Scalable Synthesis of 3,5-Disubstituted Pyridines via Iodide-Catalyzed Cyclization for Pharmaceutical Applications
The pharmaceutical and agrochemical industries continuously seek robust, scalable methodologies for constructing nitrogen-containing heterocycles, particularly pyridine derivatives, which serve as critical scaffolds in numerous bioactive molecules. Patent CN111592486B discloses a groundbreaking approach for the construction of 3,5-disubstituted pyridines through a one-step oxidative cyclization reaction involving aryl ethylene and N,N-dimethylformamide (DMF). This innovation addresses long-standing challenges in heterocyclic synthesis by utilizing a cost-effective iodide salt catalytic system coupled with peroxydisulfate oxidation. The significance of this technology lies in its ability to produce symmetrical 3,5-disubstituted pyridines with high selectivity and yield under relatively mild conditions, bypassing the need for expensive transition metal catalysts. For R&D teams focused on developing new active pharmaceutical ingredients (APIs), this method offers a streamlined pathway to access complex pyridine cores that exhibit significant biological activities, such as the anti-inflammatory properties seen in compounds like Sch-21418.
The versatility of this synthetic route is underscored by its broad substrate scope, accommodating various substituted aromatic vinyl compounds. As illustrated in the general reaction scheme below, the process effectively converts simple styrene derivatives into valuable 3,5-diarylpyridines using DMF as both the solvent and the nitrogen source. This dual functionality of DMF not only simplifies the reaction setup but also enhances the atom economy of the process, making it an attractive option for large-scale manufacturing where waste reduction is paramount.
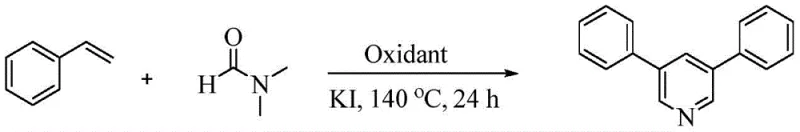
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-diarylpyridines has relied heavily on transition metal-catalyzed cross-coupling reactions or multistep cyclization protocols that often suffer from significant drawbacks. Traditional methods frequently require harsh reaction conditions, including elevated temperatures and the use of toxic heavy metal catalysts such as palladium, copper, or iron, which necessitate rigorous purification steps to meet stringent pharmaceutical purity standards. For instance, earlier approaches reported by Bennasar involved lengthy synthetic sequences starting from N-alkyl-1,4-dihydropyridine derivatives, requiring subsequent N-dealkylation and oxidation steps that resulted in low overall yields and increased operational complexity. Similarly, methods utilizing acryloyl azides or beta-nitrostyrenes often exhibited poor selectivity, generating mixtures of regioisomers that are difficult to separate, or relied on expensive and less accessible starting materials that drive up the cost of goods.
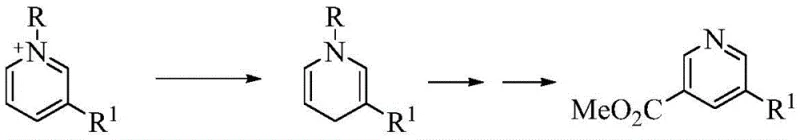
The Novel Approach
In stark contrast, the novel iodide-catalyzed oxidative cyclization described in CN111592486B represents a paradigm shift towards greener and more efficient chemical manufacturing. By employing inexpensive and readily available aryl ethylenes as carbon sources and DMF as a benign carbon-nitrogen synthon, this method drastically reduces raw material costs. The elimination of transition metals is a critical advantage, as it removes the need for costly metal scavenging processes and mitigates the risk of metal residue contamination in the final API. Furthermore, the reaction proceeds in a single pot with high selectivity for the symmetrical 3,5-disubstituted product, achieving yields of up to 69% under optimized conditions. This one-step transformation significantly shortens the production timeline and simplifies the workflow, offering a compelling solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Iodide-Catalyzed Oxidative Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific substrates. The reaction mechanism involves a complex interplay of radical species generated by the interaction of potassium iodide and potassium peroxydisulfate. Initially, DMF undergoes oxidative decomposition in the presence of the iodide catalyst to generate formaldehyde and dimethylamine intermediates. Simultaneously, the aryl ethylene substrate is oxidized to form phenylacetaldehyde. These key intermediates then engage in a cascade of condensation reactions; specifically, the phenylacetaldehyde condenses with formaldehyde to form an unsaturated aldehyde intermediate, which subsequently undergoes ammonia-aldehyde condensation with dimethylamine to generate an iminium ion species. This reactive iminium intermediate then participates in a cyclization addition with a second molecule of the aryl ethylene, ultimately closing the pyridine ring to yield the target 3,5-disubstituted product.
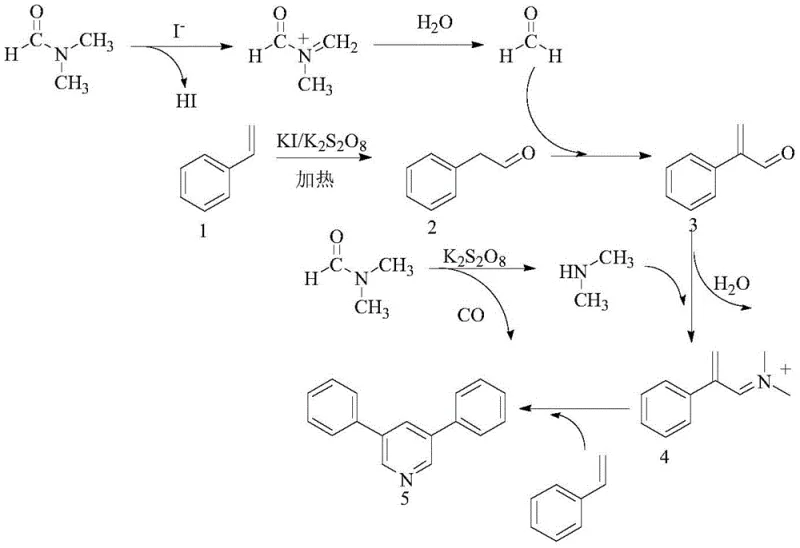
The high selectivity observed in this reaction can be attributed to the specific reactivity of the iodine-mediated radical species, which favors the formation of the symmetrical 3,5-substitution pattern over other potential regioisomers. Impurity control is inherently managed by the stoichiometry of the oxidant and the catalyst; optimization studies revealed that maintaining a specific molar ratio of peroxydisulfate to aryl ethylene (preferably 3:1 to 4:1) is essential to minimize over-oxidation by-products. Additionally, the choice of potassium iodide as the catalyst ensures a clean reaction profile, as other iodine sources or transition metal additives were found to either inhibit the reaction or lead to negligible yields. This mechanistic clarity allows process chemists to confidently scale the reaction, knowing that the formation of side products is minimized through precise control of the oxidative environment.
How to Synthesize 3,5-Disubstituted Pyridine Efficiently
To implement this synthesis in a laboratory or pilot plant setting, strict adherence to the optimized reaction parameters is necessary to achieve the reported high yields. The process utilizes a sealed tube system to maintain the integrity of the volatile components and ensure safety at elevated temperatures. The standardized protocol involves combining the aryl ethylene substrate, potassium peroxydisulfate oxidant, and potassium iodide catalyst in N,N-dimethylformamide, followed by heating at 140 °C for 24 hours. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Combine aryl ethylene (0.5 mmol), potassium peroxydisulfate (1.5 mmol), and potassium iodide (0.75 mmol) in a sealed tube with N,N-dimethylformamide (2 mL) as both solvent and reactant.
- Heat the reaction mixture in an oil bath at 140 °C for 24 hours to facilitate the oxidative cyclization process.
- Upon completion, cool to room temperature, extract with ethyl acetate, wash with aqueous sodium chloride, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodide-catalyzed methodology offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials; styrene derivatives and DMF are commodity chemicals available in bulk quantities from multiple global suppliers, ensuring supply continuity and shielding the manufacturing process from the volatility associated with specialized reagents. Furthermore, the absence of precious metal catalysts eliminates a significant cost center, as there is no need to procure expensive palladium or copper salts, nor is there a requirement for specialized waste treatment facilities to handle heavy metal effluents. This translates directly into a leaner cost structure and improved margin potential for the final commercial product.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound, primarily driven by the elimination of downstream purification costs associated with heavy metal removal. In traditional transition metal-catalyzed processes, significant resources are allocated to scavenging resins and additional chromatography steps to meet regulatory limits for metal residues. By utilizing benign iodide salts, these purification burdens are virtually removed, leading to substantial cost savings in both materials and labor. Additionally, the use of DMF as a dual-purpose solvent and reactant reduces the total volume of chemicals required, further lowering waste disposal costs and improving the overall process mass intensity.
- Enhanced Supply Chain Reliability: Reliance on commodity feedstocks such as styrenes and peroxydisulfates ensures a robust and resilient supply chain that is less susceptible to disruptions compared to routes dependent on bespoke or scarce intermediates. The simplicity of the reaction setup, which does not require inert atmosphere techniques or specialized high-pressure equipment beyond standard sealed tubes, facilitates easier technology transfer between manufacturing sites. This flexibility allows for decentralized production strategies, reducing lead times for high-purity pharmaceutical intermediates and enabling faster response to market demands.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns well with modern green chemistry principles. The avoidance of toxic heavy metals simplifies regulatory compliance and reduces the environmental footprint of the manufacturing facility. The reaction conditions, while requiring elevated temperatures, operate at atmospheric pressure within sealed vessels, posing manageable engineering challenges for scale-up from kilogram to tonne scales. The high selectivity of the reaction minimizes the generation of complex by-product mixtures, streamlining the isolation of the target compound and reducing the solvent load associated with extensive purification procedures.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and optimization studies detailed in the patent documentation, providing clarity on substrate compatibility and reaction constraints for potential licensees or manufacturing partners.
Q: What are the advantages of using iodide salts over transition metal catalysts for pyridine synthesis?
A: Iodide salts, such as potassium iodide, offer a metal-free catalytic system that eliminates the risk of heavy metal contamination in the final product. This significantly simplifies downstream purification processes, reduces environmental toxicity, and lowers overall production costs compared to traditional copper or iron-catalyzed methods.
Q: Can this synthesis method tolerate electron-withdrawing groups on the styrene substrate?
A: Yes, the method demonstrates excellent functional group tolerance. Substrates containing halogen substituents like fluorine, chlorine, and bromine on the benzene ring proceed efficiently with yields comparable to unsubstituted styrene, indicating that electronic effects do not significantly hinder the cyclization reaction.
Q: Is N,N-dimethylformamide (DMF) used solely as a solvent in this reaction?
A: No, DMF serves a dual role in this transformation. It acts as the reaction solvent to dissolve the reagents and simultaneously functions as the carbon-nitrogen source required to construct the pyridine ring, thereby streamlining the synthetic route by reducing the number of distinct reagents needed.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Disubstituted Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iodide-catalyzed cyclization technology for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3,5-disubstituted pyridine delivered meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with our technical team to evaluate the feasibility of this route for your specific project needs. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global supply of fine chemical intermediates.
