Advanced Metal-Free Synthesis of 3,5-Disubstituted Pyridines for Commercial API Production
The pharmaceutical and agrochemical industries continuously seek robust, scalable, and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly the ubiquitous pyridine scaffold. A significant breakthrough in this domain is detailed in Chinese Patent CN111592486B, which discloses a novel method for constructing 3,5-disubstituted pyridines through the cyclization reaction of aryl ethylene and N,N-dimethylformamide (DMF). This technology represents a paradigm shift from traditional transition-metal-catalyzed cross-coupling reactions, offering a metal-free alternative that utilizes inexpensive iodide salts as catalysts and peroxydisulfate as a green oxidant. For R&D directors and procurement managers alike, this innovation addresses critical pain points regarding raw material costs, catalyst toxicity, and process complexity. By leveraging simple starting materials like styrene derivatives and DMF, this one-step oxidative cyclization protocol delivers symmetrical 3,5-disubstituted pyridines with high selectivity and yield, positioning it as a highly attractive route for the commercial manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-diarylpyridines has relied heavily on transition metal-catalyzed cross-coupling reactions or multistep cyclization protocols that often suffer from significant drawbacks. Traditional methods frequently require expensive palladium or copper catalysts, which not only inflate the raw material costs but also necessitate rigorous downstream purification steps to remove trace metal residues to meet stringent pharmaceutical standards. Furthermore, many existing synthetic routes involve harsh reaction conditions, such as extremely high temperatures or the use of hazardous reagents, which pose safety risks and complicate process control. For instance, earlier approaches involving the acylation of N-alkyl-1,4-dihydropyridine derivatives followed by N-dealkylation and oxidation often result in long synthetic sequences with low overall product yields. Additionally, methods utilizing acryloyl azides or beta-nitrostyrenes may offer convenience but frequently struggle with poor selectivity, generating mixtures of regioisomers that are difficult to separate, thereby reducing the efficiency of cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology described in patent CN111592486B introduces a streamlined, one-pot oxidative cyclization strategy that circumvents these historical limitations. By employing aryl ethylene (styrene) and DMF as both solvent and reactant, the process achieves the construction of the pyridine ring in a single operational step. The use of potassium peroxydisulfate as the oxidant and potassium iodide as the catalyst creates a mild yet effective reaction environment that operates optimally at 140°C. This approach not only simplifies the operational workflow but also drastically reduces the generation of chemical waste associated with multi-step syntheses. The reaction exhibits excellent functional group tolerance, accommodating various substituents on the aromatic ring, which is crucial for the diverse needs of medicinal chemistry. As illustrated in the general reaction scheme below, the transformation proceeds efficiently to yield symmetrical 3,5-disubstituted pyridines, demonstrating the potential for reliable pharmaceutical intermediate supplier capabilities.
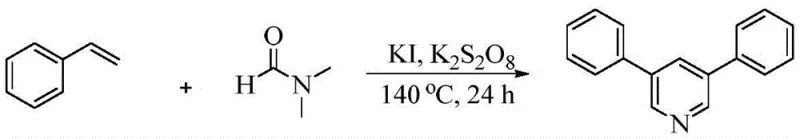
Mechanistic Insights into Iodide-Catalyzed Oxidative Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for R&D teams aiming to optimize the process for specific substrates. The reaction proceeds through a radical-mediated pathway initiated by the interaction between the iodide salt and the peroxydisulfate oxidant. Under thermal conditions, DMF undergoes decomposition to generate formaldehyde and dimethylamine, while the styrene substrate is oxidatively transformed into phenylacetaldehyde. These key intermediates then engage in a cascade of condensation reactions. Specifically, the phenylacetaldehyde condenses with formaldehyde to form an unsaturated aldehyde intermediate, which subsequently undergoes ammonia-aldehyde condensation with dimethylamine to generate an iminium ion species. This electrophilic intermediate then reacts with another molecule of styrene via a cyclization addition step to finally close the pyridine ring. This intricate dance of radical generation and ionic condensation highlights the dual role of DMF as both a carbon and nitrogen source, showcasing a high degree of atom economy.
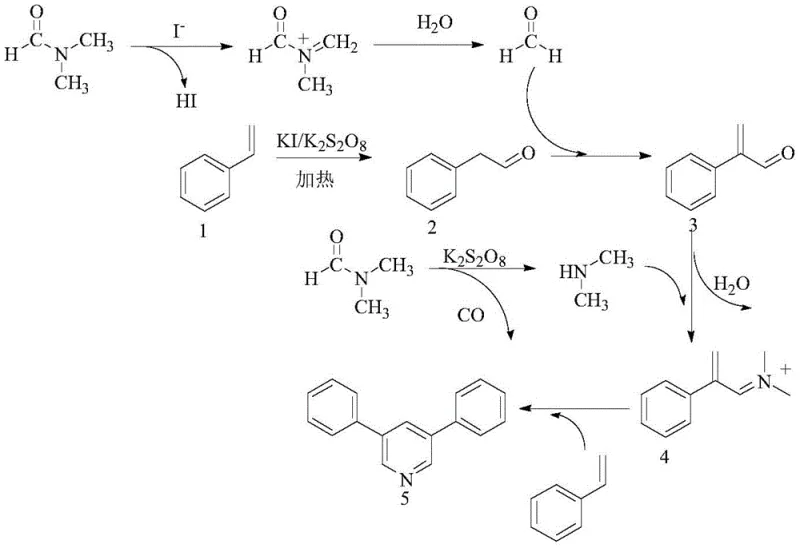
From an impurity control perspective, the mechanism offers distinct advantages. Since the reaction avoids transition metals, the risk of metal-catalyzed side reactions, such as homocoupling of the aryl halides often seen in Suzuki or Heck couplings, is eliminated. The primary by-products are typically derived from over-oxidation or incomplete condensation, which are generally easier to separate via standard chromatographic techniques compared to metal complexes. The selectivity for the 3,5-substitution pattern is driven by the symmetry of the intermediate species formed during the condensation of two equivalents of the aldehyde/styrene derivative with the nitrogen source. This inherent selectivity ensures that the crude product profile is cleaner, reducing the burden on purification teams and facilitating the production of high-purity OLED material or pharmaceutical precursors with minimal effort.
How to Synthesize 3,5-Disubstituted Pyridine Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and minimize by-product formation. The protocol involves mixing the aryl ethylene substrate with an excess of DMF, which serves as the reaction medium, along with stoichiometric amounts of potassium peroxydisulfate and catalytic quantities of potassium iodide. The mixture is heated in a sealed vessel to maintain the necessary pressure and prevent solvent loss. Detailed optimization studies indicate that maintaining the temperature at 140°C for approximately 24 hours provides the optimal balance between reaction rate and product stability. Deviating significantly from these conditions, such as lowering the temperature below 120°C or shortening the reaction time, can lead to incomplete conversion, while excessive temperatures may promote degradation. For a comprehensive understanding of the standardized operating procedures, please refer to the step-by-step guide below.
- Combine aryl ethylene (styrene derivative), potassium peroxydisulfate (oxidant), and potassium iodide (catalyst) in N,N-dimethylformamide (DMF) within a sealed reaction vessel.
- Heat the reaction mixture to an optimized temperature range of 120-150°C, preferably 140°C, and maintain stirring for a duration of 18 to 30 hours to ensure complete cyclization.
- Upon completion, cool the mixture, extract the organic phase with ethyl acetate, wash with aqueous sodium chloride, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic and logistical implications of adopting this technology are profound. The shift from precious metal catalysts to earth-abundant iodide salts represents a direct and significant reduction in raw material costs. Furthermore, the elimination of metal scavenging steps shortens the overall production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demands. The use of DMF as a dual-purpose solvent and reagent simplifies the bill of materials, reducing the complexity of inventory management and minimizing the risk of supply chain disruptions associated with sourcing specialized reagents.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with inexpensive potassium iodide results in substantial cost savings per kilogram of product. Additionally, the one-pot nature of the reaction reduces energy consumption and labor costs associated with multiple isolation and purification steps. The high selectivity of the reaction minimizes the loss of valuable starting materials to side products, further enhancing the overall process efficiency and contributing to significant cost reduction in API manufacturing.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including styrene derivatives, DMF, and potassium peroxydisulfate, are commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of shortages and price volatility often associated with specialized catalysts. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent production output and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The simplicity of the reaction setup, which does not require inert atmosphere protection or high-pressure reactors, facilitates easy scale-up from laboratory to commercial production scales. The absence of heavy metals simplifies waste treatment protocols, aligning with increasingly stringent environmental regulations. This green chemistry approach not only reduces the environmental footprint of the manufacturing process but also lowers the costs associated with waste disposal and regulatory compliance, supporting the commercial scale-up of complex heterocycles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the iodide-catalyzed synthesis of 3,5-disubstituted pyridines. These answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing a reliable foundation for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing production pipelines.
Q: What are the primary advantages of this iodide-catalyzed method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts, utilizing cheap and abundant iodide salts instead. This significantly simplifies the downstream purification process by removing the requirement for rigorous heavy metal scavenging, thereby reducing production costs and environmental impact while maintaining high selectivity.
Q: What is the substrate scope regarding electron-donating and electron-withdrawing groups?
A: The reaction demonstrates broad substrate tolerance. Styrenes substituted with halogens (fluorine, chlorine, bromine) or alkyl groups generally afford good yields comparable to unsubstituted styrene. However, strong electron-donating groups like methoxy at the para-position can inhibit the reaction, whereas ortho- and meta-methoxy substituents provide moderate yields.
Q: Can this process be scaled for industrial manufacturing of pharmaceutical intermediates?
A: Yes, the process is highly amenable to scale-up. It operates as a one-pot reaction using commercially available and inexpensive reagents (styrene, DMF, K2S2O8, KI) under relatively mild thermal conditions without the need for specialized high-pressure equipment or inert gas protection, facilitating straightforward commercial expansion.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Disubstituted Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free cyclization technology for the production of advanced pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3,5-disubstituted pyridine delivered meets the highest industry standards for quality and consistency.
We invite you to collaborate with us to leverage this innovative synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for the future.
