Advanced Synthesis of N-N Axis Chiral Pyrrole Derivatives for Oncology Drug Development
Advanced Synthesis of N-N Axis Chiral Pyrrole Derivatives for Oncology Drug Development
The landscape of oncology drug discovery is constantly evolving, driven by the need for novel scaffolds that can overcome resistance mechanisms in tumor cells. A significant breakthrough in this domain is documented in Chinese Patent CN114524701B, which details a sophisticated method for synthesizing N-N axis chiral pyrrole derivatives. This technology represents a paradigm shift from traditional dynamic kinetic resolution methods, offering a direct, asymmetric construction of the N-N axial chirality through an in-situ ring formation strategy. For pharmaceutical developers, this patent provides access to a new class of heterocyclic compounds that exhibit potent cytotoxic activity against QGP-1 pancreatic tumor cells. As a leading entity in fine chemical manufacturing, we recognize the immense value of this methodology in expanding the chemical space available for medicinal chemistry campaigns, particularly for targets requiring high stereochemical purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral compounds has been fraught with challenges, often relying on resolution techniques that inherently limit the maximum theoretical yield to 50%. Conventional approaches to N-N axis chiral systems were extremely limited, mostly restricted to dynamic kinetic resolution or desymmetrization reactions which require complex substrate pre-functionalization. Furthermore, many existing protocols rely on transition metal catalysts that pose significant risks regarding residual metal impurities, a critical concern for API intermediates intended for human use. These traditional methods often suffer from narrow substrate scope, harsh reaction conditions, and poor atom economy, making them less attractive for cost-sensitive commercial manufacturing processes where efficiency and environmental compliance are paramount.
The Novel Approach
The methodology disclosed in CN114524701B introduces a transformative organocatalytic strategy that bypasses these historical bottlenecks. By utilizing chiral phosphoric acid catalysts, the process enables the direct coupling of indoleamines or pyrroleamines with 1,4-diketone derivatives to form the desired N-N axis chiral pyrrole structures in a single pot. This approach operates under remarkably mild conditions, typically at 25°C in carbon tetrachloride, eliminating the need for energy-intensive heating or cooling cycles. The use of 3A molecular sieves as an additive further drives the equilibrium towards product formation by sequestering water, ensuring high conversion rates. This novel route not only achieves excellent yields but also delivers exceptional enantioselectivity, with some examples reaching up to 96% ee, thereby providing a reliable source of high-purity chiral building blocks.
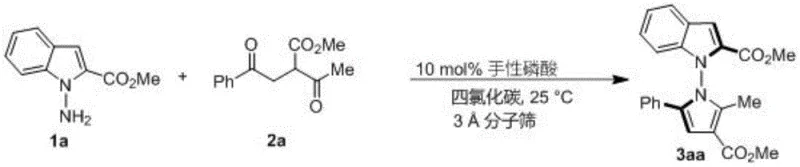
The versatility of this synthetic platform is exemplified by its ability to accommodate a wide range of substituents on both the amine and diketone components. As illustrated in the reaction scheme above, the condensation of indoleamine 1a with 1,4-diketone 2a proceeds smoothly to yield the target derivative 3aa with high fidelity. This flexibility allows medicinal chemists to rapidly generate diverse libraries of analogs for structure-activity relationship (SAR) studies without needing to redesign the core synthetic strategy for each new compound. The robustness of the reaction conditions ensures that sensitive functional groups remain intact, preserving the chemical integrity required for downstream biological testing.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
The success of this transformation hinges on the precise activation mode of the chiral phosphoric acid catalyst. These catalysts, often derived from BINOL or spiro cyclic skeletons, function as Brønsted acids that simultaneously activate the electrophilic carbonyl groups of the 1,4-diketone and organize the nucleophilic attack of the amine nitrogen. The chiral environment created by the bulky substituents on the phosphoric acid backbone (such as the 2,4,6-trimethylphenyl groups in catalyst 6j) imposes a rigid spatial arrangement on the transition state. This steric confinement effectively differentiates between the prochiral faces of the reacting species, guiding the formation of the N-N bond with a specific helical twist that results in the observed axial chirality. Understanding this mechanistic nuance is crucial for R&D directors aiming to optimize reaction parameters for specific substrate classes.
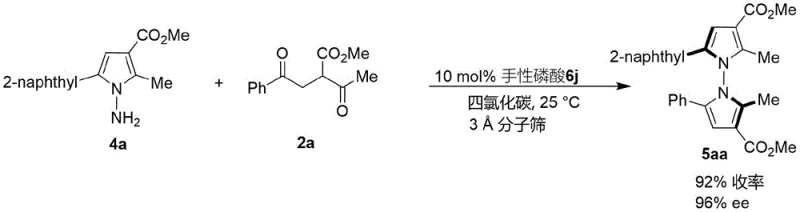
Furthermore, the mechanism inherently suppresses the formation of racemic byproducts and oligomeric impurities that often plague condensation reactions. The catalyst promotes a concerted cyclization pathway that minimizes the lifetime of reactive intermediates prone to non-selective side reactions. This high level of stereocontrol is evident in the synthesis of pyrrole-pyrrole derivatives as well, where the reaction between pyrroleamine 4a and diketone 2a yields product 5aa with 96% ee. Such precise impurity control is vital for reducing the burden on purification teams and ensuring that the final material meets the stringent specifications required for clinical grade intermediates. The ability to tune the catalyst structure allows for further optimization, making this a adaptable platform for various heterocyclic syntheses.
How to Synthesize N-N Axis Chiral Pyrrole Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational protocols to maximize yield and enantiomeric excess. The process is designed to be operationally simple, avoiding the need for inert atmosphere gloveboxes or specialized high-pressure reactors, which facilitates easier technology transfer. The standard procedure involves dissolving the amine and diketone substrates in carbon tetrachloride, adding activated 3A molecular sieves, and introducing the chiral phosphoric acid catalyst at a loading of 10 mol%. The reaction mixture is then stirred at ambient temperature (25°C) and monitored via thin-layer chromatography (TLC) until the starting materials are fully consumed. Following the reaction, the workup involves simple filtration to remove the molecular sieves and catalyst residues, followed by concentration and purification via silica gel column chromatography using a petroleum ether and ethyl acetate system.
- Prepare the reaction mixture by combining indoleamine or pyrroleamine substrates with 1,4-diketone derivatives in carbon tetrachloride solvent.
- Add 3A molecular sieves as an additive and introduce 10 mol% of a chiral phosphoric acid catalyst (preferably spiro-based derivative 6j).
- Stir the reaction at 25°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic route offers substantial strategic benefits beyond just chemical elegance. The elimination of expensive and toxic transition metals from the process flow drastically reduces the cost associated with metal scavenging and validation testing, which are mandatory steps in API manufacturing. Additionally, the use of readily available starting materials and a robust catalyst system enhances supply chain reliability, mitigating the risks associated with sourcing exotic reagents that may have long lead times or geopolitical supply constraints. The mild reaction conditions also translate to lower energy consumption and reduced safety hazards, contributing to a more sustainable and cost-effective manufacturing footprint that aligns with modern green chemistry principles.
- Cost Reduction in Manufacturing: The organocatalytic nature of this process removes the dependency on precious metal catalysts such as palladium or rhodium, which are subject to volatile market pricing and supply shortages. By replacing these with organic phosphoric acid derivatives, manufacturers can achieve significant cost savings on raw materials while simultaneously simplifying the purification workflow. The absence of heavy metals means that costly and time-consuming metal removal steps, such as treatment with specialized resins or repeated recrystallizations, can be minimized or eliminated entirely. This streamlining of the downstream processing directly lowers the cost of goods sold (COGS) and improves the overall economic viability of producing these complex chiral intermediates at scale.
- Enhanced Supply Chain Reliability: The substrates required for this synthesis, including various substituted indoleamines and 1,4-diketones, are commercially accessible or can be synthesized via established commodity chemical routes. This accessibility ensures a stable supply base, reducing the vulnerability of the production schedule to disruptions in the supply of niche reagents. Furthermore, the catalyst itself, being an organic molecule, does not face the same geopolitical mining and refining bottlenecks that affect transition metal supply chains. This stability allows for more accurate forecasting and inventory planning, ensuring that critical drug development timelines are met without unexpected delays caused by material shortages.
- Scalability and Environmental Compliance: The reaction operates at room temperature and atmospheric pressure, removing the need for complex engineering controls required for high-energy processes. This simplicity facilitates straightforward scale-up from gram-scale laboratory experiments to multi-kilogram or ton-scale commercial production without significant re-engineering of the reactor setup. Moreover, the process generates minimal hazardous waste compared to traditional methods, as it avoids the use of stoichiometric metal salts and harsh oxidants. This reduced environmental burden simplifies waste disposal compliance and lowers the associated costs, making the process more attractive for manufacturing in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
To assist our partners in evaluating this technology for their specific applications, we have compiled answers to common technical inquiries based on the patent data. These questions address key concerns regarding process robustness, catalyst performance, and the biological relevance of the resulting compounds. Understanding these details is essential for making informed decisions about integrating this synthetic route into your existing drug discovery pipelines or manufacturing operations.
Q: What are the advantages of using chiral phosphoric acid over transition metal catalysts for this synthesis?
A: Chiral phosphoric acid catalysts offer metal-free conditions which eliminate the risk of heavy metal contamination in pharmaceutical intermediates, significantly simplifying downstream purification and regulatory compliance.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the method utilizes mild reaction conditions (25°C), common solvents like carbon tetrachloride, and robust catalysts, making it highly suitable for commercial scale-up without requiring extreme pressure or temperature equipment.
Q: What is the biological significance of these N-N axis chiral pyrrole derivatives?
A: These derivatives have demonstrated strong cytotoxic activity against QGP-1 pancreatic tumor cells, indicating their high potential as lead compounds for developing new anticancer therapeutics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Pyrrole Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like this chiral pyrrole synthesis can be successfully translated into industrial reality. Our team of expert chemists is adept at optimizing reaction parameters to meet stringent purity specifications, utilizing our rigorous QC labs to guarantee that every batch of intermediate delivered meets the highest quality standards required by global regulatory bodies. We understand the critical nature of supply continuity in the pharmaceutical industry and are committed to providing a secure, scalable source of these high-value chiral building blocks.
We invite you to contact our technical procurement team to discuss how we can support your project needs with a Customized Cost-Saving Analysis tailored to your specific volume requirements. Whether you need specific COA data for a particular batch or detailed route feasibility assessments for scaling up your own synthesis, our experts are ready to provide the technical depth and commercial flexibility you need to accelerate your drug development programs efficiently.
