Scalable Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles: A Metal-Free Route for Commercial Production
Scalable Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles: A Metal-Free Route for Commercial Production
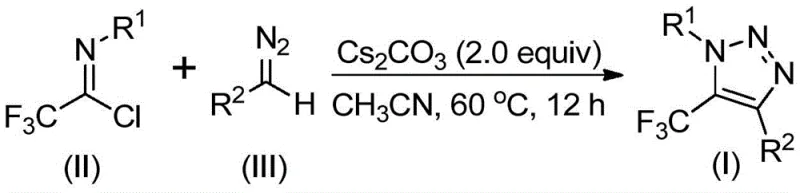
The landscape of heterocyclic chemistry is constantly evolving to meet the stringent demands of modern pharmaceutical and agrochemical manufacturing. A pivotal advancement in this field is detailed in Chinese Patent CN113121462B, which discloses a highly efficient preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. These structural motifs are ubiquitous in bioactive molecules, serving as critical pharmacophores that enhance metabolic stability and lipophilicity. The patent introduces a transformative approach that bypasses the traditional reliance on hazardous azide chemistry and expensive transition metal catalysts. By leveraging a base-promoted cyclization between trifluoroethylimidoyl chlorides and diazo compounds, this technology offers a streamlined pathway that addresses both safety concerns and cost efficiency. For R&D directors and procurement managers alike, understanding this shift from copper-catalyzed click chemistry to metal-free base promotion is essential for optimizing supply chains and reducing the overall cost of goods sold in complex API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has been dominated by two primary methodologies, both of which present significant logistical and safety hurdles for large-scale production. The first common route involves copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation. This process is inherently problematic because organic azides are notoriously toxic and possess high explosion risks, requiring specialized infrastructure and rigorous safety protocols that drive up operational costs. Furthermore, the reliance on copper catalysts introduces the risk of heavy metal contamination, necessitating additional downstream purification steps to meet the strict ppm limits imposed by regulatory bodies for pharmaceutical ingredients. The second conventional method utilizes organocatalytic 1,3-dipolar cycloaddition involving azides and trifluoromethyl ketones, which similarly suffers from the safety liabilities associated with azide handling. These legacy methods create bottlenecks in the supply chain, limiting the ability of manufacturers to rapidly scale production without compromising on safety or purity standards.
The Novel Approach
In stark contrast to these hazardous legacy protocols, the methodology described in patent CN113121462B utilizes readily available and stable starting materials: trifluoroethylimidoyl chlorides and diazo compounds. This innovative route operates under metal-free conditions, utilizing cesium carbonate as a simple, inexpensive base promoter. The elimination of transition metals not only simplifies the reaction workup but also ensures a cleaner impurity profile, which is a critical metric for quality control in fine chemical synthesis. Moreover, by avoiding the use of explosive azides, the process significantly lowers the barrier to entry for commercial scale-up, allowing facilities to operate with standard safety equipment rather than specialized blast-proof reactors. This shift represents a paradigm change in how these valuable heterocycles are manufactured, prioritizing operator safety and process robustness without sacrificing yield or substrate scope.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic elegance of this transformation lies in its simplicity and atom economy. The reaction is hypothesized to proceed through a base-promoted intermolecular nucleophilic addition-elimination sequence. Initially, the diazo compound acts as a nucleophile, attacking the electrophilic carbon of the trifluoroethylimidoyl chloride. This step facilitates the formation of a critical carbon-carbon bond while eliminating a chloride ion. Following this initial coupling, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This cyclization step is driven by the nucleophilic attack of the terminal nitrogen of the diazo moiety onto the imine carbon, closing the five-membered triazole ring. The use of cesium carbonate is particularly strategic; as a mild yet effective base, it deprotonates the reactive intermediates sufficiently to drive the cycle forward without causing decomposition of the sensitive diazo functionality. This mechanistic pathway avoids the formation of metal-carbene species typical in copper catalysis, thereby sidestepping potential side reactions such as dimerization or non-selective C-H insertion that often plague metal-mediated processes.
From an impurity control perspective, this mechanism offers distinct advantages. The absence of metal catalysts eliminates the formation of metal-complexed byproducts, which are often difficult to separate and can act as degradation initiators in the final drug product. Additionally, the reaction demonstrates excellent functional group tolerance, accommodating various substituents on both the aryl ring of the imidoyl chloride and the diazo component. Experimental data from the patent indicates that electron-donating and electron-withdrawing groups are well-tolerated, yielding products with high purity after standard silica gel chromatography. The robustness of this mechanism ensures that even with diverse substrate libraries, the impurity profile remains predictable and manageable, facilitating easier regulatory filing and validation for pharmaceutical applications.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it highly attractive for process chemistry teams looking to transfer technology from the lab to the pilot plant. The protocol requires mixing the trifluoroethylimidoyl chloride, the diazo compound, and cesium carbonate in an aprotic organic solvent, with acetonitrile being the preferred medium due to its ability to solubilize all reagents effectively. The reaction is typically conducted at moderate temperatures ranging from 50°C to 70°C, which balances reaction rate with energy consumption. Upon completion, usually within 8 to 16 hours, the workup involves simple filtration to remove inorganic salts followed by concentration. The crude product is then purified via column chromatography to afford the target triazole in high purity. This straightforward workflow minimizes unit operations and reduces the overall processing time.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via column chromatography to obtain the final triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route translates into tangible strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the raw material supply chain. Traditional azide-based routes require sourcing specialized, often regulated precursors that can be subject to supply disruptions due to safety transport restrictions. In contrast, the diazo compounds and imidoyl chlorides utilized in this patent are derived from commodity chemicals like aromatic amines and acid chlorides, which are abundantly available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency and enhances negotiation leverage for pricing. Furthermore, the stability of these starting materials allows for bulk purchasing and long-term storage without the degradation issues associated with unstable azides, enabling better inventory management and cost planning.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is substantial. By eliminating the need for expensive copper catalysts and specialized ligands, the direct material cost per kilogram of product is significantly lowered. More importantly, the removal of heavy metals from the process flow eradicates the need for costly scavenging resins or complex extraction procedures designed to reduce metal content to ppb levels. This simplification of the downstream processing reduces solvent consumption, waste generation, and labor hours, collectively driving down the manufacturing cost. Additionally, the use of cesium carbonate, a relatively inexpensive inorganic base, further contributes to a favorable cost structure compared to precious metal catalytic systems.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain reliability. Processes involving explosive azides often face stricter regulatory scrutiny and longer lead times for facility approvals and audits. By adopting a safer, azide-free methodology, manufacturers can accelerate the timeline for production slot allocation and reduce the administrative burden associated with hazardous material handling. This agility allows for faster response times to market demand fluctuations. The robustness of the reaction conditions, which tolerate a wide range of temperatures and solvents, also means that production is less susceptible to minor variations in utility supplies, ensuring consistent output and on-time delivery performance for downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental challenges, particularly regarding waste disposal. This novel synthesis generates minimal hazardous waste compared to traditional methods. The absence of heavy metal residues simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. The reaction operates under mild thermal conditions, reducing energy consumption for heating and cooling. These factors align perfectly with modern green chemistry principles and corporate sustainability goals. The ability to easily scale this reaction from gram to kilogram scales, as demonstrated in the patent examples, provides a clear path for commercial expansion without the need for massive capital investment in new safety infrastructure, ensuring long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is this new synthesis method safer than traditional copper-catalyzed routes?
A: Traditional methods often rely on toxic and explosive organic azides. This novel protocol utilizes stable diazo compounds and imidoyl chlorides, eliminating the safety hazards associated with handling high-energy azide intermediates.
Q: What are the optimal reaction conditions for scaling this process?
A: The patent specifies using cesium carbonate as a promoter in acetonitrile at temperatures between 50°C and 70°C. This mild condition set facilitates easier thermal management during commercial scale-up compared to cryogenic or high-pressure alternatives.
Q: Does this method require expensive transition metal catalysts?
A: No, the process is entirely metal-free. It relies on a base-promoted mechanism, which removes the need for costly copper catalysts and the subsequent rigorous purification steps required to remove trace metal residues from pharmaceutical products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemical market. Our technical team has thoroughly analyzed the potential of the metal-free cyclization route described in patent CN113121462B and is prepared to leverage this technology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole intermediate meets the highest industry standards for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss your project needs,索取 specific COA data for our available intermediates, and review our comprehensive route feasibility assessments. Let us help you secure a reliable, safe, and cost-effective supply of these vital heterocyclic building blocks for your next generation of therapeutic or agricultural products.
