesis is continually evolving, driven by the demand for high-purity chiral intermediates that serve as the backbone for advanced pharmaceutical agents and specialized catalysts. Patent CN111848322B introduces a groundbreaking methodology for the resolution of axially chiral oxindole-substituted styrene compounds, addressing a critical gap in the availability of these structurally complex molecules. Traditionally, the synthesis of axially chiral styrenes has relied heavily on transition metal catalysis or limited nucleophilic addition strategies, which often suffer from narrow substrate scope and harsh reaction conditions. This new approach leverages the power of chiral phosphoric acid organocatalysis to achieve highly enantioselective resolution under remarkably mild conditions. By utilizing a specific BINOL-derived chiral phosphoric acid catalyst, the process enables the efficient kinetic resolution of racemic precursors, yielding products with exceptional optical purity. This technological leap not only expands the library of accessible chiral ligands but also provides a robust pathway for generating novel organocatalysts, positioning it as a vital tool for modern medicinal chemistry and process development teams seeking reliable pharmaceutical intermediate supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral styrene skeletons has been a formidable challenge in organic synthesis, primarily due to the high energy barrier associated with rotation around the aryl-alkene bond. Conventional strategies reported in literature, such as those involving palladium-catalyzed allylation or titanium-mediated additions, often require stringent anhydrous conditions and expensive metal precursors. These metal-dependent pathways introduce significant complications regarding product purification, as trace metal residues must be rigorously removed to meet regulatory standards for pharmaceutical applications. Furthermore, many existing methods exhibit limited tolerance for diverse functional groups, restricting the structural diversity of the resulting chiral scaffolds. The reliance on stoichiometric chiral auxiliaries or complex ligand synthesis further exacerbates the cost and operational complexity, making large-scale production economically unfeasible for many organizations. Consequently, there has been a persistent industry need for a more atom-economical and operationally simple strategy that avoids the pitfalls of heavy metal contamination while delivering high stereochemical fidelity.
The Novel Approach
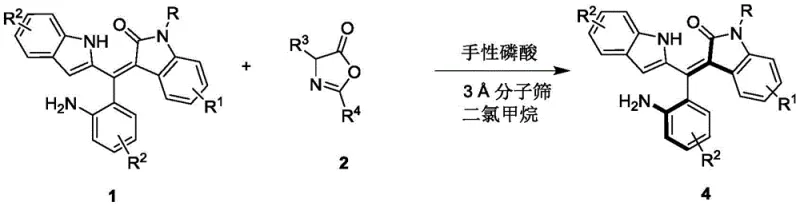
The methodology disclosed in the patent represents a paradigm shift by employing a metal-free organocatalytic system that utilizes readily available azlactone derivatives as resolving agents. Instead of constructing the chiral axis from scratch through difficult bond-forming reactions, this innovative process performs a dynamic kinetic resolution or selective transformation on pre-formed racemic mixtures. The use of dichloromethane as a solvent and 3 Å molecular sieves as additives creates an optimized microenvironment that enhances the interaction between the substrate and the chiral catalyst. This approach allows for the processing of a wide array of substrates with varying electronic and steric properties, as evidenced by the successful resolution of compounds bearing electron-withdrawing trifluoromethyl groups and electron-donating methoxy substituents. The reaction proceeds efficiently at low temperatures, typically around 0°C, which minimizes side reactions and thermal degradation, ensuring that the delicate axially chiral integrity of the molecule is preserved throughout the synthesis. This mildness translates directly into operational safety and reduced energy consumption, key factors for sustainable manufacturing.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Resolution
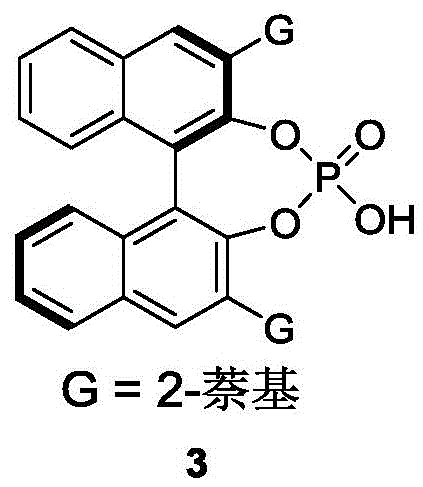
The core of this technological advancement lies in the precise molecular recognition facilitated by the chiral phosphoric acid catalyst, specifically the BINOL-derived structure substituted with bulky 2-naphthyl groups (Compound 3). This catalyst functions as a dual hydrogen-bond donor, activating the electrophilic azlactone species while simultaneously organizing the nucleophilic styrene substrate within a well-defined chiral pocket. The steric bulk of the 2-naphthyl groups at the 3,3'-positions of the BINOL backbone creates a confined space that differentiates between the two enantiomers of the racemic starting material with high fidelity. Through a network of non-covalent interactions, including hydrogen bonding and pi-pi stacking, the catalyst lowers the activation energy for the reaction of one specific enantiomer while sterically hindering the other. This differential reactivity is the driving force behind the high enantiomeric excess (ee) values observed, which can reach up to 96% in optimized examples. The mechanism avoids the formation of unstable intermediates common in metal catalysis, leading to a cleaner reaction profile and simplified downstream processing.
Impurity control is inherently built into this mechanistic framework due to the high specificity of the organocatalyst. Unlike broad-spectrum metal catalysts that might promote various side reactions such as polymerization or over-alkylation, the chiral phosphoric acid directs the reaction pathway exclusively towards the desired addition product. The use of molecular sieves plays a critical role in sequestering water, which could otherwise hydrolyze the sensitive azlactone ring or deactivate the acidic catalyst. This attention to moisture control ensures consistent batch-to-batch reproducibility, a critical parameter for industrial scale-up. Furthermore, the resulting products possess a stable axially chiral configuration that resists racemization under standard storage conditions, guaranteeing long-term shelf stability for the high-purity pharmaceutical intermediate. The ability to fine-tune the catalyst structure by modifying the substituents on the phosphoric acid backbone offers additional levers for optimizing selectivity for specific challenging substrates, providing R&D teams with a versatile platform for method development.
How to Synthesize Axially Chiral Oxindole-Substituted Styrene Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible roadmap for generating these valuable chiral building blocks. The process begins with the preparation of the reaction mixture in a dry environment, emphasizing the importance of excluding moisture to maintain catalyst activity. By carefully controlling the stoichiometry between the racemic styrene precursor and the azlactone resolving agent, operators can maximize the yield of the desired enantiomer while minimizing waste. The reaction is monitored via thin-layer chromatography (TLC) to ensure complete conversion before proceeding to workup, preventing the carryover of unreacted starting materials that could complicate purification. Following the reaction, a simple filtration step removes the solid molecular sieves, and the product is isolated through standard silica gel column chromatography using a petroleum ether and dichloromethane mixture. This straightforward workup procedure underscores the practicality of the method for both laboratory discovery and pilot plant operations.
- Prepare the reaction mixture by combining the racemic styrene precursor (Compound 1) and the azlactone derivative (Compound 2) in dichloromethane solvent with a molar ratio of approximately 1: 0.58.
- Add 3 Å molecular sieves as an additive and introduce the chiral phosphoric acid catalyst (specifically the BINOL-derived catalyst with 2-naphthyl groups) at a loading of 10 mol%.
- Stir the reaction mixture at a controlled low temperature of 0°C until TLC indicates completion, then filter off the molecular sieves and purify the resulting product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this organocatalytic resolution technology offers substantial strategic benefits that extend beyond mere technical performance. The elimination of precious metal catalysts such as palladium or titanium removes a significant cost driver and supply risk associated with volatile metal markets. Transitioning to an organic small-molecule catalyst stabilizes the raw material cost structure and simplifies the supply chain by reducing dependency on specialized metal suppliers. Furthermore, the absence of heavy metals drastically reduces the burden on quality control laboratories, as extensive testing for residual metal content is no longer required, accelerating the release of batches for downstream use. The mild reaction conditions also imply lower energy costs for heating or cooling, contributing to a reduced carbon footprint and aligning with corporate sustainability goals. These factors collectively enhance the overall economic viability of producing complex chiral intermediates, making it an attractive option for cost-sensitive manufacturing campaigns.
- Cost Reduction in Manufacturing: The shift from metal-based catalysis to organocatalysis fundamentally alters the cost equation by removing the need for expensive ligand synthesis and metal scavenging steps. Traditional methods often require costly purification techniques like preparative HPLC or specialized resin treatments to remove trace metals to ppm levels, which adds significant time and expense to the production cycle. By utilizing a metal-free system, the downstream purification process is drastically simplified, often requiring only standard column chromatography or crystallization. This reduction in processing steps translates directly into lower labor costs and higher throughput capacity. Additionally, the catalyst loading is relatively low (10 mol%), and the potential for catalyst recovery or recycling in future iterations could further drive down the cost per kilogram of the final product, offering significant margin improvement opportunities for large-scale production.
- Enhanced Supply Chain Reliability: The starting materials for this resolution process, including the racemic styrene precursors and azlactones, are derived from commodity chemicals that are widely available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks that often plague specialized metal catalyst supply chains. The robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality or minor fluctuations in environmental parameters, ensuring consistent output even in varied manufacturing settings. For supply chain managers, this reliability translates into more predictable lead times and the ability to maintain leaner inventory levels without fear of production stoppages. The scalability of the process from gram to kilogram scale without significant re-optimization further supports a agile supply chain capable of responding quickly to market demand shifts.
- Scalability and Environmental Compliance: The use of dichloromethane as a solvent, while common, is managed effectively within this closed-loop style reaction, and the overall process generates minimal hazardous waste compared to metal-mediated couplings which produce heavy metal sludge. The mild temperature requirements (0°C) reduce the energy load on industrial reactors, allowing for the use of standard cooling systems rather than cryogenic setups. This ease of thermal management facilitates safer scale-up to multi-ton production volumes, as the risk of thermal runaway is negligible. From an environmental compliance standpoint, the absence of toxic metal waste streams simplifies wastewater treatment and disposal procedures, reducing the regulatory burden on manufacturing facilities. This alignment with green chemistry principles not only improves the environmental profile of the product but also future-proofs the manufacturing process against increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is critical for R&D directors evaluating the feasibility of integrating this method into existing synthetic routes and for procurement teams assessing the long-term value proposition. The answers reflect the specific advantages of the chiral phosphoric acid system over legacy technologies, highlighting its role in enabling the efficient production of high-value chiral intermediates.
Q: What are the primary advantages of this organocatalytic resolution over traditional metal-catalyzed methods?
A: This method eliminates the need for expensive and potentially toxic transition metals like palladium or titanium, thereby simplifying purification processes and reducing heavy metal residue risks in the final pharmaceutical intermediate. Additionally, it operates under significantly milder conditions (0°C) compared to many metal-catalyzed couplings.
Q: What level of enantioselectivity can be achieved with this resolution protocol?
A: The patented process demonstrates excellent stereocontrol, achieving enantiomeric excess (ee) values as high as 96% for various substrates, ensuring the production of high-purity chiral building blocks essential for downstream catalytic applications.
Q: Can the resolved styrene compounds be further functionalized into active catalysts?
A: Yes, the resolved axially chiral styrenes serve as versatile precursors that can be derivatized into chiral tertiary amine catalysts, which have been shown to effectively catalyze asymmetric [4+2] cyclization reactions with superior performance compared to commercial alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Oxindole-Substituted Styrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced organocatalytic resolution technology in accelerating the development of next-generation therapeutics and catalysts. As a premier CDMO partner, we possess the technical expertise and infrastructure to seamlessly translate this patented methodology from the laboratory bench to commercial manufacturing scales. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency and quality. We operate state-of-the-art rigorous QC labs equipped to verify stringent purity specifications, including high-performance liquid chromatography (HPLC) for enantiomeric excess determination, guaranteeing that every batch meets the exacting standards required for pharmaceutical applications. Our commitment to process excellence ensures that the high ee values and yields demonstrated in the patent are maintained and optimized during scale-up.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your specific project requirements. Whether you need custom synthesis of specific derivatives or large-scale supply of the core resolved intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your current manufacturing challenges. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this metal-free resolution strategy can enhance your supply chain resilience and reduce overall production costs. Let us be your partner in turning complex chemical challenges into commercial successes.
