Advanced Rhodium-Catalyzed Synthesis of Acetamide Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking sustainable pathways to construct essential amide bonds, which are ubiquitous motifs in active pharmaceutical ingredients (APIs) and agrochemicals. A groundbreaking approach detailed in Chinese Patent CN112812032B introduces a highly efficient rhodium-catalyzed aminocarbonylation strategy that fundamentally shifts the paradigm from hazardous traditional methods to green chemistry principles. This innovative protocol utilizes dimethyl carbonate (DMC) not merely as a benign solvent but critically as the sole carbon monoxide and methyl source, reacting directly with readily available nitro compounds. By eliminating the need for pre-functionalized amines or toxic carbonyl sources, this technology offers a streamlined, atom-economical route to diverse acetamide derivatives. For R&D directors and process chemists, this represents a significant leap forward in synthetic efficiency, providing a robust platform for generating high-purity intermediates while adhering to increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of acetamide scaffolds has relied heavily on classical methylation and carbonylation strategies that pose severe safety and environmental challenges. Traditional methylating agents such as diazomethane, dimethyl sulfate, and methyl iodide are fraught with hazards; diazomethane is notoriously explosive, while dimethyl sulfate and methyl iodide are highly toxic, corrosive, and carcinogenic. Furthermore, these conventional pathways typically necessitate the use of stoichiometric amounts of strong bases to drive the reaction, resulting in the generation of substantial quantities of inorganic salt waste that requires costly disposal and treatment. In the realm of carbonylation, processes like the Monsanto acetic acid synthesis often employ hydroiodic acid (HI) as a co-catalyst, which induces severe corrosion in reactor equipment, thereby increasing maintenance costs and limiting operational longevity. These legacy methods create significant bottlenecks for procurement and supply chain teams due to the complex handling requirements, regulatory burdens associated with hazardous waste, and the volatility of raw material pricing for specialized reagents.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112812032B leverages the unique reactivity of dimethyl carbonate under rhodium catalysis to overcome these entrenched limitations. By employing DMC as a dual-function reagent—acting simultaneously as the C1 building block and the reaction medium—the process eliminates the need for external carbon monoxide gas or toxic methyl halides. The use of nitro compounds as nitrogen surrogates further simplifies the supply chain, as nitroarenes are generally more stable, cheaper, and more widely available than their corresponding aniline counterparts. This novel approach operates under relatively mild thermal conditions without the requirement for corrosive acidic promoters, thereby preserving reactor integrity and reducing equipment depreciation. The transition to this green chemistry platform allows manufacturers to drastically simplify their operational workflows, removing the burden of neutralizing stoichiometric base waste and enabling a cleaner, more sustainable production cycle that aligns with modern ESG (Environmental, Social, and Governance) goals.
Mechanistic Insights into Rh-Catalyzed Aminocarbonylation
The core of this technological advancement lies in the sophisticated catalytic cycle driven by a dirhodium tetracarbonyl dichloride precursor in conjunction with a bidentate phosphine ligand, specifically 1,3-bis(diphenylphosphine)propane (DPPP). The reaction mechanism initiates with the activation of the nitro compound, which undergoes reduction and subsequent carbonylation in situ. Tungsten hexacarbonyl [W(CO)6] serves as a solid CO surrogate, releasing carbon monoxide slowly to maintain a safe pressure profile within the reactor, while sodium iodide acts as a crucial promoter to facilitate the activation of the dimethyl carbonate. The rhodium center coordinates with the nitro substrate and the in-situ generated CO species, orchestrating the formation of the amide bond through a series of migratory insertion and reductive elimination steps. This intricate interplay ensures high turnover frequencies and exceptional selectivity, minimizing the formation of side products such as ureas or over-methylated species that often plague less controlled systems.
One of the most compelling aspects of this catalytic system is its remarkable tolerance for a broad spectrum of functional groups, a critical factor for R&D teams designing complex molecular architectures. The mild nature of the rhodium-DPPP catalyst system allows for the successful transformation of substrates bearing sensitive moieties including halogens (fluorine, chlorine, bromine), electron-withdrawing trifluoromethyl groups, and various alkoxy substituents without compromising yield or purity. As illustrated in the general reaction scheme below, the process seamlessly converts diverse nitroarenes and aliphatic nitro compounds into their corresponding acetamides with high efficiency. This robustness implies that the impurity profile of the final product is significantly cleaner compared to harsher acidic or basic methods, reducing the burden on downstream purification units and ensuring that the final API intermediate meets stringent quality specifications required by global regulatory bodies.
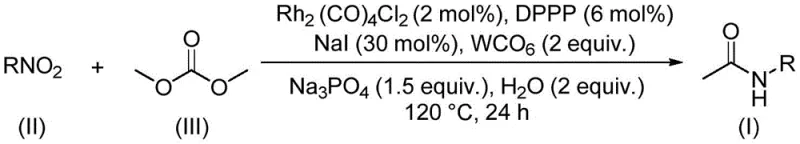
How to Synthesize Acetamide Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting involves a straightforward procedure that balances reagent stoichiometry with thermal control to maximize yield. The standard protocol dictates charging a sealed reaction vessel with the rhodium catalyst (2 mol%), DPPP ligand (6 mol%), tungsten carbonyl (2 equivalents), sodium phosphate (1.5 equivalents), sodium iodide (30 mol%), and a small amount of water (2 equivalents) alongside the nitro substrate. Dimethyl carbonate is added in excess to serve as both the reactant and the solvent, ensuring adequate solubility of all components. The mixture is then heated to 120°C and maintained for approximately 24 hours to drive the reaction to completion. Following the reaction, the workup is remarkably simple, involving filtration to remove insoluble salts followed by silica gel chromatography, although on a commercial scale, crystallization or distillation would be optimized to replace chromatography for cost efficiency.
- Combine dirhodium tetracarbonyl dichloride, DPPP ligand, tungsten carbonyl, sodium phosphate, sodium iodide, water, nitro compound, and dimethyl carbonate in a sealed tube.
- Heat the reaction mixture to 120°C and maintain stirring for 24 hours to ensure complete conversion.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target acetamide compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed DMC methodology presents a transformative opportunity to optimize cost structures and mitigate supply risks. The primary economic driver is the substitution of expensive, hazardous, and logistically challenging reagents with commodity chemicals. Dimethyl carbonate is produced on a massive industrial scale and is priced significantly lower than specialized methylating agents, while nitro compounds are ubiquitous bulk chemicals with stable supply chains. By eliminating the need for stoichiometric bases and the subsequent waste treatment associated with inorganic salts, the process inherently reduces the variable costs related to effluent disposal and environmental compliance. Furthermore, the avoidance of corrosive additives like hydroiodic acid extends the lifespan of reactor vessels and piping, leading to substantial long-term capital expenditure savings on equipment maintenance and replacement.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the convergence of cheap raw materials and simplified downstream processing. Since dimethyl carbonate acts as the solvent, there is no need to purchase and recover separate volatile organic solvents, which reduces both material costs and energy consumption during solvent removal. The high atom economy of the reaction means that a larger proportion of the input mass ends up in the final product, minimizing raw material waste. Additionally, the use of a heterogeneous-like workup (filtration of salts) simplifies the isolation process, potentially allowing for the elimination of complex extraction sequences. These factors collectively contribute to a significantly reduced cost of goods sold (COGS), making the final acetamide intermediates more competitive in the global market without sacrificing quality.
- Enhanced Supply Chain Reliability: Relying on hazardous reagents like diazomethane or methyl iodide often introduces fragility into the supply chain due to strict transportation regulations and limited supplier availability. In contrast, dimethyl carbonate and nitro compounds are classified as safer chemicals with widespread global production, ensuring a continuous and reliable flow of materials even during market disruptions. The stability of these starting materials also allows for longer storage times without degradation, enabling manufacturers to maintain strategic stockpiles without the risk of spoilage. This resilience is crucial for maintaining consistent production schedules and meeting the just-in-time delivery demands of downstream pharmaceutical clients, thereby strengthening the overall reliability of the supply network.
- Scalability and Environmental Compliance: From a scale-up perspective, the absence of gaseous carbon monoxide cylinders and explosive reagents makes this process inherently safer and easier to scale from kilogram to multi-ton production. The reaction conditions (120°C) are well within the operating range of standard glass-lined or stainless steel reactors, requiring no specialized high-pressure equipment. Environmentally, the process aligns perfectly with green chemistry principles by utilizing a biodegradable solvent and generating minimal waste, which simplifies the permitting process for new manufacturing lines. This environmental compatibility reduces the regulatory burden and accelerates the time-to-market for new products, providing a strategic advantage in regions with strict environmental enforcement.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on catalyst performance, substrate scope, and process safety. Understanding these nuances is essential for process engineers and technical buyers evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What are the advantages of using dimethyl carbonate over traditional methylating agents?
A: Dimethyl carbonate serves as a non-toxic, biodegradable alternative to hazardous reagents like diazomethane or dimethyl sulfate, acting simultaneously as a green solvent and C1 source without generating stoichiometric salt waste.
Q: Does this rhodium-catalyzed method tolerate diverse functional groups?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully accommodating substrates with halogens, trifluoromethyl groups, alkoxy groups, and heterocycles with high yields.
Q: Is the catalyst system cost-effective for large-scale production?
A: The method utilizes commercially available and relatively inexpensive rhodium precursors like dirhodium tetracarbonyl dichloride at low loading (2 mol%), making it economically viable for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acetamide Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this rhodium-catalyzed aminocarbonylation technology in reshaping the production landscape for acetamide intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-scale operations. Our state-of-the-art facilities are equipped to handle the specific thermal and catalytic requirements of this process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards of the global pharmaceutical industry. We are committed to delivering high-purity acetamide compounds that serve as robust building blocks for next-generation therapeutics.
We invite procurement leaders and R&D innovators to collaborate with us to leverage this advanced synthetic route for your specific project needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this green chemistry approach can optimize your budget. We encourage you to reach out today to request specific COA data for our acetamide portfolio and to discuss detailed route feasibility assessments that can accelerate your development timelines and secure your supply chain for the future.
