Scalable Production of 2-Trifluoromethyl Benzimidazoles Using Recyclable Heterogeneous Copper Catalysts
Introduction to Advanced Benzimidazole Synthesis
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, particularly benzimidazoles, which serve as critical scaffolds in numerous bioactive molecules and functional materials. As detailed in the recent patent literature, specifically CN113429349A, a significant breakthrough has been achieved in the preparation of 2-trifluoromethyl substituted benzimidazole compounds through a novel heterogeneous catalytic approach. This technology addresses long-standing challenges in organic synthesis by replacing traditional homogeneous catalytic systems with a recyclable copper-doped carbon nitride catalyst, thereby enhancing both the economic and environmental viability of the process. The introduction of the trifluoromethyl group is strategically vital, as it significantly modulates the physicochemical properties of the parent molecule, improving lipophilicity, metabolic stability, and bioavailability, which are paramount parameters for drug candidates targeting central nervous system disorders or infectious diseases.
Furthermore, the versatility of this synthetic route allows for the precise engineering of molecular structures to meet specific therapeutic requirements, making it an invaluable tool for medicinal chemists and process development teams alike. By leveraging mild reaction conditions ranging from 70°C to 90°C and utilizing readily available starting materials such as trifluoroethylimidoyl chloride and various amines, this method represents a paradigm shift towards more sustainable and efficient manufacturing protocols. The ability to synthesize a broad spectrum of derivatives with high reaction efficiency underscores the practical applicability of this invention, positioning it as a cornerstone technology for the reliable production of high-value pharmaceutical intermediates on a commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted benzimidazoles has relied heavily on methods that suffer from significant operational and economic drawbacks, hindering their widespread adoption in large-scale manufacturing. Traditional routes often involve the condensation of o-amino aniline with trifluoroacetic acid or aldehyde, reductive cyclization of o-nitroaniline, or direct trifluoromethylation using expensive and hazardous reagents. These conventional pathways frequently necessitate severe reaction conditions, such as high temperatures or strong acidic environments, which can lead to poor selectivity, decomposition of sensitive functional groups, and the generation of complex impurity profiles that are difficult to remove. Moreover, earlier reports utilizing homogeneous copper catalysts, while effective in small-scale laboratory settings, present formidable challenges in industrial applications due to the difficulty in separating the catalyst from the final product, leading to potential heavy metal contamination and increased purification costs.
The Novel Approach
In stark contrast to these legacy methods, the innovative protocol disclosed in the patent utilizes a copper-doped graphitic carbon nitride (Cu/g-C3N4) heterogeneous catalyst to drive the cascade cyclization reaction with exceptional efficiency and selectivity. This novel approach facilitates the reaction between trifluoroethylimidoyl chloride and amines under mild thermal conditions, typically between 70°C and 90°C, in the presence of a base like potassium carbonate. The use of a heterogeneous catalyst not only simplifies the post-reaction workup—allowing for easy filtration and recovery of the catalyst—but also enables the catalyst to be recycled multiple times with only a marginal decrease in activity, as evidenced by retention of catalytic efficiency over three cycles. This advancement drastically reduces the environmental footprint and operational complexity associated with benzimidazole synthesis.
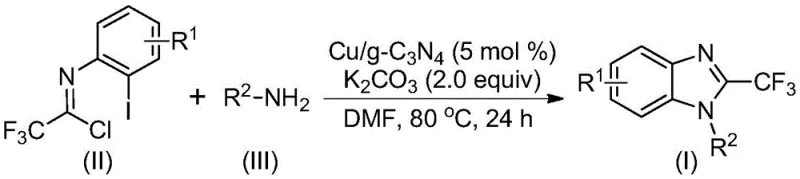
The reaction mechanism proceeds through a streamlined sequence where the trifluoroethylimidoyl chloride and amine first undergo a nucleophilic addition-elimination to form an amidine intermediate. Subsequently, the copper catalyst inserts into the carbon-iodine bond of the aryl ring, forming a transient copper complex that undergoes intramolecular cyclization upon base-mediated elimination of hydrogen iodide. This elegant mechanistic pathway avoids the harsh reagents and extreme conditions of the past, offering a cleaner, safer, and more atom-economical route to these valuable heterocyclic compounds. The generality of this method is further highlighted by its tolerance to various substituents, allowing for the modular construction of diverse libraries of benzimidazole derivatives essential for modern drug discovery programs.
Mechanistic Insights into Cu/g-C3N4 Catalyzed Cyclization
The core of this technological advancement lies in the unique properties of the copper-doped carbon nitride catalyst, which acts as a robust platform for facilitating the C-N bond formation required for benzimidazole ring closure. The graphitic carbon nitride support provides a high surface area and excellent thermal stability, ensuring that the active copper species remain dispersed and accessible throughout the reaction duration. Mechanistically, the copper centers coordinate with the iodine atom of the substrate, activating the carbon-iodine bond for oxidative addition. This step is critical as it generates the organocopper intermediate necessary for the subsequent intramolecular nucleophilic attack by the nitrogen atom of the amidine moiety. The presence of the base, typically potassium carbonate, plays a dual role by neutralizing the generated hydrogen iodide and deprotonating the intermediate to drive the cyclization forward, ultimately yielding the 2-trifluoromethyl benzimidazole scaffold via reductive elimination.
From an impurity control perspective, the mild nature of this catalytic system is instrumental in minimizing side reactions that often plague harsher synthetic routes. The specificity of the heterogeneous catalyst ensures that the reaction proceeds primarily through the desired cyclization pathway, reducing the formation of polymeric byproducts or over-reacted species that can complicate downstream purification. The ability to tune the electronic properties of the catalyst by varying the copper loading or the carbon nitride precursor offers an additional layer of control over the reaction kinetics and selectivity. This level of mechanistic understanding allows process chemists to optimize reaction parameters precisely, ensuring consistent product quality and high purity levels that meet the stringent specifications required for pharmaceutical applications.
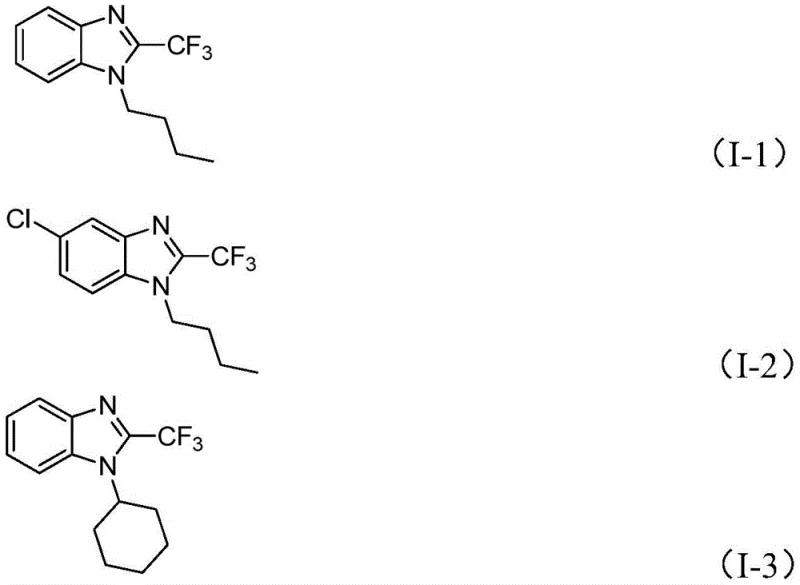
Furthermore, the structural diversity achievable through this method is exemplified by the successful synthesis of various derivatives, including those with alkyl chains like n-butyl and cyclohexyl, as well as aromatic substitutions. The compatibility with different R1 and R2 groups demonstrates the robustness of the catalytic cycle against steric and electronic variations in the substrate. For instance, the synthesis of compounds such as I-1, I-2, and I-3 showcases the method's ability to accommodate both electron-rich and electron-deficient aromatic rings without compromising yield. This broad substrate scope is a testament to the versatility of the Cu/g-C3N4 system, making it a powerful tool for generating diverse chemical libraries for biological screening and structure-activity relationship (SAR) studies in the development of new therapeutic agents.
How to Synthesize 2-Trifluoromethyl Benzimidazole Efficiently
The implementation of this synthesis route in a laboratory or pilot plant setting is straightforward and relies on standard organic synthesis techniques, making it highly accessible for process development teams. The procedure begins with the preparation of the heterogeneous catalyst, followed by the combination of the key building blocks—trifluoroethylimidoyl chloride and the chosen amine—in a polar aprotic solvent such as DMF. The reaction is driven by thermal energy and the presence of the base, requiring minimal specialized equipment beyond a standard heating mantle and filtration setup. This simplicity translates directly into reduced capital expenditure and faster turnaround times for producing gram-to-kilogram quantities of the target intermediate. For a detailed breakdown of the standardized operating procedures and specific stoichiometric ratios optimized for maximum yield, please refer to the technical guide below.
- Prepare the heterogeneous catalyst by doping copper sulfate onto graphitic carbon nitride support.
- Mix trifluoroethylimidoyl chloride, amine, potassium carbonate, and the Cu/g-C3N4 catalyst in DMF solvent.
- Heat the reaction mixture at 70-90°C for 18-30 hours, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this heterogeneous catalytic process offers compelling strategic advantages that extend far beyond mere technical feasibility. The shift from homogeneous to heterogeneous catalysis fundamentally alters the cost structure of manufacturing by eliminating the need for expensive ligand systems and complex metal scavenging processes that are typically required to meet residual metal specifications in API production. The catalyst itself is derived from inexpensive and abundant precursors like copper sulfate and melamine-derived carbon nitride, ensuring a stable and low-cost supply chain for the catalytic material. Furthermore, the recyclability of the catalyst means that the effective cost per kilogram of product decreases significantly over multiple batches, providing a clear path to margin improvement as production volumes scale up.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of costly homogeneous catalysts and the associated downstream purification steps required to remove trace metals. By utilizing a solid-supported catalyst that can be filtered off and reused, manufacturers can drastically simplify the workup procedure, reducing solvent consumption and waste disposal costs. The use of cheap, commodity chemicals like potassium carbonate and DMF further anchors the variable costs at a low level, making the overall process highly competitive compared to traditional multi-step syntheses that require cryogenic conditions or exotic reagents.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as substituted anilines and trifluoroacetic acid derivatives ensures a robust and resilient supply chain that is less susceptible to disruptions. Unlike proprietary reagents that may have single-source suppliers, the key inputs for this reaction are produced by multiple global chemical manufacturers, mitigating the risk of shortages or price volatility. Additionally, the mild reaction conditions reduce the stress on reactor equipment and lower the energy requirements for heating and cooling, contributing to more predictable and stable production schedules that can reliably meet customer demand.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns perfectly with the industry's push towards greener chemistry principles. The heterogeneous nature of the catalyst minimizes heavy metal waste, and the high atom economy of the cyclization reaction reduces the generation of organic byproducts. This results in a cleaner process that is easier to permit and operate within strict environmental regulations, facilitating smoother scale-up from pilot plants to multi-ton commercial production facilities without the need for extensive retrofitting of waste treatment infrastructure.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology for their specific applications, we have compiled a set of frequently asked questions based on the detailed experimental data and mechanistic studies provided in the patent documentation. These answers address common concerns regarding catalyst performance, substrate compatibility, and process safety, offering a clear picture of what can be expected when implementing this synthesis route in a GMP environment. Understanding these nuances is critical for project planning and risk assessment, ensuring that all stakeholders are aligned on the technical capabilities and limitations of the proposed manufacturing strategy.
Q: What are the advantages of using Cu/g-C3N4 over homogeneous copper catalysts?
A: The heterogeneous Cu/g-C3N4 catalyst offers superior recyclability, maintaining significant catalytic efficiency over multiple cycles, and eliminates the need for complex heavy metal removal steps required with homogeneous systems.
Q: What is the substrate scope for this benzimidazole synthesis?
A: The method tolerates a wide range of substituents including alkyl, halogen, and trifluoromethyl groups on the aromatic ring, as well as various aliphatic and aromatic amines, allowing for diverse molecular design.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes cheap, commercially available starting materials and mild reaction conditions (70-90°C), making it highly amenable to scale-up from laboratory to industrial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development pipelines, and we are uniquely positioned to support your needs with our advanced manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can seamlessly transition your projects from early-stage discovery to full-scale commercial supply. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-trifluoromethyl benzimidazole meets the highest standards of quality and consistency required by the global pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss how this innovative catalytic technology can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the synthesis of complex fluorinated heterocycles.
