Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
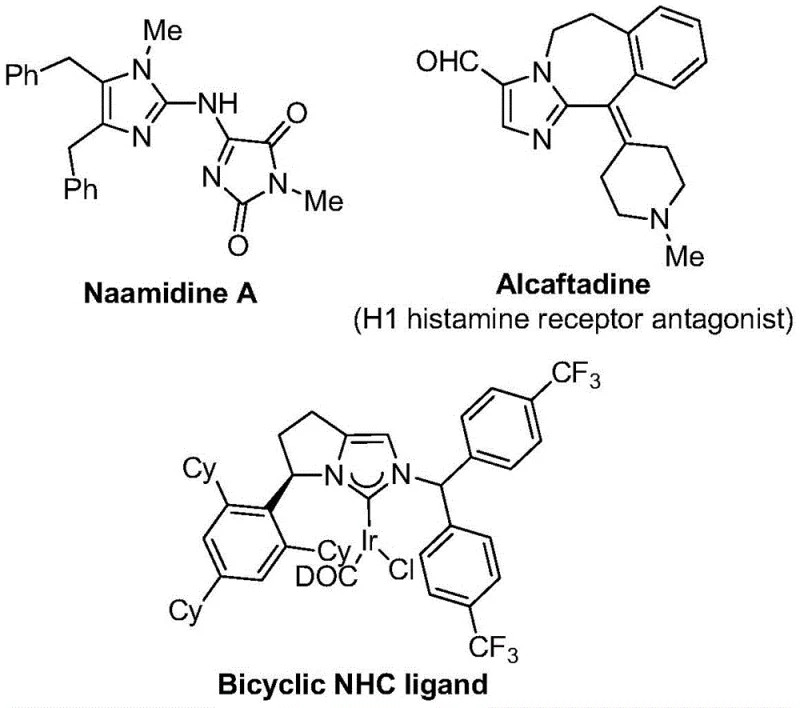
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups, due to their profound impact on metabolic stability and lipophilicity. A significant breakthrough in this domain is detailed in Chinese patent CN111423381B, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted imidazole compounds. This technology leverages a transition metal palladium-catalyzed carbonylation series reaction, utilizing readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryl iodide salts. The strategic introduction of the trifluoromethyl moiety into the imidazole backbone addresses critical challenges in drug design, enhancing the bioavailability of potential therapeutic agents. As a leading entity in the chemical sector, understanding the nuances of this patent is essential for developing a reliable pharmaceutical intermediate supplier strategy that meets the rigorous demands of modern drug discovery.
The structural diversity and biological significance of imidazole derivatives cannot be overstated, as evidenced by their presence in numerous bioactive natural products and synthetic drugs. The patent highlights that imidazole compounds serve not only as key pharmacophores but also as versatile ligands in coordination catalysis and components in ionic liquids. The specific innovation described allows for the synthesis of diversified substituted imidazoles bearing trifluoromethyl groups through rational substrate design. This capability is paramount for medicinal chemists aiming to optimize lead compounds, as the electron-withdrawing nature of the CF3 group can drastically alter the electronic properties of the heterocycle. By mastering this synthetic route, manufacturers can offer high-purity OLED material precursors or specialized agrochemical intermediates, thereby expanding their portfolio into high-value market segments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles functionalized with trifluoromethyl groups has been fraught with significant technical hurdles and safety concerns. Conventional literature often relies on the direct reaction of synthons possessing a trifluoromethyl group with suitable substrates, frequently employing reagents like trifluorodiazoethane. While effective in certain contexts, trifluorodiazoethane is known for its instability and potential explosiveness, posing severe risks during large-scale commercial operations. Furthermore, existing methods utilizing trifluoroethylimidoyl halides have not been widely exploited or optimized, leaving a gap in practical, scalable methodologies. The reliance on harsh reaction conditions, expensive catalysts, or difficult-to-handle gaseous reagents in traditional protocols often results in poor atom economy and complicated downstream processing. These limitations hinder the ability of procurement teams to secure cost-effective raw materials and complicate the supply chain continuity for critical API intermediates.
The Novel Approach
In stark contrast to legacy techniques, the novel approach outlined in the patent utilizes a mild, one-pot multicomponent reaction that elegantly constructs the imidazole ring while simultaneously installing the trifluoromethyl group. The core transformation involves the coupling of trifluoroethylimidoyl chloride, propargylamine, and a diaryl iodonium salt under palladium catalysis. This method operates at a remarkably mild temperature of 30°C, eliminating the need for energy-intensive heating or cryogenic cooling. The use of formic acid and acetic anhydride serves as a safe and convenient source of carbon monoxide in situ, bypassing the hazards associated with handling high-pressure CO gas cylinders. This streamlined process not only simplifies the operational workflow but also enhances the overall safety profile of the manufacturing plant. By adopting this methodology, companies can achieve substantial cost savings in specialty chemical production through reduced energy consumption and simplified equipment requirements.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
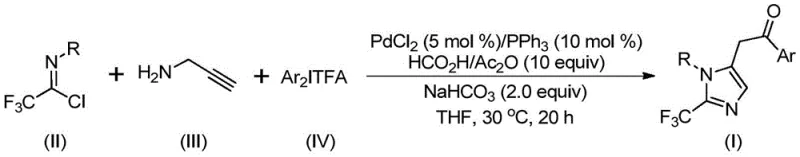
The mechanistic pathway of this transformation is a sophisticated sequence of organometallic steps that ensures high regioselectivity and yield. Initially, an intermolecular carbon-nitrogen bond is formed under basic conditions to generate a trifluoroacetamidine intermediate, which subsequently undergoes isomerization. The palladium catalyst then activates the alkyne moiety of the propargylamine derivative through palladation, forming a key alkenyl palladium species. This intermediate undergoes further isomerization to an alkyl palladium complex, setting the stage for the crucial carbonylation step. The carbon monoxide, released from the decomposition of formic acid and acetic anhydride, inserts into the palladium-carbon bond to create an acyl palladium intermediate. Finally, oxidative addition of the diaryl iodonium salt generates a tetravalent palladium species, which undergoes reductive elimination to release the final 2-trifluoromethyl-substituted imidazole product and regenerate the active catalyst. Understanding this cycle is vital for R&D directors focusing on impurity control and process optimization.
Impurity control in this system is inherently managed by the specificity of the palladium cycle and the mild reaction conditions. The use of sodium bicarbonate as a base helps neutralize acidic byproducts without promoting unwanted side reactions that stronger bases might induce. The choice of tetrahydrofuran (THF) as the preferred solvent ensures excellent solubility of all reactants, facilitating homogeneous catalysis and minimizing the formation of oligomeric byproducts often seen in heterogeneous systems. The tolerance for various functional groups, including halogens and electron-withdrawing nitro groups, suggests that the catalytic cycle is robust against competitive coordination or deactivation. This robustness translates to a cleaner crude reaction profile, reducing the burden on purification teams and ensuring that the final product meets stringent purity specifications required for pharmaceutical applications. The ability to tune the electronic properties of the aryl rings on both the imidoyl chloride and the iodonium salt allows for precise modulation of the reaction kinetics.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis protocol requires careful attention to reagent stoichiometry and mixing order to maximize efficiency. The patent describes a procedure where palladium chloride and triphenylphosphine are combined with the substrates in an organic solvent, creating a reactive environment for the cascade reaction. The molar ratios are optimized to ensure complete conversion while minimizing excess reagent waste, typically employing a slight excess of the imidoyl chloride and iodonium salt relative to the alkyne. The reaction proceeds smoothly at ambient-like temperatures, making it accessible for facilities without specialized high-temperature reactors. For detailed operational parameters and specific stoichiometric ratios tailored to your production scale, please refer to the standardized synthesis steps provided below.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid, trifluoroethylimidoyl chloride, propargylamine, and diaryl iodide in an organic solvent like THF.
- Stir the reaction mixture at a mild temperature of 30°C for a duration of 16 to 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl substituted imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers transformative advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for complex heterocycles. The primary benefit lies in the drastic simplification of the supply chain for raw materials; trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts are all commercially available commodities that can be sourced from multiple vendors, reducing dependency on single-source suppliers. This availability mitigates the risk of supply disruptions and stabilizes pricing volatility. Furthermore, the elimination of hazardous gases like carbon monoxide from the external supply chain removes significant regulatory and logistical burdens associated with transporting and storing toxic compressed gases. The mild reaction conditions also imply lower energy costs and reduced wear and tear on reactor vessels, contributing to a more sustainable and economically viable manufacturing process.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive palladium chloride compared to more exotic noble metal catalysts, coupled with the high atom economy of the multicomponent reaction. By avoiding the need for specialized high-pressure equipment required for traditional carbonylations, capital expenditure for new production lines is significantly lowered. The simplicity of the workup, involving basic filtration and chromatography, reduces labor hours and solvent consumption during the isolation phase. Additionally, the high yields reported across a broad range of substrates mean that less starting material is wasted, directly improving the cost of goods sold (COGS) for the final intermediate. These factors collectively enable a competitive pricing structure for high-purity pharmaceutical intermediates in the global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining long-term supply contracts with major pharmaceutical clients. Since the reaction tolerates a wide array of functional groups, the same core process can be adapted to produce a library of diverse derivatives without needing to requalify entirely new synthetic routes. This flexibility allows suppliers to respond rapidly to changing customer demands for specific analogues during the drug development lifecycle. The use of stable, shelf-stable reagents further enhances reliability, as inventory can be held for longer periods without degradation, ensuring that production can resume quickly after any unforeseen interruptions. This stability is crucial for reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the absence of exothermic hazards and the use of common organic solvents like THF. The mild temperature profile (30°C) simplifies heat management in large reactors, removing the need for complex cooling jackets or heating mantles. From an environmental standpoint, the in situ generation of carbon monoxide minimizes the release of greenhouse gases and eliminates the risk of leaks associated with gas cylinders. The waste stream is primarily composed of organic solvents and inorganic salts, which can be treated using standard wastewater treatment protocols familiar to most chemical plants. This alignment with green chemistry principles supports corporate sustainability goals and ensures compliance with increasingly strict environmental regulations globally.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope and operational details. These insights are derived directly from the experimental data and embodiments presented in the patent documentation, ensuring accuracy and relevance. Understanding these nuances helps in making informed decisions about process adoption and resource allocation. For more specific technical data or custom synthesis requests, our team is ready to provide further assistance.
Q: What are the optimal reaction conditions for this palladium-catalyzed synthesis?
A: The patent specifies reacting the components at 30°C for 16-24 hours in an aprotic solvent like tetrahydrofuran (THF), using PdCl2 and PPh3 as the catalytic system.
Q: Can this method tolerate diverse functional groups on the aryl rings?
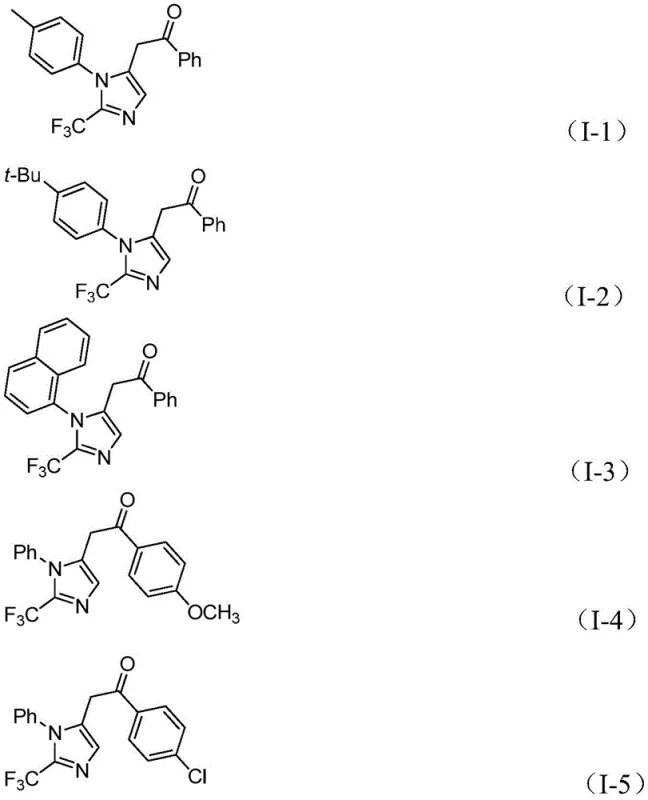
A: Yes, the method demonstrates excellent substrate compatibility, successfully accommodating substituents such as methyl, tert-butyl, chlorine, bromine, trifluoromethyl, nitro, and methoxy groups on both the imidoyl chloride and the diaryl iodonium salt.
Q: Is the post-treatment process complex for industrial application?
A: No, the post-processing is straightforward, involving simple filtration followed by standard silica gel column chromatography, which facilitates easy scale-up and purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in patent CN111423381B for driving innovation in the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. Whether you require this specific imidazole derivative for oncology research or as a building block for agrochemicals, our infrastructure is designed to support your growth with reliability and precision.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates how this specific route can optimize your current supply chain economics. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your unique molecular targets. Let us help you navigate the complexities of fine chemical manufacturing with confidence and efficiency.
