Optimizing Asenapine Intermediate Production Through Novel Copper-Catalyzed Coupling Strategies
Optimizing Asenapine Intermediate Production Through Novel Copper-Catalyzed Coupling Strategies
The pharmaceutical landscape for antipsychotic medications continues to evolve, driven by the need for more efficient and cost-effective synthetic routes for active pharmaceutical ingredients (APIs). A pivotal development in this domain is documented in Chinese Patent CN101563312A, which outlines a groundbreaking method for synthesizing key intermediates of Asenapine, a potent schizophrenic drug. This patent introduces a strategic shift in raw material selection, moving away from scarce brominated precursors to readily available chlorinated acids. For R&D directors and procurement specialists alike, this transition represents a significant opportunity to streamline supply chains and reduce manufacturing overheads. The core innovation lies in the copper-catalyzed coupling of 2-chlorophenylacetic acid with 4-chlorophenol, a reaction that surprisingly proceeds with high efficiency despite the lower reactivity typically associated with aryl chlorides compared to aryl bromides.
This technical insight report delves deep into the mechanistic nuances and commercial implications of this patented process. By leveraging inexpensive starting materials and a robust catalytic system, manufacturers can achieve substantial cost reductions in pharmaceutical intermediate manufacturing. The methodology described not only addresses the economic constraints of traditional synthesis but also enhances the scalability of the production process. As global demand for mental health treatments rises, securing a reliable pharmaceutical intermediate supplier who can deliver high-purity compounds through such optimized routes becomes critical. The following analysis provides a comprehensive breakdown of the chemical transformations, highlighting how this novel approach overcomes historical bottlenecks in Asenapine synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Asenapine and its precursors has relied heavily on the use of methyl 2-bromophenylacetate as a key starting material. As illustrated in the prior art reaction pathways, this conventional approach presents several formidable challenges for industrial scale-up. Firstly, methyl 2-bromophenylacetate is not a commodity chemical; it is difficult to obtain in bulk quantities and often requires complex, multi-step preparation itself, which inherently drives up the cost of goods sold (COGS). Secondly, the reliance on brominated species introduces volatility into the supply chain, as bromine-based reagents are subject to greater market fluctuation and regulatory scrutiny compared to their chlorinated counterparts. Furthermore, the handling of brominated intermediates often necessitates stricter safety protocols due to their potential lachrymatory and toxic properties, adding layers of operational complexity to the manufacturing environment.
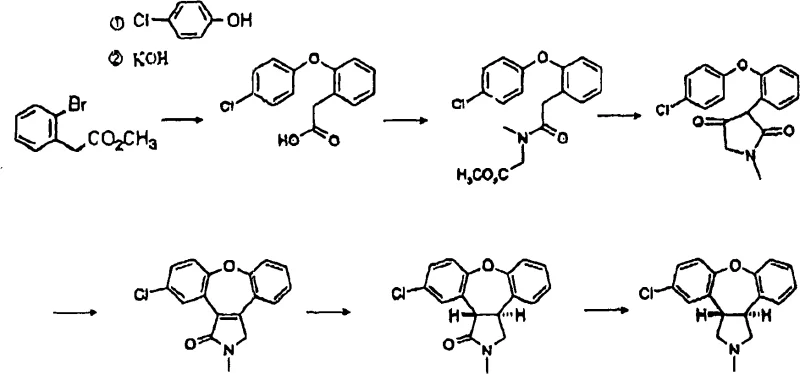
In addition to sourcing difficulties, the conventional route depicted in earlier patents often suffers from lower atom economy and generates significant waste streams associated with the protection and deprotection steps required for the ester functionality. The necessity to convert the ester to the acid later in the sequence adds unnecessary unit operations, consuming time, energy, and solvents. For a procurement manager, these inefficiencies translate directly into higher purchase prices and longer lead times for high-purity pharmaceutical intermediates. The cumulative effect of these limitations is a fragile supply chain that struggles to meet the rigorous demands of modern Good Manufacturing Practice (GMP) standards while maintaining competitive pricing structures. Consequently, there is an urgent industry need for a synthetic strategy that bypasses these brominated bottlenecks entirely.
The Novel Approach
The innovative methodology presented in Patent CN101563312A fundamentally重构 s the synthetic landscape by substituting the problematic brominated ester with 2-chlorophenylacetic acid. This simple yet profound change unlocks a cascade of benefits, primarily centered around the availability and cost of raw materials. 2-Chlorophenylacetic acid is a widely produced commodity chemical, ensuring a stable and continuous supply stream that is immune to the shortages often plaguing specialized brominated reagents. The new reaction pathway, as comprehensively detailed in the patent figures, demonstrates that this chlorinated acid can be directly coupled with 4-chlorophenol to form the critical diaryl ether linkage. This direct coupling eliminates the need for ester hydrolysis steps early in the sequence, thereby simplifying the overall process flow and reducing the total number of processing stages required to reach the key intermediate.
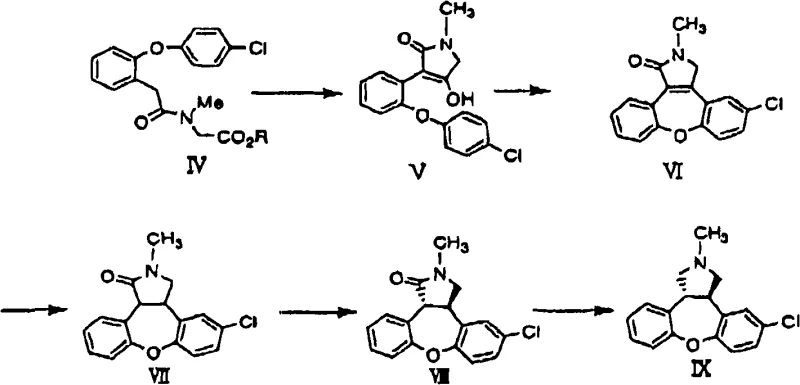
Moreover, the novel approach leverages a sophisticated copper-catalyzed system that activates the relatively inert aryl chloride bond, a feat that was previously considered challenging without expensive palladium catalysts. By utilizing copper(I) halides in conjunction with amino acid ligands, the process achieves high conversion rates at moderate temperatures. This technological leap allows for the commercial scale-up of complex pharmaceutical intermediates with significantly reduced capital expenditure on catalyst recovery systems. The ability to use cheap, abundant chlorinated feedstocks while maintaining high reaction yields positions this method as a superior alternative for cost reduction in API manufacturing. It effectively transforms a niche, high-cost synthesis into a robust, commodity-driven process that aligns perfectly with the strategic goals of supply chain heads looking to optimize inventory turnover and minimize risk.
Mechanistic Insights into Copper-Catalyzed Ullmann-Type Coupling
The heart of this synthetic breakthrough lies in the mechanistic intricacies of the copper-catalyzed Ullmann-type coupling reaction. Unlike traditional nucleophilic aromatic substitutions that require harsh conditions or electron-withdrawing groups, this protocol employs a catalytic cycle involving copper(I) species coordinated with amino acid ligands, such as N-methylglycine. The mechanism initiates with the oxidative addition of the copper catalyst into the carbon-chlorine bond of the 2-chlorophenylacetic acid. This step is facilitated by the electron-donating nature of the ligand, which stabilizes the copper center and lowers the activation energy barrier for the cleavage of the strong C-Cl bond. Following oxidative addition, the 4-chlorophenol, activated by a base to form a phenoxide anion, undergoes transmetallation with the copper complex. This creates a transient organocopper species where both the aryl group and the phenoxy group are coordinated to the metal center.
The final step in the catalytic cycle is reductive elimination, which forge the new carbon-oxygen bond to yield the desired diaryl ether product while regenerating the active copper(I) catalyst. A critical aspect of this mechanism is the remarkable regioselectivity observed during the reaction. The patent data explicitly notes that the substitution occurs exclusively at the 2-position of the phenylacetic acid ring, leaving the chlorine atom on the 4-chlorophenol ring untouched. This selectivity is paramount for R&D directors concerned with impurity profiles, as it prevents the formation of bis-coupled byproducts or polymeric tars that are common in less controlled coupling reactions. The use of bases like cesium carbonate or potassium carbonate further enhances the reaction kinetics by ensuring a high concentration of the nucleophilic phenoxide species. Understanding this mechanistic pathway is essential for troubleshooting potential scale-up issues, such as catalyst deactivation or incomplete conversion, ensuring that the process remains robust from the laboratory bench to the 100 MT production scale.
How to Synthesize 2-(2-(4-chlorophenoxy)phenyl)acetic Acid Efficiently
The practical execution of this synthesis involves a carefully orchestrated sequence of reaction conditions designed to maximize yield and purity. The process begins with the charging of 2-chlorophenylacetic acid and 4-chlorophenol into a reactor containing a high-boiling solvent such as diethylene glycol dimethyl ether (diglyme) or toluene. The addition of a copper(I) halide catalyst, typically copper(I) bromide or iodide, along with an amino acid ligand, sets the stage for the coupling event. The reaction mixture is then heated to temperatures ranging between 120°C and 160°C, a thermal window that provides sufficient energy for the catalytic cycle to proceed rapidly without degrading the sensitive functional groups. Detailed standardized synthesis steps for this transformation are provided in the guide below, outlining precise molar ratios and workup procedures to ensure reproducibility.
- React 2-chlorophenylacetic acid with 4-chlorophenol in the presence of a copper(I) catalyst and base in diglyme or toluene at 120-160°C.
- Convert the resulting carboxylic acid intermediate into its reactive acyl halide derivative using thionyl chloride.
- Condense the acyl halide with N-methylglycine ethyl ester under basic conditions to form the key amide intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers transformative advantages that extend far beyond simple chemical curiosity. The primary driver of value is the drastic simplification of the raw material portfolio. By switching to 2-chlorophenylacetic acid, companies can leverage existing global supply networks for commodity chemicals, thereby insulating themselves from the volatility associated with specialized brominated reagents. This shift not only stabilizes pricing but also significantly shortens lead times for high-purity pharmaceutical intermediates, as suppliers can maintain larger stockpiles of the chlorinated acid. Furthermore, the elimination of the ester hydrolysis step reduces the consumption of solvents and reagents, leading to a leaner manufacturing process with a smaller environmental footprint. These operational efficiencies translate directly into improved margins and a more competitive position in the global marketplace.
- Cost Reduction in Manufacturing: The replacement of expensive, hard-to-source methyl 2-bromophenylacetate with廉价 2-chlorophenylacetic acid results in substantial raw material cost savings. Additionally, the streamlined process flow reduces the number of unit operations, lowering energy consumption and labor costs associated with multiple isolation and purification steps. The use of earth-abundant copper catalysts instead of precious metals further contributes to a lower cost basis, making the final API more affordable to produce without compromising quality standards.
- Enhanced Supply Chain Reliability: Relying on commodity-grade starting materials ensures a consistent and reliable supply chain, mitigating the risk of production stoppages due to raw material shortages. The robustness of the copper-catalyzed coupling reaction means that the process is less sensitive to minor variations in reagent quality, providing a buffer against supply chain disruptions. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the strict delivery deadlines demanded by downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The reaction conditions employed, such as the use of toluene and moderate temperatures, are fully compatible with existing industrial infrastructure, facilitating seamless scale-up from pilot plant to commercial production. Moreover, the reduction in waste generation and the avoidance of hazardous brominated byproducts simplify waste treatment processes, ensuring compliance with increasingly stringent environmental regulations. This alignment with green chemistry principles enhances the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These answers are derived directly from the experimental data and claims within Patent CN101563312A, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this technology for their own production lines. The insights provided here cover aspects ranging from catalyst selection to impurity control, ensuring a holistic view of the process capabilities.
Q: What is the primary advantage of using 2-chlorophenylacetic acid over methyl 2-bromophenylacetate?
A: 2-Chlorophenylacetic acid is significantly more commercially available and cost-effective compared to the brominated ester analogue, reducing raw material procurement complexity and overall production costs.
Q: How does the copper catalyst system ensure regioselectivity in this synthesis?
A: The catalytic system selectively activates the chlorine atom at the 2-position of the phenylacetic acid ring for nucleophilic substitution, while leaving the 4-chlorophenol chlorine intact, ensuring high purity of the desired ether linkage.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes robust solvents like toluene and diglyme and operates at moderate temperatures (120-160°C), making it highly scalable and compatible with standard industrial reactor setups without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asenapine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical industry. Our team of expert chemists has extensively evaluated the methodology described in Patent CN101563312A and possesses the technical capability to implement this copper-catalyzed coupling strategy at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Asenapine intermediate adheres to the highest international standards.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage these advanced synthetic technologies. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce manufacturing costs through the adoption of this innovative and economically superior synthetic pathway.
