Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
Introduction to Next-Generation Quinolinone Synthesis
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and sustainable pathways to access biologically active scaffolds. A significant breakthrough in this domain is detailed in patent CN113045489B, which discloses a novel preparation method for 3-arylquinoline-2(1H) ketone derivatives. These quinolinone cores are ubiquitous in medicinal chemistry, serving as critical structural motifs in a wide array of therapeutic agents ranging from antibiotics and antiplatelet drugs to potent antitumor compounds and endothelin receptor antagonists. The traditional reliance on multi-step syntheses or harsh reagents has often bottlenecked the supply chain for these valuable intermediates. However, this new methodology leverages a palladium-catalyzed aminocarbonylation strategy that utilizes benzisoxazole not merely as a substrate, but ingeniously as a dual source of both nitrogen and the formyl group. This innovation drastically simplifies the synthetic logic, offering a streamlined route that aligns perfectly with the needs of modern pharmaceutical development for speed and reliability.
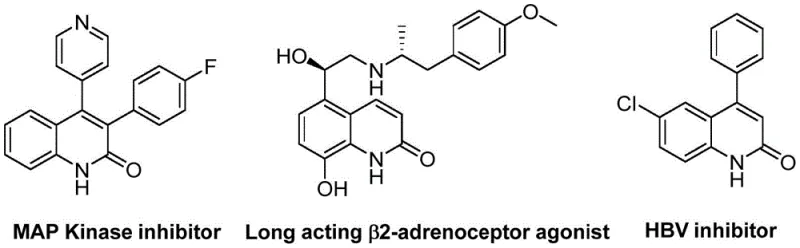
As illustrated in the structural diversity of bioactive molecules, the ability to rapidly construct the quinolinone core with various aryl substitutions is paramount for drug discovery teams. The patent highlights that this method tolerates a broad spectrum of functional groups, allowing chemists to introduce diverse substituents without compromising the integrity of the sensitive heterocyclic ring. For a reliable pharmaceutical intermediate supplier, mastering such versatile chemistry is essential to support the rapid iteration required in lead optimization phases. By shifting away from classical condensation reactions towards this transition-metal catalyzed approach, manufacturers can offer clients a more robust platform for generating libraries of quinolinone derivatives, ultimately accelerating the timeline from benchtop discovery to clinical candidate selection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)one derivatives has relied heavily on classical named reactions such as the Vilsmeier-Haack, Knorr, and Friedlander condensations. While these methods are well-established in academic literature, they often suffer from significant drawbacks when translated to industrial settings. For instance, the Vilsmeier-Haack reaction typically requires the use of phosphorus oxychloride and DMF under strictly anhydrous conditions, generating substantial amounts of corrosive waste and posing severe safety hazards during scale-up. Furthermore, the Friedlander condensation, although atom-economical in theory, often demands high temperatures and strong acidic or basic catalysts that can degrade sensitive functional groups present on the starting aniline or ketone substrates. These harsh conditions frequently lead to complex impurity profiles, necessitating rigorous and costly purification steps that erode overall process efficiency. Additionally, many traditional routes require pre-functionalized starting materials that are themselves expensive or difficult to source, creating supply chain vulnerabilities for procurement managers seeking cost-effective solutions.
The Novel Approach
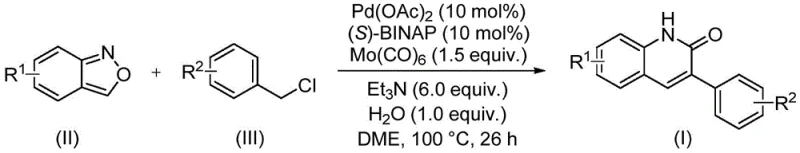
In stark contrast, the novel approach described in the patent utilizes a palladium-catalyzed carbonylation coupling reaction that operates under significantly milder and more controlled conditions. By employing benzisoxazole and benzyl chloride compounds as the primary building blocks, the reaction circumvents the need for hazardous gaseous carbon monoxide, instead utilizing molybdenum hexacarbonyl as a safe, solid CO surrogate. This substitution alone represents a major safety advancement, removing the requirement for specialized high-pressure equipment typically associated with carbonylation reactions. The reaction proceeds smoothly at 100°C in ethylene glycol dimethyl ether (DME), a solvent that effectively dissolves the reactants while maintaining thermal stability. The use of a chiral ligand, (S)-1,1'-binaphthyl-2,2'-bisdiphenylphosphine, alongside palladium acetate ensures high catalytic activity and selectivity, minimizing the formation of side products. This methodological shift not only enhances the safety profile of the manufacturing process but also improves the overall yield and purity of the final 3-arylquinolin-2(1H) ketone derivatives, making it a superior choice for commercial production.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The core of this technological advancement lies in the intricate catalytic cycle facilitated by the palladium complex. The reaction initiates with the oxidative addition of the benzyl chloride to the active Pd(0) species generated in situ from palladium acetate and the chiral phosphine ligand. This step forms a benzyl-palladium(II) intermediate, which is then poised for carbonyl insertion. Uniquely, the carbonyl source is derived from the decomposition of molybdenum hexacarbonyl under the reaction conditions, releasing CO which subsequently inserts into the Pd-C bond to form an acyl-palladium species. Simultaneously, the benzisoxazole undergoes a ring-opening process, likely facilitated by the basic environment provided by triethylamine and the presence of water, releasing the necessary nitrogen nucleophile. This nitrogen species then attacks the acyl-palladium complex, followed by reductive elimination to forge the new C-N bond and close the quinolinone ring, regenerating the Pd(0) catalyst to continue the cycle. This elegant mechanism allows for the concurrent formation of the heterocyclic ring and the installation of the aryl group in a single pot operation.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature thermal cyclizations. The specificity of the palladium catalyst ensures that the carbonylation occurs selectively at the benzylic position, avoiding random acylation of the aromatic rings which is a common issue in Friedel-Crafts type reactions. Furthermore, the use of water as a co-reagent in precise stoichiometric amounts (1.0 equivalent) aids in the hydrolysis of the intermediate imine or nitrile species without promoting excessive hydrolysis of the final lactam product. The broad functional group tolerance observed, accommodating groups such as cyano, trifluoromethyl, methoxy, and halogens, suggests that the catalytic cycle is robust against electronic variations in the substrate. This robustness is critical for R&D directors who need to ensure that the synthetic route remains viable even as the molecular structure evolves during the drug development process, thereby reducing the risk of late-stage process failures.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction parameters to maximize the benefits of this patented technology. The process is designed to be operationally simple, utilizing commercially available starting materials that do not require complex pre-synthesis or purification. The standard protocol involves charging a sealed reaction vessel with the palladium catalyst system, the solid CO source, the base, and the two key organic substrates in DME solvent. Maintaining the temperature at 100°C for a duration of 26 hours is critical to drive the reaction to full conversion, as shorter times may result in incomplete consumption of the benzyl chloride, while excessively long times offer diminishing returns and increased energy costs. Following the reaction, the workup procedure is straightforward, involving filtration to remove metal residues and inorganic salts, followed by standard silica gel chromatography to isolate the pure product. Detailed standardized synthesis steps see the guide below.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in a sealed tube with DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant reduction of raw material complexity and associated costs. By utilizing benzisoxazole and benzyl chlorides, which are commodity chemicals available from multiple global suppliers, the dependency on bespoke or exotic reagents is eliminated. This commoditization of inputs enhances supply chain resilience, ensuring that production schedules are not disrupted by the shortage of a single niche precursor. Moreover, the replacement of gaseous carbon monoxide with a solid surrogate like molybdenum hexacarbonyl removes the need for specialized gas handling infrastructure and safety protocols, leading to substantial capital expenditure savings and lower operational overheads. The simplified downstream processing further contributes to cost efficiency by reducing solvent consumption and waste disposal fees, aligning with increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the high atom economy and the use of inexpensive catalysts relative to the value of the final product. Since the benzisoxazole acts as a dual-purpose reagent, the total number of reactants required per mole of product is reduced, directly lowering the bill of materials. Additionally, the high yields reported, often exceeding 90% for optimized substrates, mean that less starting material is wasted, and the throughput of the manufacturing facility is maximized. The elimination of cryogenic conditions or ultra-high pressure reactors further reduces utility costs, making the cost reduction in pharmaceutical intermediate manufacturing substantial and sustainable over the long term without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the reaction conditions and the availability of reagents. Unlike processes that rely on moisture-sensitive organometallics or unstable intermediates, this method tolerates the presence of water and uses stable solids, simplifying storage and logistics. The broad substrate scope means that a single manufacturing line can be adapted to produce a wide variety of quinolinone derivatives simply by swapping the benzyl chloride input, providing flexibility to respond to changing market demands. This adaptability reduces lead time for high-purity pharmaceutical intermediates, allowing clients to accelerate their own development timelines with confidence in a steady supply of critical building blocks.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or tonne levels is facilitated by the homogeneous nature of the catalytic system and the moderate temperature requirements. The absence of hazardous gases and the use of relatively benign solvents like DME simplify the engineering controls required for large-scale reactors. From an environmental standpoint, the process generates less hazardous waste compared to traditional methods involving phosphorus halides or strong mineral acids. The ability to recover and potentially recycle the palladium catalyst, although not explicitly detailed in the patent, is a standard practice in such systems that further enhances the green chemistry profile. This alignment with environmental compliance standards makes the commercial scale-up of complex heterocyclic intermediates more feasible and socially responsible.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the practical implementation and scope of this synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for process chemists. Understanding the nuances of catalyst loading, substrate compatibility, and purification requirements is essential for successful technology transfer. The following questions address the most critical aspects of adopting this palladium-catalyzed route for the production of quinolinone derivatives.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl source, eliminating the need for separate carbonylation reagents like toxic carbon monoxide gas and simplifying the reaction stoichiometry significantly.
Q: What is the typical yield range for this palladium-catalyzed method?
A: The patented process demonstrates high reaction efficiency, with isolated yields frequently exceeding 90% for various substrates, including those with electron-withdrawing or electron-donating groups.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the use of commercially available starting materials, mild reaction conditions (100°C), and straightforward post-processing involving filtration and chromatography makes this route highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality intermediates play in the success of drug development programs. Our team of expert chemists has extensively evaluated the patented palladium-catalyzed aminocarbonylation route and confirmed its potential for delivering superior results in terms of purity and yield. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need grams for initial screening or tonnes for clinical trials, our capacity is ready to meet your demands. Our state-of-the-art facilities are equipped with rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of 3-arylquinolin-2(1H)-one derivative meets the highest industry standards for impurity profiles and physical properties.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific target molecule, exploring how this efficient route can optimize your budget. We encourage you to reach out today to obtain specific COA data for our existing library of quinolinone derivatives or to discuss route feasibility assessments for novel analogs. Let us be your trusted partner in transforming complex chemical challenges into commercial realities, driving your pipeline forward with speed and precision.
