Advanced Rhodium-Catalyzed Synthesis of Polysubstituted Pyrroles for Commercial Scale Production
Advanced Rhodium-Catalyzed Synthesis of Polysubstituted Pyrroles for Commercial Scale Production
The chemical industry is constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly those with significant utility in pharmaceuticals and advanced materials. Patent CN112851674B introduces a groundbreaking methodology for the synthesis of polysubstituted pyrrole compounds, utilizing a transition metal rhodium-catalyzed double dehydrogenative coupling reaction. This innovative approach leverages internal enamines and internal alkynes as primary building blocks, facilitating a one-step construction of the target molecular architecture under relatively mild thermal conditions. The significance of this technology lies in its ability to bypass traditional multi-step sequences, thereby streamlining the production of high-value nitrogen heterocycles that are essential for modern drug discovery and functional material science. By employing a bimolecular vinylogous reaction strategy, this process overcomes previous limitations associated with unimolecular pathways, offering a robust platform for generating diverse chemical libraries.
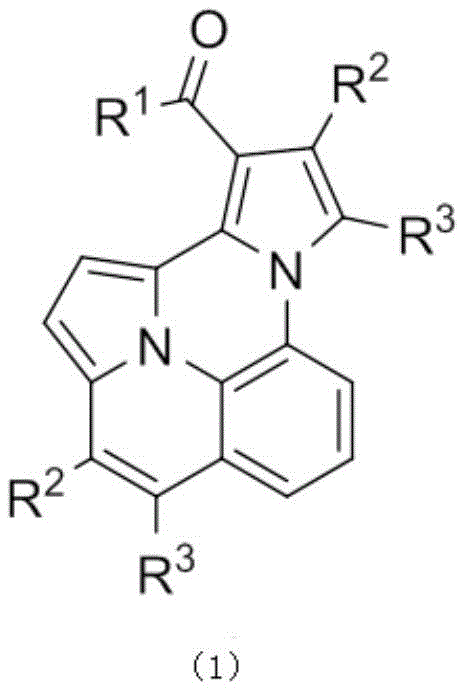
Furthermore, the structural versatility enabled by this patent allows for extensive functionalization, where substituents such as halogens, alkyl groups, and electron-donating or withdrawing moieties can be precisely incorporated into the pyrrole core. This level of control is paramount for R&D teams aiming to fine-tune the electronic and steric properties of final active ingredients or performance chemicals. The resulting compounds exhibit notable fluorescent characteristics, expanding their potential application spectrum beyond simple intermediates to include roles as fluorescent whitening agents, laser dyes, and photosensitizers. For supply chain managers, the implication is a versatile product line that can serve multiple market sectors ranging from agrochemicals to electronic materials, all derived from a single, scalable synthetic platform.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted pyrroles has been fraught with challenges related to regioselectivity, harsh reaction conditions, and limited substrate scope. Traditional methods often rely on pre-functionalized starting materials that require tedious preparation steps, leading to increased waste generation and higher overall production costs. Many conventional protocols necessitate the use of strong acids or bases, which can be incompatible with sensitive functional groups, thereby restricting the chemical diversity of the accessible library. Additionally, older catalytic systems frequently suffer from poor turnover numbers or require stoichiometric amounts of toxic heavy metals, creating significant environmental and disposal burdens for manufacturing facilities. The reliance on unimolecular cyclization strategies further limits the ability to introduce distinct substituents at specific positions on the pyrrole ring without complex protecting group manipulations.
The Novel Approach
In stark contrast, the methodology disclosed in CN112851674B utilizes a direct oxidative coupling strategy that merges an internal enamine with an internal alkyne in a single operational step. This bimolecular approach significantly simplifies the synthetic route by eliminating the need for pre-activated halides or organometallic reagents typically required in cross-coupling reactions. The use of a rhodium catalyst in conjunction with a copper oxidant enables a highly selective C-H activation process, ensuring that the new carbon-carbon bonds are formed precisely where needed without affecting other sensitive parts of the molecule. This not only enhances the atom economy of the process but also drastically reduces the number of purification steps required downstream. The ability to operate at temperatures around 110°C in common solvents like dichloroethane makes this process readily adaptable to existing reactor infrastructure without necessitating specialized high-pressure or cryogenic equipment.
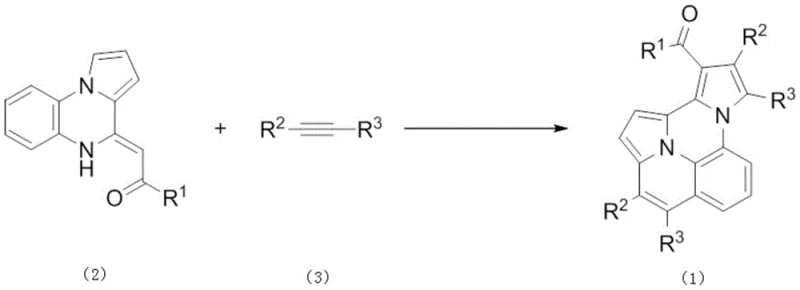
Mechanistic Insights into Rhodium-Catalyzed Double Dehydrogenative Coupling
The core of this technological advancement lies in the sophisticated catalytic cycle mediated by the rhodium species, likely involving a Rh(III)/Rh(I) redox manifold. The reaction initiates with the coordination of the internal enamine to the cationic rhodium center, followed by a directed C-H activation at the vinyl position to form a stable rhodacycle intermediate. This metallacycle then undergoes migratory insertion with the internal alkyne, effectively stitching the two carbon frameworks together. Subsequent beta-hydride elimination or oxidative dehydrogenation steps, facilitated by the copper acetate co-oxidant, restore the aromaticity of the pyrrole system and regenerate the active Rh(III) catalyst. Understanding this mechanism is crucial for process chemists, as it highlights the importance of maintaining anhydrous conditions and precise stoichiometry of the oxidant to prevent catalyst deactivation or side reactions such as homocoupling of the alkyne.
From an impurity control perspective, the high selectivity of the rhodium catalyst minimizes the formation of regioisomers, which are often difficult to separate in traditional pyrrole syntheses. The specific choice of ligands, such as the pentamethylcyclopentadienyl group in the dimeric catalyst precursor, provides a steric environment that favors the desired bimolecular insertion over competing pathways. Furthermore, the use of copper acetate as a terminal oxidant ensures that any reduced rhodium species are efficiently re-oxidized, maintaining a steady state concentration of the active catalyst throughout the prolonged reaction time of 24 hours. This mechanistic robustness translates directly to batch-to-batch consistency, a critical parameter for regulatory compliance in pharmaceutical manufacturing where impurity profiles must remain strictly defined and reproducible across large-scale production runs.
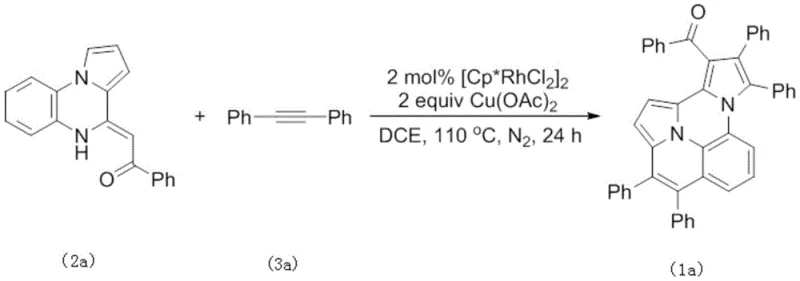
How to Synthesize Polysubstituted Pyrrole Efficiently
To implement this synthesis effectively, one must begin with the preparation of the key internal enamine precursor, which can be synthesized from readily available ketene dithioacetals and ortho-aminophenyl-pyrrole derivatives using Lewis acid catalysis. Once the starting materials are secured, the main coupling reaction is performed by combining the enamine and alkyne in a suitable solvent with the rhodium catalyst and copper oxidant under an inert atmosphere. The detailed standardized synthesis steps, including precise weighing, addition sequences, and workup procedures, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the internal enamine precursor by reacting appropriate ketene dithioacetals with ortho-aminophenyl-pyrrole derivatives under Lewis acid catalysis.
- Combine the internal enamine and internal alkyne in a solvent such as dichloroethane with a rhodium catalyst like [Cp*RhCl2]2 and copper acetate oxidant.
- Heat the reaction mixture to 110°C under nitrogen atmosphere for 24 hours, then purify the resulting polysubstituted pyrrole via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this rhodium-catalyzed protocol offers substantial strategic benefits regarding cost structure and logistical reliability. The primary advantage stems from the use of commodity chemicals as starting materials; internal alkynes and enamines are widely available from bulk chemical suppliers, reducing dependency on niche or custom-synthesized precursors that often carry long lead times and price volatility. By consolidating multiple synthetic transformations into a single pot, the process inherently reduces the consumption of solvents, energy, and labor hours associated with intermediate isolation and purification. This streamlined workflow not only lowers the direct cost of goods sold but also minimizes the physical footprint required for manufacturing, allowing for higher throughput within existing facility constraints.
- Cost Reduction in Manufacturing: The elimination of stoichiometric organometallic reagents and harsh activating agents significantly decreases the raw material expenditure per kilogram of product. Furthermore, the high efficiency of the catalytic system means that expensive rhodium metal is used in minute quantities and can potentially be recovered from the reaction residue, further enhancing the economic viability of the process. The reduction in waste generation also translates to lower disposal costs and reduced environmental compliance fees, contributing to a leaner and more sustainable cost model for large-scale production campaigns.
- Enhanced Supply Chain Reliability: Since the reaction tolerates a wide range of functional groups and substituents, manufacturers are not locked into a single specific supplier for highly specialized substrates. This flexibility allows procurement teams to source starting materials from multiple vendors, mitigating the risk of supply disruptions caused by geopolitical issues or production failures at a single site. The robustness of the reaction conditions, which do not require extreme pressures or temperatures, also ensures that the process can be reliably transferred between different manufacturing sites or contract organizations without significant re-validation efforts.
- Scalability and Environmental Compliance: The use of dichloroethane or dioxane as solvents, while requiring careful handling, is well-established in industrial settings with mature recovery and recycling protocols. The reaction generates minimal hazardous byproducts compared to traditional methods that might produce heavy metal sludge or acidic waste streams. This cleaner profile simplifies the permitting process for new production lines and aligns with increasingly stringent global environmental regulations, ensuring long-term operational continuity without the threat of regulatory shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to assist decision-makers in evaluating the feasibility of this technology for their specific applications.
Q: What are the optimal reaction conditions for this rhodium-catalyzed coupling?
A: The patent specifies that optimal yields (up to 89%) are achieved using 2 mol% [Cp*RhCl2]2 catalyst, 2 equivalents of Cu(OAc)2 oxidant, in dichloroethane (DCE) at 110°C for 24 hours under nitrogen.
Q: Can this method tolerate diverse functional groups on the substrates?
A: Yes, the method demonstrates wide substrate adaptability. The R groups can include various aryl substituents such as fluorine, chlorine, bromine, methyl, tert-butyl, methoxy, and cyano groups in ortho, meta, or para positions.
Q: What are the primary industrial applications of these pyrrole compounds?
A: These compounds serve as chromatographic analysis standards, fluorescent whitening agents, laser dyes, and valuable intermediates in the pharmaceutical industry due to their distinct fluorescent characteristics and structural versatility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this rhodium-catalyzed technology in accelerating the development of next-generation pharmaceuticals and functional materials. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory bench to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of polysubstituted pyrrole delivered meets the highest international standards for quality and consistency required by top-tier global enterprises.
We invite you to leverage our technical expertise to optimize your supply chain and reduce time-to-market for your critical projects. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals and drive value for your organization.
