Advanced Pd-Catalyzed Multi-Component Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
Introduction to Patent CN115353511A: A Breakthrough in Safe Carbonylation
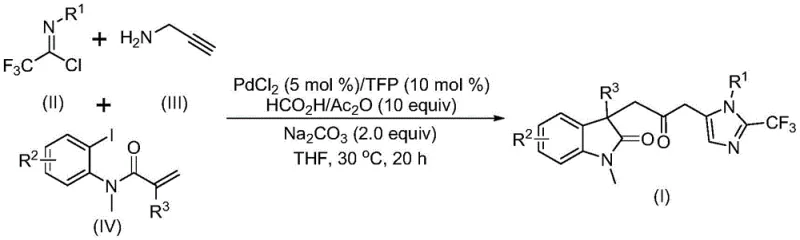
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds, which serve as the core backbone for numerous bioactive molecules. Patent CN115353511A introduces a highly efficient and operationally simple preparation method for carbonyl-bridged biheterocyclic compounds, specifically targeting the synthesis of indolinone-imidazole derivatives. This technology represents a significant leap forward in organic synthesis by replacing the traditional, hazardous use of gaseous carbon monoxide with a safe, in-situ generation system using formic acid and acetic anhydride. For R&D directors and process chemists, this innovation eliminates the need for specialized high-pressure equipment and toxic gas handling protocols, thereby streamlining the workflow for creating valuable pharmacophores. The method employs a palladium-catalyzed multi-component cascade reaction that seamlessly integrates trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives into a single pot. By operating at a mild temperature of 30°C, this process not only ensures high reaction efficiency but also preserves the integrity of sensitive functional groups, making it an ideal candidate for the late-stage functionalization of complex drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbonyl-bridged biheterocyclic systems has relied heavily on direct coupling reactions or oxidative cyclizations that often suffer from poor atom economy and harsh reaction conditions. Traditional carbonylation strategies typically necessitate the use of pressurized carbon monoxide gas, which poses severe safety risks including toxicity, flammability, and the requirement for specialized containment infrastructure. Furthermore, many existing protocols involve multi-step sequences with intermediate isolation, leading to accumulated yield losses and increased waste generation. The reliance on stoichiometric oxidants or expensive transition metal complexes in older methods further exacerbates the cost profile, making large-scale production economically unviable for many generic pharmaceutical applications. Additionally, the substrate scope in conventional approaches is frequently limited, failing to tolerate electron-withdrawing groups or sterically hindered substituents that are common in modern drug design. These limitations create significant bottlenecks for procurement managers and supply chain heads who require reliable, scalable, and cost-effective routes to access these critical intermediates without compromising on safety or environmental standards.
The Novel Approach
In stark contrast, the novel approach detailed in CN115353511A utilizes a transition metal palladium-catalyzed carbonylation cascade that operates under exceptionally mild and safe conditions. By employing a mixture of formic acid and acetic anhydride as a carbon monoxide surrogate, the method completely circumvents the hazards associated with handling CO gas cylinders, effectively transforming a high-risk operation into a standard bench-top procedure. This one-pot multi-component strategy allows for the simultaneous formation of multiple chemical bonds, drastically reducing the number of unit operations and solvent usage compared to stepwise syntheses. The reaction proceeds efficiently at 30°C in common organic solvents like tetrahydrofuran (THF), demonstrating remarkable compatibility with a wide array of functional groups including halogens, alkyls, and nitro groups. This versatility enables the rapid diversification of the molecular scaffold, providing medicinal chemists with a powerful tool for structure-activity relationship (SAR) studies. Moreover, the use of commercially available and inexpensive starting materials such as propargylamine and acrylamide derivatives ensures that the raw material costs remain low, directly addressing the economic concerns of procurement teams while maintaining high purity standards essential for pharmaceutical applications.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation involves a sophisticated sequence of organometallic steps orchestrated by the palladium catalyst system comprising PdCl2 and trifurylphosphine (TFP). The cycle likely initiates with the oxidative addition of zero-valent palladium into the carbon-iodine bond of the acrylamide substrate, generating an aryl-palladium(II) species. This intermediate subsequently undergoes an intramolecular Heck-type reaction to form a divalent alkyl-palladium intermediate, establishing the initial cyclic framework. Crucially, the carbon monoxide required for the carbonyl bridge is released in-situ from the decomposition of the formic acid/acetic anhydride mixture, which then inserts into the palladium-carbon bond to yield an acyl-palladium intermediate. Parallel to this metal cycle, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine species, which undergoes isomerization to become the active nucleophile. The final stage involves the activation of this amidine by the acyl-palladium complex, triggering an intramolecular cyclization that constructs the imidazole ring and releases the final carbonyl-bridged biheterocyclic product while regenerating the active palladium catalyst. This intricate interplay between the metal cycle and the organic transformation ensures high turnover numbers and minimizes the formation of side products, resulting in the clean impurity profiles observed in the experimental data.
From an impurity control perspective, the mild reaction temperature of 30°C plays a pivotal role in suppressing thermal degradation pathways and non-selective radical reactions that often plague high-temperature processes. The use of a specific ligand system (TFP) enhances the stability of the palladium center, preventing the formation of palladium black and ensuring consistent catalytic activity throughout the 12 to 20-hour reaction window. Furthermore, the stoichiometric balance of reagents, particularly the slight excess of propargylamine and acrylamide relative to the imidoyl chloride, drives the equilibrium towards the desired product, minimizing the accumulation of unreacted starting materials. Post-reaction processing involves simple filtration and silica gel treatment followed by column chromatography, which effectively removes residual palladium species and organic by-products. This streamlined purification protocol is critical for meeting the stringent purity specifications required for pharmaceutical intermediates, as it avoids complex extraction procedures that could introduce new contaminants. The robustness of this mechanism against varying electronic properties of the substrates ensures that the process remains reliable even when scaling up to gram-level quantities, providing a solid foundation for industrial adoption.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and mixing protocols to ensure optimal catalyst performance and product yield. The process begins with the preparation of the catalytic system in an anhydrous environment to prevent premature deactivation of the palladium species. Detailed standardized synthetic steps for this specific transformation are provided in the guide below, outlining the exact addition order and workup procedures necessary to achieve the reported high yields.
- Prepare the reaction mixture by combining palladium chloride catalyst, trifurylphosphine ligand, sodium carbonate base, and the formic acid/acetic anhydride CO source in an organic solvent like THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the substituted acrylamide derivative to the catalytic system under inert atmosphere.
- Maintain the reaction at a mild temperature of 30°C for 12 to 20 hours, followed by filtration, silica gel treatment, and column chromatography purification to isolate the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers substantial strategic advantages that extend beyond mere chemical efficiency. The elimination of toxic carbon monoxide gas from the supply chain removes a significant logistical and regulatory burden, as there is no longer a need to procure, store, and monitor hazardous gas cylinders. This shift not only simplifies inventory management but also drastically reduces the insurance and compliance costs associated with handling dangerous goods. Furthermore, the reliance on cheap and readily available starting materials such as propargylamine and acrylamide derivatives ensures a stable and resilient supply base, mitigating the risk of raw material shortages that can disrupt production schedules. The ability to run the reaction at ambient temperatures (30°C) translates directly into lower energy consumption compared to processes requiring reflux or cryogenic conditions, contributing to a reduced carbon footprint and lower utility bills. These factors combined create a compelling business case for integrating this technology into existing manufacturing lines, offering a pathway to significant cost reduction in API manufacturing without sacrificing quality or throughput.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction setup and the avoidance of expensive safety infrastructure. By removing the need for high-pressure autoclaves and gas monitoring systems, capital expenditure for new production lines is significantly lowered. Additionally, the high atom economy of the multi-component reaction means that a larger proportion of the raw material mass is incorporated into the final product, reducing waste disposal costs. The use of a palladium catalyst, while a precious metal, is employed at low loading levels (5 mol%), and the efficiency of the cycle ensures that the cost per kilogram of product remains competitive. Qualitative analysis suggests that the overall cost of goods sold (COGS) can be optimized through these efficiency gains, making the final intermediates more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of commodity chemicals that are widely produced and available from multiple vendors globally. Unlike specialized reagents that may have long lead times or single-source dependencies, the key substrates for this reaction are standard industrial chemicals. This diversity in sourcing options provides procurement teams with greater negotiating power and flexibility, ensuring that production is not halted due to supplier issues. Moreover, the robustness of the reaction conditions allows for manufacturing in a wider range of facilities, including those without specialized high-pressure capabilities, thereby expanding the potential network of contract manufacturing organizations (CMOs) capable of executing the synthesis. This decentralization of production capability enhances overall supply security and reduces lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is facilitated by the absence of gaseous reagents and the use of liquid-phase reagents that are easy to pump and meter. The mild thermal profile reduces the risk of thermal runaway, a critical safety consideration during scale-up, allowing for larger batch sizes with confidence. From an environmental standpoint, the in-situ generation of CO minimizes emissions, and the simplified workup reduces the volume of aqueous and organic waste streams generated per unit of product. This aligns with increasingly strict environmental regulations and corporate sustainability goals, positioning manufacturers who adopt this technology as leaders in green chemistry. The combination of safety, scalability, and environmental friendliness makes this method highly attractive for long-term commercial partnerships and regulatory filings.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the practical aspects of the technology for potential partners and technical stakeholders evaluating its feasibility for their specific projects.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: Unlike conventional methods that require handling toxic and hazardous carbon monoxide gas cylinders, this patent utilizes a formic acid and acetic anhydride mixture to generate carbon monoxide in-situ, significantly enhancing operational safety and reducing regulatory compliance burdens.
Q: What represents the optimal reaction temperature for this multi-component cascade?
A: The patent specifies a remarkably mild reaction temperature of 30°C, which minimizes energy consumption and prevents thermal degradation of sensitive functional groups often found in complex pharmaceutical intermediates.
Q: Can this process accommodate diverse substrate variations for drug discovery?
A: Yes, the method demonstrates excellent substrate compatibility, tolerating various substituents such as halogens, alkyl groups, alkoxy groups, and nitro groups on the aromatic rings, allowing for the rapid generation of diverse chemical libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN115353511A for accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale innovation to industrial reality is seamless. Our state-of-the-art facilities are equipped to handle complex multi-step syntheses with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to guarantee product quality. We understand that the successful commercialization of pharmaceutical intermediates requires not just chemical expertise but also a deep commitment to safety, regulatory compliance, and supply chain reliability, all of which are core tenets of our operational philosophy.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in palladium-catalyzed transformations can drive value and efficiency in your supply chain.
