Optimizing Asenapine Intermediate Production: A Technical Breakthrough in Stereocontrol
The pharmaceutical industry's demand for high-purity central nervous system (CNS) agents continues to drive innovation in intermediate synthesis, particularly for complex polycyclic structures like Asenapine. Patent CN101175741B discloses a pivotal advancement in the preparation of trans-5-chloro-2-methyl-2,3,3a,12b-tetrahydro-1H-dibenzo[2,3:6,7]oxepino[4,5-c]pyrrole, a critical precursor to the antipsychotic agent Asenapine (Org 5222). This technical disclosure addresses long-standing stereochemical challenges that have historically plagued the commercial viability of this molecule. By shifting from a direct reduction strategy to a sophisticated ring-opening and recyclization protocol, the patent offers a robust pathway that significantly enhances diastereomeric purity and overall process efficiency. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediate suppliers, understanding this mechanistic shift is essential for securing a stable supply chain for next-generation CNS therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
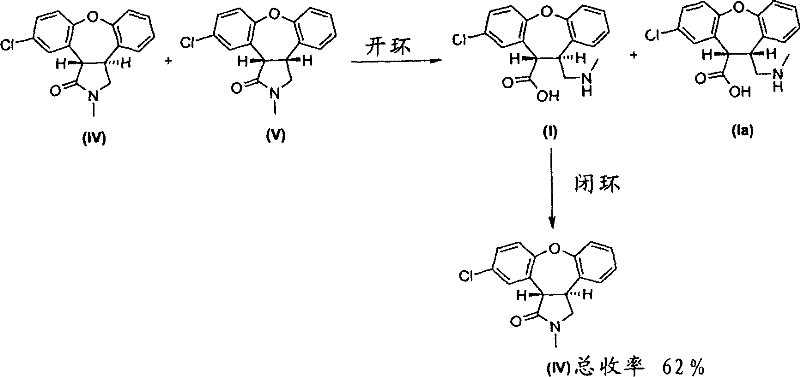
Historically, the synthesis of the target trans-lactam has relied on the magnesium-mediated reduction of an unsaturated alkene amide precursor, specifically compound (III). As illustrated in the prior art, this reduction step is inherently non-selective regarding stereochemistry, producing a crude mixture where the desired trans-isomer (IV) is vastly outnumbered by the unwanted cis-isomer (V) in a ratio of approximately 1:4. To salvage any value from this process, manufacturers were forced to employ tedious isomerization loops using strong bases like DBN (1,5-diazabicyclo[4.3.0]non-5-ene) to equilibrate the mixture. Even after multiple cycles of isomerization and silica gel chromatography, the cumulative total recovery of the desired trans-isomer remained dismally low, hovering around 38%. This reliance on repetitive chromatographic purification not only inflates manufacturing costs but also introduces significant bottlenecks in commercial scale-up of complex pharmaceutical intermediates, making the process economically unattractive for large-volume API production.
The Novel Approach
In stark contrast, the novel methodology presented in the patent circumvents these stereochemical bottlenecks by introducing a strategic ring-opening step. Instead of struggling to separate the difficult lactam isomers directly, the process converts the lactam mixture into open-chain amino acid derivatives. This transformation is remarkably stereospecific, yielding a trans-to-cis amino acid ratio of 10:1, a dramatic improvement over the 1:4 ratio seen in the lactam form. This shift in molecular architecture allows for the efficient separation of the trans-isomer through standard crystallization or extraction techniques, effectively bypassing the need for expensive and time-consuming chromatography. By recycling the purified trans-amino acid back into the lactam form, the process achieves a total yield of approximately 62% from the starting alkene amide, representing a substantial leap in process efficiency and resource utilization.
Mechanistic Insights into Stereospecific Lactam Hydrolysis
The core innovation lies in the base-catalyzed hydrolysis of the lactam ring, which acts as a stereochemical filter. When the mixture of trans-lactam (IV) and cis-lactam (V) is treated with a stoichiometric excess of a strong alkaline base, such as potassium hydroxide or sodium hydroxide, in an alcoholic solvent under reflux, the amide bond cleaves to form the corresponding amino acid carboxylates. The patent data suggests that the transition state for the hydrolysis of the trans-isomer is kinetically or thermodynamically favored under these specific conditions, or conversely, that the cis-isomer is less susceptible to hydrolysis or undergoes rapid epimerization during the process. The result is a solution enriched heavily with the trans-amino acid derivative (I). This open-chain structure possesses different solubility and crystallization properties compared to the rigid bicyclic lactams, facilitating purification. Once isolated, typically as a hydrochloride salt or zwitterion, the trans-amino acid is subjected to cyclization conditions—often heating in an aromatic solvent like toluene with additives like sodium acetate—to reform the lactam ring while strictly preserving the established trans-stereochemistry.
Furthermore, the control of impurities is intrinsically linked to this ring-opening strategy. In the conventional route, the cis-isomer (V) is a persistent impurity that requires active chemical conversion (isomerization) to be useful. In the new process, the cis-amino acid (Ia) remains in the mother liquor or aqueous phase during the isolation of the trans-product, effectively removing it from the process stream without additional chemical steps. This "purification by transformation" approach minimizes the carryover of diastereomeric impurities into the final API, ensuring that the subsequent reduction to Asenapine proceeds with high fidelity. The ability to isolate the intermediate as a stable hydrochloride salt or zwitterion also provides flexibility in storage and handling, reducing the risk of degradation or racemization during long-term inventory holding.
How to Synthesize Trans-Asenapine Lactam Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for executing this high-yield transformation. It begins with the preparation of the lactam mixture, followed by the critical hydrolysis step using alcoholic KOH, and concludes with the acidification and recyclization of the purified amino acid. This sequence replaces the inefficient chromatographic loops of the past with a streamlined crystallization-driven workflow.
- Perform stereospecific ring-opening of the lactam mixture using excess strong base in alcoholic solvent to generate amino acid derivatives.
- Separate the trans-amino acid derivative from the cis-isomer via crystallization or extraction, leveraging the improved 10: 1 ratio.
- Cyclize the purified trans-amino acid derivative back to the lactam form using aromatic solvents and additives like sodium acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates directly into tangible operational benefits and risk mitigation. The primary advantage is the drastic simplification of the purification train. By eliminating the need for multiple rounds of silica gel chromatography and DBN isomerization cycles, the process reduces the consumption of expensive stationary phases and organic solvents. This reduction in material intensity leads to cost reduction in pharmaceutical intermediate manufacturing by lowering both raw material costs and waste disposal fees associated with chromatographic effluents. Additionally, the higher overall yield means that less starting material is required to produce the same amount of final product, further optimizing the cost of goods sold (COGS) and improving the margin profile for the final API.
- Cost Reduction in Manufacturing: The shift from a 38% yield process to a 62% yield process fundamentally alters the economics of production. Higher yields mean reduced raw material consumption per kilogram of output. Furthermore, replacing chromatography with crystallization significantly lowers processing time and energy consumption. The elimination of repetitive isomerization loops reduces labor hours and reactor occupancy time, allowing for faster batch turnover and increased facility throughput without the need for capital expansion.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like potassium hydroxide, ethanol, and toluene ensures that the supply chain is not vulnerable to shortages of specialized reagents. The robustness of the crystallization steps provides a consistent quality output that is less prone to the variability often seen in chromatographic separations. This consistency is crucial for maintaining regulatory compliance and ensuring reducing lead time for high-purity pharmaceutical intermediates, as batches are less likely to fail quality control specifications due to residual impurities or solvent issues.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in solvent usage and the elimination of silica gel waste align with green chemistry principles. The process generates less hazardous waste, simplifying effluent treatment and reducing the environmental footprint of the manufacturing site. The scalability is proven by the patent's description of kilogram-scale operations, demonstrating that the chemistry holds up when moving from the bench to pilot and commercial scales, thereby securing a reliable pharmaceutical intermediate supplier status for partners adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this stereocontrolled synthesis. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for process evaluation.
Q: How does the new ring-opening method improve yield compared to traditional reduction?
A: Traditional magnesium reduction yields a 1:4 trans-to-cis mixture requiring complex isomerization loops. The novel ring-opening strategy achieves a 10:1 ratio immediately, boosting overall recovery from approximately 38% to 62%.
Q: What reagents are critical for the stereospecific hydrolysis step?
A: The process utilizes stoichiometric excesses of strong alkaline bases, such as potassium hydroxide or sodium hydroxide, in C1 to C6 alcoholic solvents under reflux conditions to ensure complete and selective conversion.
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the method eliminates the need for repetitive silica gel chromatography cycles used in older methods, relying instead on crystallization and standard extraction, which are highly amenable to multi-kilogram and ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asenapine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex CNS agents like Asenapine depends on mastering the nuances of stereochemical control. Our technical team has extensively analyzed the pathways described in CN101175741B and possesses the expertise to implement this advanced ring-opening strategy effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of monitoring diastereomeric ratios with high precision, guaranteeing that every batch meets stringent purity specifications required by global regulatory bodies.
We invite pharmaceutical partners to collaborate with us to leverage this superior synthetic route for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this yield-enhancing technology can impact your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemistry available in the market.
