Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-Ones for Scalable Pharmaceutical Manufacturing
Introduction to High-Efficiency Triazole Synthesis
The 1,2,4-triazole-3-one scaffold represents a privileged structural motif in modern medicinal chemistry, extensively utilized in the development of potent bioactive agents ranging from antifungal and anti-inflammatory drugs to antitumor and anticonvulsant therapies. As illustrated in the diverse biological applications shown below, this heterocyclic core is integral to molecules acting as tyrosinase inhibitors, CB receptor modulators, and NK1 antagonists, underscoring its critical value in drug discovery pipelines. 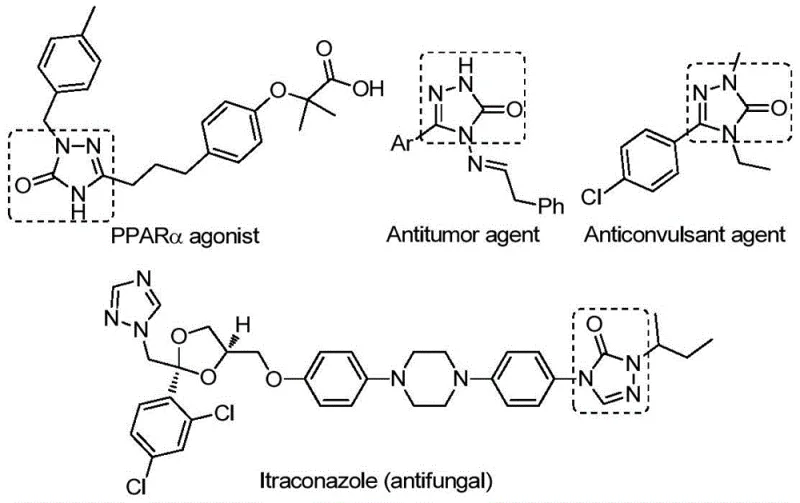 Recent advancements detailed in patent CN112538054B have introduced a transformative preparation method that addresses longstanding synthetic bottlenecks, utilizing a transition metal palladium-catalyzed carbonylation tandem cyclization strategy. This innovative approach leverages readily available chlorohydrazones and sodium azide as starting materials, operating under relatively mild thermal conditions to achieve high reaction efficiency and exceptional substrate compatibility. For research and development teams seeking reliable pharmaceutical intermediate suppliers, this methodology offers a robust pathway to access diverse derivatives that were previously difficult to synthesize, thereby accelerating the timeline from lead optimization to clinical candidate selection.
Recent advancements detailed in patent CN112538054B have introduced a transformative preparation method that addresses longstanding synthetic bottlenecks, utilizing a transition metal palladium-catalyzed carbonylation tandem cyclization strategy. This innovative approach leverages readily available chlorohydrazones and sodium azide as starting materials, operating under relatively mild thermal conditions to achieve high reaction efficiency and exceptional substrate compatibility. For research and development teams seeking reliable pharmaceutical intermediate suppliers, this methodology offers a robust pathway to access diverse derivatives that were previously difficult to synthesize, thereby accelerating the timeline from lead optimization to clinical candidate selection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazole-3-one ring system has been plagued by significant synthetic challenges that hinder efficient process development and scale-up. Traditional protocols often rely on the cyclization of benzoyl hydrazides with urea under strong basic conditions, or the tandem reaction of hydrazides with isocyanates, which frequently necessitates the use of hazardous reagents and stringent moisture-free environments. Other established routes involve the condensation of thioamides with hydrazines at elevated temperatures or the reaction of acyl isocyanates with monosubstituted hydrazines, all of which suffer from narrow substrate scopes and poor functional group tolerance. These conventional methods are typically characterized by complicated multi-step sequences, low overall yields, and the absolute requirement for pre-activation of reaction substrates, which drastically increases both the cost of goods and the environmental footprint of the manufacturing process. Furthermore, the harsh reaction conditions often lead to the formation of difficult-to-remove impurities, complicating downstream purification and jeopardizing the purity specifications required for active pharmaceutical ingredients.
The Novel Approach
In stark contrast to these legacy techniques, the novel palladium-catalyzed protocol described in the patent data introduces a streamlined, one-pot synthesis that fundamentally reshapes the economic and operational landscape of triazole production. By employing a sophisticated catalytic system comprising Pd2(dba)3 and Xantphos ligand alongside TFBen as a solid carbon monoxide source, this method enables the direct conversion of chlorohydrazones into the target heterocycles with remarkable efficiency. The reaction proceeds smoothly in common organic solvents like 1,4-dioxane at temperatures around 100°C, eliminating the need for high-pressure carbon monoxide gas cylinders and significantly enhancing laboratory safety.  This approach not only simplifies the operational workflow but also demonstrates excellent compatibility with a wide array of substituents, including alkyl, aryl, and halogen groups, allowing for the rapid generation of diverse chemical libraries. For procurement managers focused on cost reduction in API manufacturing, this shift towards milder conditions and cheaper starting materials translates directly into reduced energy consumption and lower raw material expenditures.
This approach not only simplifies the operational workflow but also demonstrates excellent compatibility with a wide array of substituents, including alkyl, aryl, and halogen groups, allowing for the rapid generation of diverse chemical libraries. For procurement managers focused on cost reduction in API manufacturing, this shift towards milder conditions and cheaper starting materials translates directly into reduced energy consumption and lower raw material expenditures.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, involving a intricate sequence of oxidative addition, migratory insertion, and rearrangement steps that ensure high fidelity in bond formation. The cycle initiates with the oxidative addition of the palladium(0) catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, generating a reactive divalent palladium intermediate that serves as the cornerstone for subsequent transformations. Simultaneously, the TFBen additive undergoes thermal decomposition to release carbon monoxide in situ, which then inserts into the carbon-palladium bond to form a crucial acyl-palladium species. This acyl intermediate subsequently reacts with sodium azide to generate an acyl azide compound, which spontaneously undergoes a Curtius rearrangement to yield a highly reactive isocyanate intermediate. The final ring closure is achieved through an intramolecular nucleophilic addition where the hydrazine nitrogen attacks the electrophilic carbon of the isocyanate, locking the structure into the stable 1,2,4-triazole-3-one framework.  Understanding this detailed mechanism is vital for R&D directors, as it highlights the precise control over impurity profiles; by avoiding high-energy radical pathways common in thermal cyclizations, this catalytic route minimizes side reactions and ensures a cleaner crude product profile that facilitates easier purification.
Understanding this detailed mechanism is vital for R&D directors, as it highlights the precise control over impurity profiles; by avoiding high-energy radical pathways common in thermal cyclizations, this catalytic route minimizes side reactions and ensures a cleaner crude product profile that facilitates easier purification.
Furthermore, the choice of ligands and solvents plays a pivotal role in stabilizing the catalytic species and promoting the turnover frequency of the cycle. The use of Xantphos, a bidentate phosphine ligand with a large bite angle, is instrumental in facilitating the reductive elimination step and preventing catalyst deactivation, while aprotic solvents like dioxane provide the optimal dielectric environment for the ionic intermediates involved in the azide substitution. The structural versatility of the starting chlorohydrazones, where R1 and R2 can be varied from simple alkyl chains to complex aromatic systems, allows for the modular synthesis of a vast array of derivatives without compromising the integrity of the catalytic cycle.  This mechanistic robustness ensures that even sterically hindered or electronically diverse substrates can be converted with high yields, providing supply chain heads with the confidence that the process can accommodate structural changes required during lead optimization without necessitating a complete process redevelopment.
This mechanistic robustness ensures that even sterically hindered or electronically diverse substrates can be converted with high yields, providing supply chain heads with the confidence that the process can accommodate structural changes required during lead optimization without necessitating a complete process redevelopment.
How to Synthesize 1,2,4-Triazole-3-One Efficiently
Implementing this synthesis in a practical setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and minimize waste. The standard protocol involves charging a reaction vessel with the palladium catalyst, ligand, TFBen, chlorohydrazone, and sodium azide in a molar ratio optimized for complete conversion, typically using a slight excess of the azide source to drive the equilibrium forward. The mixture is then heated to a controlled temperature range of 100 to 120 degrees Celsius and maintained under agitation for a period of 16 to 30 hours, ensuring sufficient time for the slow carbonylation and rearrangement steps to reach completion. Following the reaction, the workup procedure is notably straightforward, involving simple filtration to remove inorganic salts followed by silica gel treatment and column chromatography to isolate the pure product. This operational simplicity makes the method highly attractive for both laboratory-scale discovery and pilot-plant operations, reducing the technical barrier to entry for manufacturing this valuable class of compounds.
- Combine Pd2(dba)3 catalyst, Xantphos ligand, TFBen carbon monoxide source, chlorohydrazone substrate, and sodium azide in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 100-120°C and maintain stirring for 16 to 30 hours to allow the tandem cyclization and carbonylation to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity 1,2,4-triazole-3-one product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial strategic advantages that align perfectly with the goals of modern pharmaceutical supply chains seeking resilience and cost-efficiency. The reliance on chlorohydrazones and sodium azide as primary building blocks is particularly significant, as these commodities are widely available from global chemical markets at competitive price points, mitigating the risk of supply disruptions associated with exotic or proprietary reagents. Moreover, the elimination of high-pressure carbon monoxide gas removes a major safety hazard and regulatory burden, simplifying facility requirements and lowering insurance and compliance costs associated with handling toxic gases. The high atom economy and clean reaction profile further contribute to sustainability goals by reducing the volume of hazardous waste generated per kilogram of product, which is increasingly critical for meeting environmental regulations in chemical manufacturing.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of inexpensive, commodity-grade starting materials and the avoidance of complex multi-step sequences that characterize older synthetic routes. By consolidating the synthesis into a single catalytic pot reaction, manufacturers can significantly reduce labor hours, solvent consumption, and energy usage associated with intermediate isolations and purifications. The high yields reported across a broad substrate scope mean that less raw material is wasted on failed batches or low-conversion runs, leading to a more predictable and lower cost of goods sold. Additionally, the use of a solid CO surrogate like TFBen eliminates the need for specialized gas handling infrastructure, representing a capital expenditure saving for facilities upgrading their production capabilities.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the reaction conditions, which tolerate a wide variety of functional groups without requiring sensitive protection-deprotection strategies. This flexibility allows for the sourcing of diverse substituted chlorohydrazones from multiple vendors, preventing single-source dependency and enabling procurement teams to negotiate better terms. The scalability of the reaction, demonstrated effectively from milligram to gram scales in the patent data, suggests a smooth path to tonnage production without the nonlinear scale-up issues often encountered with exothermic or gas-dependent reactions. Consequently, partners can rely on consistent delivery schedules and stable quality attributes, which are essential for maintaining uninterrupted clinical trial supplies and commercial launches.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly superior to traditional methods, as it avoids the generation of sulfur-containing waste from thioamide routes or the use of corrosive bases required for urea condensations. The reaction operates in standard organic solvents that can be readily recovered and recycled, further minimizing the ecological footprint of the manufacturing process. The mild thermal conditions reduce the energy intensity of the operation, contributing to lower carbon emissions and aligning with corporate sustainability targets. For facilities operating under strict environmental permits, this cleaner technology offers a compliant pathway to expand production capacity without triggering additional regulatory hurdles or requiring expensive scrubber upgrades.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis, drawing directly from the experimental data and scope defined in the patent literature. These insights are intended to clarify the operational boundaries and potential applications of the technology for stakeholders evaluating its adoption. By understanding these nuances, technical teams can better assess the fit of this methodology within their existing process portfolios and identify opportunities for optimization.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis?
A: Unlike traditional methods requiring harsh conditions or pre-activated substrates, this protocol uses cheap chlorohydrazones and sodium azide under mild heating (100°C), offering broader substrate tolerance and higher efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability with simple operation, inexpensive reagents, and robust functional group compatibility, making it ideal for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the role of TFBen in this reaction mechanism?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a solid carbon monoxide substitute, releasing CO in situ under heating to facilitate the carbonylation step without requiring hazardous gas cylinders.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates that meet the rigorous demands of modern drug development, and our expertise in advanced catalytic methodologies positions us as an ideal partner for your project needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 1,2,4-triazole-3-one delivered meets your exacting standards for identity, potency, and impurity profiles. Our commitment to technical excellence means we can rapidly adapt this patented chemistry to produce custom derivatives tailored to your specific structure-activity relationship studies.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can optimize your supply chain and reduce your overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your target molecule and volume requirements. We encourage you to contact us today to obtain specific COA data for our standard catalog items or to initiate a dialogue regarding route feasibility assessments for your proprietary compounds, ensuring a secure and efficient supply of these vital pharmaceutical building blocks.
