Revolutionizing 3,5-Disubstituted Pyridine Production via Metal-Free Oxidative Cyclization for Global Supply Chains
Revolutionizing 3,5-Disubstituted Pyridine Production via Metal-Free Oxidative Cyclization for Global Supply Chains
The landscape of heterocyclic chemistry is undergoing a significant transformation with the emergence of sustainable, metal-free synthetic methodologies. A pivotal development in this arena is documented in Chinese Patent CN111518021B, which details a robust method for constructing 3,5-disubstituted pyridines using mixed styrene derivatives and N,N-dimethylformamide (DMF). This technology represents a paradigm shift for manufacturers of pharmaceutical intermediates, moving away from reliance on precious transition metals towards a more economical iodide-catalyzed oxidative system. The core innovation lies in the ability to perform a one-pot cyclization reaction that simultaneously utilizes DMF as both a solvent and a critical carbon-nitrogen source, thereby streamlining the synthetic route. For R&D directors and procurement specialists alike, this patent offers a compelling solution to the persistent challenges of cost, purity, and scalability in the production of complex pyridine scaffolds found in numerous bioactive molecules.
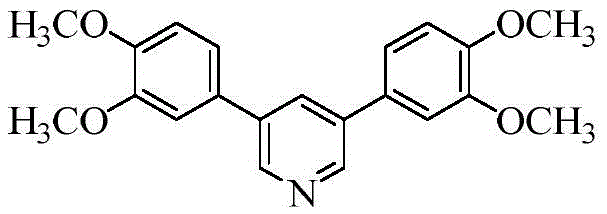
Historically, the synthesis of 3,5-diarylpyridines has been dominated by transition metal-catalyzed cross-coupling reactions, which often necessitate harsh conditions and expensive catalysts such as palladium or copper complexes. These conventional routes frequently suffer from low atom economy and generate significant amounts of heavy metal waste, posing severe challenges for environmental compliance and downstream purification. Furthermore, alternative small-molecule multicomponent cyclizations reported in earlier literature often exhibit poor selectivity or require complex multi-step sequences involving unstable intermediates like acryloyl azides. The limitations of these prior art methods include not only high operational costs but also the difficulty in controlling impurity profiles, which is critical for high-purity pharmaceutical intermediates. The new approach described in the patent addresses these deficiencies by leveraging a simple yet effective oxidative system that operates under relatively mild thermal conditions without the need for exotic reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for accessing the 3,5-disubstituted pyridine core often rely heavily on the functionalization of pre-existing pyridine rings or the cyclization of specialized precursors that are not readily available on a bulk scale. For instance, methods involving the acylation of N-alkyl-1,4-dihydropyridine derivatives followed by N-dealkylation and oxidation are plagued by long reaction sequences and cumulative yield losses at each step. Similarly, iron-mediated syntheses from beta-nitrostyrenes, while operationally simple, utilize substrates that are significantly more expensive than simple styrenes, driving up the raw material costs. The reliance on transition metals introduces another layer of complexity; removing trace metal residues to meet stringent regulatory standards for drug substances requires additional processing steps such as scavenging or recrystallization, which further erodes profit margins. Moreover, many of these older methods struggle to efficiently produce asymmetrical derivatives, limiting their utility in the diverse library synthesis required for modern drug discovery programs.
The Novel Approach
In stark contrast, the novel methodology disclosed in CN111518021B utilizes widely available styrene derivatives and DMF to construct the pyridine ring in a single oxidative step. This approach fundamentally alters the economic equation by replacing costly specialty chemicals with commodity feedstocks. The reaction proceeds via an iodide-catalyzed mechanism where potassium peroxydisulfate serves as the terminal oxidant, generating the necessary reactive intermediates in situ. This metal-free protocol not only simplifies the workup procedure—eliminating the need for heavy metal removal—but also demonstrates excellent functional group tolerance. The process is capable of producing both symmetrical and asymmetrical 3,5-disubstituted pyridines simply by adjusting the ratio of the mixed styrene inputs. This flexibility is a major advantage for commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to rapidly access diverse chemical space without retooling entire production lines.
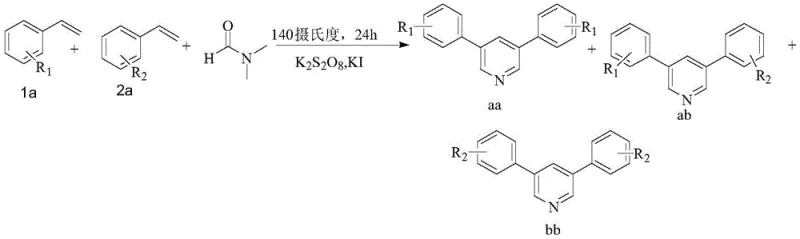
Mechanistic Insights into Iodide-Catalyzed Oxidative Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction initiates with the decomposition of DMF under the influence of the potassium iodide and potassium peroxydisulfate system, generating formaldehyde and dimethylamine as key reactive species. Concurrently, the styrene derivative undergoes oxidation to form a phenylacetaldehyde intermediate. This aldehyde then condenses with the formaldehyde derived from DMF to create an unsaturated ketone intermediate. Subsequent condensation with dimethylamine yields an iminium ion species, which acts as a potent electrophile. This electrophile then reacts with a second molecule of styrene (either the same or different from the first) to close the ring and form the pyridine core. The elegance of this mechanism lies in its modularity; by varying the electronic nature of the substituents on the styrene rings, chemists can influence the ratio of symmetrical versus asymmetrical products. Electron-donating groups such as tert-butyl or methyl generally enhance the reaction efficiency compared to electron-withdrawing halogens, providing a predictive model for substrate selection.
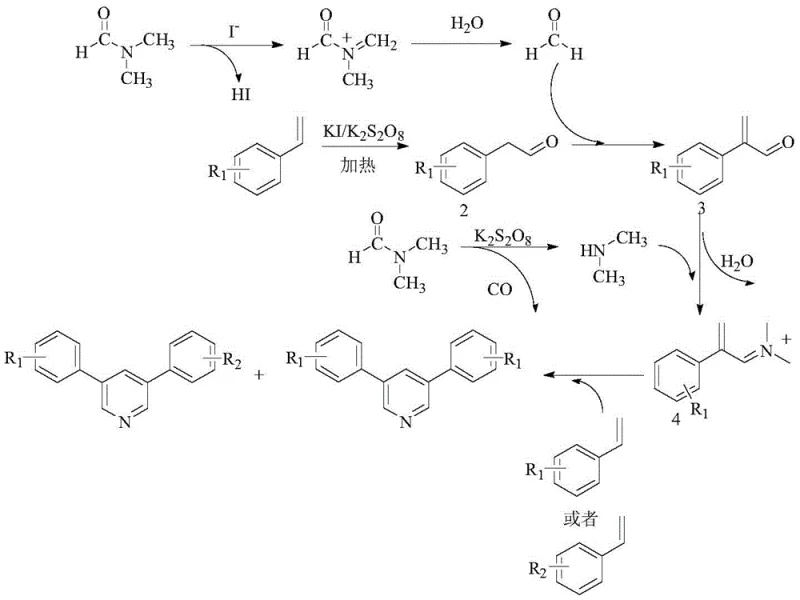
Impurity control in this system is inherently managed by the stoichiometry of the oxidant and the catalyst. Experimental data indicates that maintaining a precise molar ratio of peroxydisulfate to styrene (optimally around 3-4:1) is critical to preventing over-oxidation or polymerization side reactions. Similarly, the concentration of the iodide catalyst must be carefully tuned; insufficient catalyst leads to incomplete conversion, while excess can promote side reactions that lower the overall yield. The reaction produces a mixture of three potential pyridine isomers (AA, AB, and BB) when mixed styrenes are used, with the asymmetrical AB product often being favored due to statistical probability and electronic matching. This predictable distribution allows for efficient separation strategies during purification, ensuring that the final high-purity OLED material or pharmaceutical intermediate meets strict specifications. The absence of metal catalysts also means that the impurity profile is organic in nature, which is generally easier to characterize and remove compared to inorganic metal traces.
How to Synthesize 3,5-Disubstituted Pyridines Efficiently
The practical implementation of this synthesis route is straightforward and amenable to standard laboratory and pilot plant equipment. The process involves charging a reaction vessel with the selected styrene derivatives, DMF, potassium peroxydisulfate, and potassium iodide, followed by heating to the optimized temperature. The simplicity of the reagent setup reduces the risk of operational errors and facilitates rapid technology transfer between sites. Detailed standardized synthetic steps, including precise addition rates and quenching protocols, are outlined in the technical guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the reaction mixture by combining mixed styrene derivatives (Formula 1 and Formula 2), N,N-dimethylformamide (DMF), potassium peroxydisulfate (oxidant), and potassium iodide (catalyst) in a sealed vessel.
- Heat the reaction mixture to an optimized temperature range of 130-140°C and maintain stirring for approximately 24 hours to facilitate oxidative cyclization.
- Upon completion, cool the mixture, extract with ethyl acetate, wash with brine, dry over anhydrous sodium sulfate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers tangible strategic benefits that extend beyond mere chemical novelty. The primary driver for adoption is the significant reduction in raw material costs achieved by substituting expensive transition metal catalysts and specialized precursors with commodity chemicals like styrene and DMF. This shift directly impacts the Cost of Goods Sold (COGS), making the final intermediates more competitive in the global market. Furthermore, the elimination of heavy metals simplifies the regulatory compliance landscape, reducing the time and resources spent on quality control testing for metal residues. This efficiency gain translates into faster batch release times and improved supply chain agility.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with inexpensive iodide salts results in substantial cost savings on catalyst procurement. Additionally, the use of DMF as both solvent and reactant reduces the volume of auxiliary chemicals required, lowering waste disposal costs. The one-pot nature of the reaction minimizes unit operations, reducing energy consumption and labor hours associated with intermediate isolation and purification steps. These cumulative efficiencies drive down the overall manufacturing cost, allowing for more aggressive pricing strategies in competitive tenders.
- Enhanced Supply Chain Reliability: Styrene derivatives and DMF are produced on a massive industrial scale globally, ensuring a stable and continuous supply of raw materials. Unlike specialized organometallic reagents that may suffer from supply chain disruptions or long lead times, the feedstocks for this process are readily available from multiple vendors. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, further enhancing supply chain resilience.
- Scalability and Environmental Compliance: The absence of toxic heavy metals simplifies the treatment of process effluents, aligning with increasingly stringent environmental regulations. Waste streams are primarily organic and can be managed through standard incineration or recovery processes, reducing the environmental footprint of the manufacturing site. The reaction conditions (140°C) are well within the operating range of standard stainless steel reactors, facilitating easy scale-up from kilogram to tonne quantities without the need for specialized pressure vessels or exotic materials of construction. This scalability ensures that the technology can meet growing market demand without significant capital investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative cyclization technology. These answers are derived directly from the experimental data and optimization studies presented in the patent documentation, providing a reliable basis for decision-making.
Q: What are the primary advantages of this metal-free synthesis method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts, significantly simplifying downstream purification processes and reducing heavy metal residue risks in pharmaceutical intermediates.
Q: Can this process produce asymmetrical 3,5-disubstituted pyridines efficiently?
A: Yes, by utilizing a mixture of different styrene derivatives (Formula 1 and Formula 2), the reaction successfully generates asymmetrical products alongside symmetrical ones, with the asymmetrical AB-type often favored depending on substituent electronic effects.
Q: What represents the optimal reaction condition for maximizing yield in this cyclization?
A: Experimental optimization indicates that using potassium peroxydisulfate (1.5 mmol) and potassium iodide (0.75 mmol) at 140°C for 24 hours provides the highest total yield of approximately 56% for the pyridine product mixture.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Disubstituted Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthetic route for the production of high-value heterocyclic intermediates. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging this innovative technology, we can offer our partners a reliable source of 3,5-disubstituted pyridines that meet the highest standards of quality and consistency required by the global pharmaceutical industry.
We invite you to collaborate with us to explore how this cost-effective synthesis method can enhance your supply chain efficiency. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals. Let us be your partner in driving innovation and efficiency in your chemical supply chain.
