Advanced Manufacturing of Lenalidomide Intermediates via Optimized Catalytic Hydrogenation
Advanced Manufacturing of Lenalidomide Intermediates via Optimized Catalytic Hydrogenation
The pharmaceutical landscape for oncology treatments continues to evolve, with Lenalidomide standing as a cornerstone therapy for multiple myeloma and myelodysplastic syndromes. Recent advancements in process chemistry, specifically detailed in patent CN106957299B, have introduced a transformative approach to synthesizing this critical active pharmaceutical ingredient (API). This innovation addresses long-standing inefficiencies in the production of the key intermediate, 3-(4-nitro-1-oxoisoindolin-2-yl)piperidine-2,6-dione, and its subsequent conversion to Lenalidomide. By leveraging a streamlined two-step sequence involving nucleophilic substitution and catalytic hydrogenation, the technology offers a pathway to higher purity and reduced environmental impact. For global supply chain leaders, this represents a significant opportunity to secure a more reliable lenalidomide intermediate supplier capable of meeting stringent regulatory demands while optimizing cost structures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for Lenalidomide have been plagued by substantial operational complexities and economic inefficiencies that hinder scalable manufacturing. Early methodologies, such as those described by Fang Feng et al., relied on multi-step sequences involving N-(benzyloxycarbonyl)-L-glutamine, necessitating cumbersome esterification and debenzylation steps that drastically extended production timelines. Furthermore, alternative pathways disclosed in patents like CN103601717A utilized bromination strategies that frequently generated dibromo-impurities, complicating purification and lowering overall yields. Other established methods, such as those referenced in US Patent 5656325517, resulted in products with undesirable dark coloration and required prolonged reaction times in large volumes of solvent, creating significant waste disposal challenges. Additionally, processes utilizing iron powder reduction, while cost-effective in reagent terms, produced vast quantities of iron sludge that were difficult to filter and posed severe environmental contamination risks.
The Novel Approach
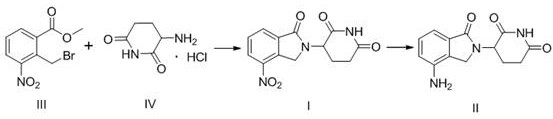
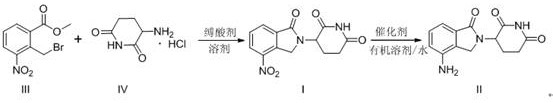
In stark contrast to these legacy methods, the novel process outlined in CN106957299B introduces a highly efficient strategy that bypasses unnecessary synthetic detours. This methodology initiates with the direct condensation of methyl 2-bromomethyl-3-nitrobenzoate and 3-amino-2,6-piperidinedione hydrochloride, utilizing inexpensive inorganic bases as acid scavengers. This single-step formation of the nitro-intermediate eliminates the need for protecting groups and reduces the total number of unit operations. The subsequent conversion to the final API is achieved through a clean catalytic hydrogenation using palladium on carbon in a mixed solvent system. This approach not only simplifies the workflow but also ensures the production of a white to off-white solid with superior purity profiles, effectively resolving the coloration and impurity issues endemic to previous techniques.
Mechanistic Insights into Pd/C Catalyzed Reduction and Cyclization
The core of this technological breakthrough lies in the precise control of reaction parameters during the cyclization and reduction phases. In the initial step, the nucleophilic attack of the amine nitrogen on the benzylic carbon of the bromo-ester is facilitated by the presence of inorganic bases such as sodium carbonate or potassium bicarbonate. These bases effectively neutralize the hydrochloride salt of the piperidine dione, generating the free amine in situ without promoting unwanted hydrolysis of the ester moiety. The choice of solvent, ranging from tetrahydrofuran to acetonitrile, is critical for maintaining the solubility of both reactants while stabilizing the transition state, ensuring high conversion rates to the nitro-intermediate (I) with minimal formation of regio-isomers or oligomeric byproducts.
Following the isolation of the intermediate, the reduction of the nitro group to the primary amine is executed using a heterogeneous palladium catalyst under controlled hydrogen pressure. The mechanistic advantage here is the use of a mixed solvent system comprising an organic component like DMF and water. This specific solvent ratio enhances the mass transfer of hydrogen gas to the catalyst surface while keeping the organic substrate in solution, preventing premature precipitation that could shield reactive sites. The mild reaction conditions, typically between 30-80°C and 0.3-0.8 MPa, prevent the over-reduction of the imide carbonyls, a common side reaction in less optimized systems. This selectivity is paramount for maintaining the structural integrity of the glutarimide ring, which is essential for the biological activity of the final drug substance.
How to Synthesize Lenalidomide Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and workup procedures to maximize yield and purity. The process begins with the reflux of the bromo-ester and amine salt in a suitable organic solvent with an inorganic base, followed by a straightforward aqueous quench and recrystallization to isolate the nitro-intermediate. Detailed standardized operating procedures for temperature control, catalyst loading, and filtration are essential for reproducibility. For a comprehensive breakdown of the specific reaction conditions, stoichiometric ratios, and purification protocols validated in the patent examples, please refer to the technical guide below.
- Condense methyl 2-bromomethyl-3-nitrobenzoate with 3-amino-2,6-piperidinedione hydrochloride using an inorganic base in an organic solvent.
- Isolate the nitro-intermediate through aqueous workup and recrystallization to ensure high purity.
- Perform catalytic hydrogenation using Pd/C in a mixed solvent system of DMF and water to reduce the nitro group to the amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond simple chemical efficiency. By eliminating the need for complex chiral starting materials like protected glutamine and avoiding the use of stoichiometric metal reductants like iron powder, the process fundamentally alters the cost structure of manufacturing. The reliance on commodity chemicals such as inorganic bases and standard heterogeneous catalysts reduces exposure to volatile raw material markets. Furthermore, the simplified downstream processing, characterized by easy filtration and crystallization, significantly lowers utility consumption and labor costs associated with extended purification cycles, offering a compelling value proposition for cost reduction in pharmaceutical intermediates manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive protecting group chemistry and the use of catalytic rather than stoichiometric reduction agents drive down the bill of materials. The process avoids the high costs associated with removing heavy metal residues from iron-based reductions, thereby reducing waste treatment expenses. Additionally, the higher purity of the crude intermediate minimizes the loss of material during recrystallization, leading to substantial cost savings through improved overall yield.
- Enhanced Supply Chain Reliability: The starting materials, specifically methyl 2-bromomethyl-3-nitrobenzoate and 3-amino-2,6-piperidinedione hydrochloride, are commercially available from multiple global vendors, mitigating the risk of single-source dependency. The robustness of the reaction conditions allows for flexible scheduling and reduces the likelihood of batch failures due to sensitive parameters. This stability ensures a consistent flow of high-purity lenalidomide intermediates, securing the supply chain against disruptions common with more fragile synthetic routes.
- Scalability and Environmental Compliance: The process is inherently green, replacing toxic iron sludge generation with a clean hydrogenation step that produces only water as a byproduct. The use of recyclable palladium catalysts and the potential for solvent recovery in the mixed DMF/water system align with strict environmental regulations. This environmental friendliness facilitates easier permitting for commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to expand capacity without incurring prohibitive environmental compliance costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into existing production facilities.
Q: How does this novel route improve upon traditional Lenalidomide synthesis methods?
A: Traditional methods often suffer from long synthetic routes, the use of expensive protecting groups like Cbz, or the generation of difficult-to-remove iron sludge. This novel approach utilizes a direct condensation of readily available starting materials followed by a clean catalytic hydrogenation, significantly shortening the process and eliminating heavy metal waste.
Q: What are the key advantages of the solvent system used in the hydrogenation step?
A: The use of a mixed solvent system comprising an organic solvent like DMF and water optimizes the solubility of the intermediate while facilitating the reaction kinetics. This balance allows for effective reduction at moderate temperatures and pressures, avoiding the need for excessive solvent volumes or hazardous reagents found in older protocols.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed with industrial scalability in mind. It employs robust inorganic bases and standard heterogeneous catalysts (Pd/C) that are easily filtered and recovered. The simplified workup procedures reduce energy consumption and waste generation, making it highly viable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lenalidomide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust supply chains for life-saving oncology medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Lenalidomide intermediate meets the highest international standards. Our commitment to quality assurance means that clients can rely on us for consistent product performance, essential for maintaining regulatory approval and patient safety.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply of this critical API intermediate remains uninterrupted and cost-effective.
