Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Applications
Introduction to Advanced Triazole Synthesis Technology
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign synthetic routes. A pivotal development in this sector is documented in patent CN113105402B, which discloses a novel preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds. These heterocyclic scaffolds are critical structural motifs found in a wide array of bioactive molecules, including blockbuster drugs such as Maraviroc, Sitagliptin, and Deferasirox, as illustrated in the structural diversity shown below. The ability to efficiently construct these cores, particularly those bearing both trifluoromethyl and acyl groups, represents a significant technological breakthrough for medicinal chemists and process engineers alike.
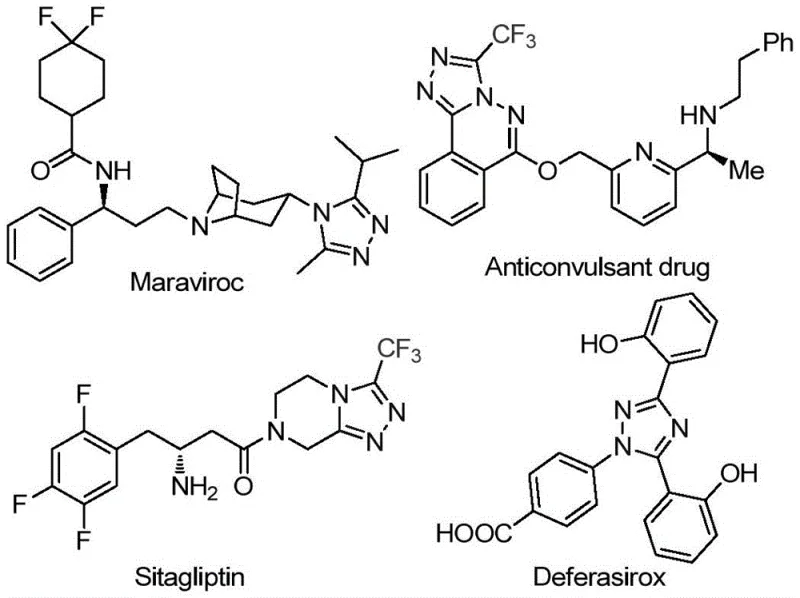
This patented methodology addresses long-standing challenges in heterocyclic chemistry by eliminating the reliance on precious metal catalysts and harsh reaction conditions. For a reliable pharmaceutical intermediate supplier, adopting such a robust protocol means enhancing the purity profile of the final product while simultaneously streamlining the supply chain. The introduction of a trifluoromethyl group into the triazole ring is particularly valuable, as it markedly improves the metabolic stability, lipophilicity, and bioavailability of the parent compound, making this synthesis route highly attractive for the development of next-generation active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles has often relied on transition metal catalysis or multi-step sequences that suffer from low atom economy and complex workup procedures. Traditional approaches frequently necessitate the use of expensive palladium or copper catalysts, which not only inflate the raw material costs but also introduce the risk of heavy metal contamination in the final product. For procurement managers, this translates into higher costs for metal scavenging resins and more rigorous quality control testing to ensure compliance with strict ICH Q3D guidelines regarding elemental impurities. Furthermore, many conventional routes require stringent anhydrous and oxygen-free conditions, demanding specialized equipment like Schlenk lines or gloveboxes, which limits scalability and increases the capital expenditure required for manufacturing facilities.
The Novel Approach
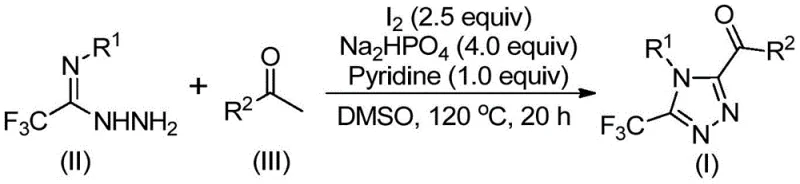
In stark contrast, the method disclosed in CN113105402B utilizes a metal-free, iodine-promoted strategy that operates under relatively mild and operationally simple conditions. As depicted in the general reaction scheme below, the process leverages the oxidative power of elemental iodine in dimethyl sulfoxide (DMSO) to facilitate the transformation. This approach bypasses the need for toxic heavy metals entirely, thereby removing a major bottleneck in downstream processing. The reaction tolerates a wide range of functional groups on both the aryl ketone and the hydrazide components, allowing for the rapid generation of diverse chemical libraries. This flexibility is crucial for R&D teams aiming to optimize lead compounds, as it enables the facile exploration of structure-activity relationships (SAR) without being constrained by incompatible protecting groups or sensitive functionalities.
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of oxidation and condensation reactions, centered around the unique reactivity of iodine in polar aprotic solvents. The process likely initiates with an iodine-mediated Kornblum oxidation of the aryl ethyl ketone substrate. In the presence of DMSO, the ketone is oxidized to an aryl diketone intermediate, a key species that sets the stage for ring closure. This oxidative step is critical, as it generates the necessary electrophilic centers required for the subsequent nucleophilic attack by the nitrogen atoms of the trifluoroethylimide hydrazide. The use of DMSO is not merely as a solvent but as an active participant in the oxygen transfer process, ensuring high conversion rates without the need for external oxidants like peroxides or hypervalent iodine reagents.
Following the formation of the diketone, the reaction proceeds through a dehydration condensation with the trifluoroethylimide hydrazide to form a hydrazone intermediate. Under the continued influence of iodine and the basic environment provided by pyridine and sodium dihydrogen phosphate, this intermediate undergoes an intramolecular cyclization. This final ring-closing step constructs the 1,2,4-triazole core while simultaneously installing the trifluoromethyl group at the 3-position and the acyl group at the 5-position. The precise control over regioselectivity is a hallmark of this method, ensuring that the desired 3,4,5-substitution pattern is obtained with high fidelity. This mechanistic clarity allows process chemists to fine-tune reaction parameters, such as temperature and stoichiometry, to maximize yield and minimize the formation of regioisomeric impurities.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazoles Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the sequential addition of reagents and temperature control to ensure optimal outcomes. The protocol is designed to be user-friendly, avoiding the complexities associated with air-sensitive chemistry. By adhering to the specific molar ratios and thermal profiles outlined in the patent, manufacturers can achieve consistent results across different batch sizes. The following guide summarizes the critical operational steps derived from the patent data, serving as a foundational reference for process development teams looking to adopt this technology for cost reduction in pharmaceutical intermediate manufacturing.
- Combine aryl ethyl ketone and elemental iodine in dimethyl sulfoxide (DMSO) and heat to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the mixture to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iodine-promoted synthesis method offers profound advantages that extend beyond mere chemical efficiency. For supply chain heads and procurement managers, the shift towards metal-free catalysis represents a strategic opportunity to de-risk the supply of critical intermediates. The elimination of precious metals removes a significant variable cost component and mitigates the supply volatility associated with commodities like palladium. Furthermore, the simplified purification process reduces the consumption of silica gel and solvents during chromatography, leading to substantial waste reduction and lower disposal costs. This aligns perfectly with modern green chemistry initiatives and helps companies meet increasingly stringent environmental regulations without compromising on production speed.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive catalysts with inexpensive elemental iodine and the use of commodity chemicals like aryl ketones and DMSO. By eliminating the need for costly metal scavengers and reducing the number of purification steps, the overall cost of goods sold (COGS) is significantly lowered. Additionally, the high atom economy of the reaction ensures that a larger proportion of the starting materials are incorporated into the final product, minimizing waste and maximizing resource utilization. This efficiency translates directly into improved profit margins for manufacturers and more competitive pricing for downstream clients seeking high-purity intermediates.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to a more resilient supply chain. Since the reaction does not require strictly anhydrous or oxygen-free conditions, it can be performed in standard reactor vessels without the need for specialized inert atmosphere infrastructure. This ease of operation facilitates faster technology transfer between sites and reduces the likelihood of batch failures due to environmental factors. Moreover, the starting materials, such as aryl ethyl ketones and trifluoroethylimide hydrazides, are commercially available and widely sourced, reducing the risk of raw material shortages. This reliability ensures consistent delivery schedules, which is paramount for maintaining continuous API production lines.
- Scalability and Environmental Compliance: Scalability is a key strength of this methodology, as evidenced by its successful demonstration from gram to multi-gram scales in the patent examples. The use of DMSO, a high-boiling polar solvent, allows for reactions to be run at elevated temperatures safely, promoting faster kinetics and shorter cycle times. From an environmental standpoint, the absence of heavy metals simplifies the treatment of effluent streams, making it easier to comply with wastewater discharge standards. The reduction in hazardous waste generation not only lowers disposal fees but also enhances the company's sustainability profile, a factor that is increasingly important to investors and regulatory bodies alike.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis technology, we have compiled a set of frequently asked questions based on the detailed specifications provided in the patent documentation. These insights address common concerns regarding reaction safety, impurity profiles, and scale-up potential. Understanding these nuances is essential for technical decision-makers who are evaluating new synthetic routes for integration into their existing manufacturing portfolios. The answers provided below reflect the specific advantages and operational parameters defined in the intellectual property.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the process described in patent CN113105402B utilizes elemental iodine as a promoter instead of toxic or expensive heavy metal catalysts, significantly simplifying downstream purification and reducing environmental impact.
Q: What are the typical reaction conditions for this triazole formation?
A: The reaction proceeds in dimethyl sulfoxide (DMSO) solvent. It involves a two-stage heating process: initially at 90-110°C for oxidation, followed by 110-130°C for cyclization, without the need for strict anhydrous or oxygen-free conditions.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method is easily scalable from gram levels to industrial production due to the use of cheap, commercially available raw materials and simple operational procedures that do not require specialized inert atmosphere equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN113105402B for the production of high-value pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Whether you require custom synthesis of complex triazole derivatives or large-scale supply of established intermediates, our infrastructure is designed to meet the exacting demands of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecular targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your drug development timeline with our reliable and efficient manufacturing capabilities.
