Advanced Metal-Free Synthesis of Triazolopyrimidines for Commercial Scale-Up and API Production
Introduction to Novel Triazolopyrimidine Synthesis Technology
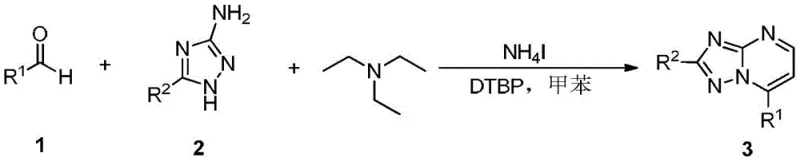
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for nitrogen-containing heterocycles, particularly [1,2,4]triazolo[1,5-a]pyrimidines, due to their profound biological activities ranging from PDE2a inhibition to antitumor and antiviral properties. A groundbreaking approach detailed in Chinese patent CN112778317B introduces a highly efficient, transition-metal-free methodology for constructing these privileged scaffolds. This innovation addresses critical bottlenecks in traditional organic synthesis by employing a one-pot tandem reaction strategy that utilizes readily available aldehydes and 3-amino-1,2,4-triazoles. By leveraging an ammonium iodide and di-tert-butyl peroxide (DTBP) catalytic system in toluene at 130°C, this method achieves exceptional regioselectivity and yield without the need for expensive palladium or copper catalysts. For R&D directors and procurement specialists, this represents a paradigm shift towards greener, more cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of [1,2,4]triazolo[1,5-a]pyrimidine cores has relied heavily on the condensation of 3-aminopyrazoles with β-dicarbonyl compounds or α,β-unsaturated carbonyl species. While these classical pathways are well-documented in academic literature, they suffer from significant practical drawbacks when applied to industrial settings. The primary challenge lies in the lack of regiocontrol, which frequently results in complex mixtures of structural isomers that are notoriously difficult to separate. This necessitates extensive chromatographic purification, drastically reducing the overall process efficiency and increasing the cost of goods sold (COGS). Furthermore, many traditional protocols require harsh reaction conditions or stoichiometric amounts of hazardous reagents, posing safety risks and environmental compliance issues that modern supply chains strive to eliminate. The presence of residual metal catalysts in the final product is another persistent concern, particularly for API applications where strict limits on heavy metals are enforced by regulatory bodies worldwide.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112778317B offers a streamlined solution that bypasses these historical inefficiencies through a direct oxidative cyclization mechanism. By utilizing a simple combination of an aldehyde, a 3-amino-1,2,4-triazole, and triethylamine in the presence of NH4I and DTBP, the reaction proceeds through a clean, one-pot sequence to deliver the target heterocycle in a single operational step. This approach not only eliminates the formation of regioisomers but also removes the dependency on transition metals, thereby simplifying the workup procedure to basic extraction and crystallization. The use of triethylamine serves a dual purpose as both a base and a reactant, significantly lowering raw material costs compared to specialized organometallic reagents. For procurement managers, this translates to a more resilient supply chain with reduced exposure to volatile metal prices and simplified logistics for hazardous waste disposal, ensuring a more sustainable and economically viable production model for complex heterocyclic intermediates.
Mechanistic Insights into Metal-Free Oxidative Cyclization
The success of this synthetic route hinges on a sophisticated radical-mediated mechanism initiated by the thermal decomposition of di-tert-butyl peroxide (DTBP). Upon heating to 130°C in toluene, DTBP generates tert-butoxy radicals which abstract hydrogen atoms from the triethylamine or interact with the ammonium iodide to generate reactive iodine species. These active intermediates facilitate the oxidative coupling between the aldehyde carbonyl carbon and the amino group of the triazole ring. The reaction likely proceeds through an imine intermediate followed by an intramolecular cyclization driven by the electrophilic activation of the triazole ring. The absence of transition metals suggests that the iodine species act as a redox mediator, shuttling electrons to promote the aromatization of the pyrimidine ring without becoming consumed in the process. This mechanistic elegance ensures high atom economy and minimizes the generation of inorganic salt byproducts, which is a critical factor for maintaining high purity standards in pharmaceutical manufacturing.
From an impurity control perspective, the specificity of this radical pathway is advantageous because it avoids the non-selective background reactions often seen with Lewis acid catalysts. The reaction conditions are tuned to favor the formation of the thermodynamically stable [1,2,4]triazolo[1,5-a]pyrimidine scaffold over potential kinetic byproducts. The use of toluene as a solvent provides an optimal boiling point that matches the activation energy required for the DTBP initiation while allowing for easy removal post-reaction. For quality assurance teams, this means the impurity profile is predictable and manageable, with the major impurities being unreacted starting materials that are easily removed during the aqueous workup described in the patent examples. This level of control is essential for scaling up the process to multi-kilogram batches while maintaining the stringent purity specifications required for clinical trial materials.
How to Synthesize [1,2,4]Triazolo[1,5-a]pyrimidines Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and addition order of reagents to maximize yield and safety. The patent specifies a molar ratio of 1:1:2:1:3 for the aldehyde, aminotriazole, triethylamine, ammonium iodide, and DTBP respectively, which has been optimized to drive the reaction to completion. The process begins by dissolving the solid reagents in toluene within a pressure-rated sealed tube or reactor, followed by the addition of the liquid components. Heating is maintained at 130°C for approximately 10 hours, after which the reaction is quenched with water and extracted with ethyl acetate. Detailed standard operating procedures regarding temperature ramping rates and quenching protocols are critical for ensuring reproducibility and safety during scale-up operations.
- Dissolve the aldehyde substrate, 3-amino-1,2,4-triazole derivative, and triethylamine in toluene solvent within a sealed reaction vessel.
- Add ammonium iodide and di-tert-butyl peroxide (DTBP) as the oxidant system to the reaction mixture under stirring.
- Heat the reaction mixture to 130°C for approximately 10 hours to facilitate the one-pot tandem cyclization and isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement officers, the adoption of this metal-free synthesis route offers substantial strategic advantages that extend beyond simple yield improvements. The elimination of precious metal catalysts such as palladium or rhodium removes a significant cost driver and supply risk, as the global market for these metals is often subject to geopolitical instability and price volatility. Furthermore, the reliance on commodity chemicals like triethylamine, ammonium iodide, and toluene ensures that raw material sourcing is straightforward and can be localized to multiple suppliers, enhancing supply chain resilience. The simplified purification process, which avoids complex column chromatography in favor of standard extraction and crystallization, reduces solvent consumption and processing time, leading to a smaller environmental footprint and lower utility costs per kilogram of product manufactured.
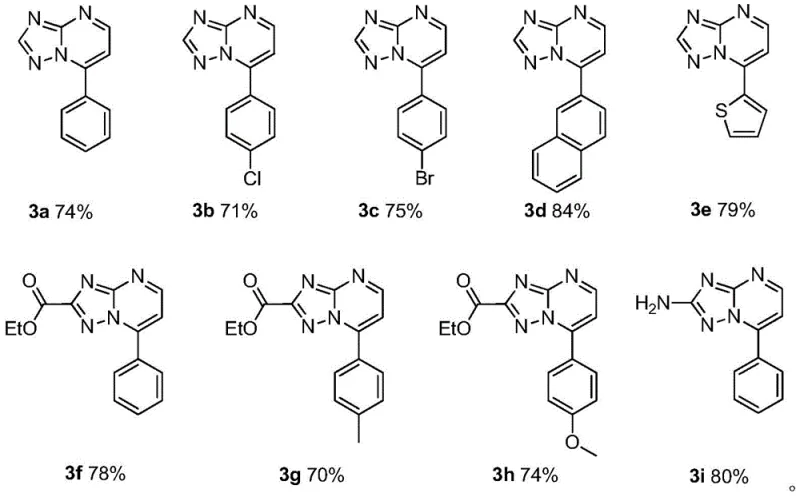
- Cost Reduction in Manufacturing: The economic impact of switching to this protocol is driven primarily by the removal of expensive catalytic systems and the reduction in downstream processing steps. Traditional methods often require scavengers to remove trace metals, adding both material costs and processing time; this new method inherently avoids that expense. Additionally, the high yields reported (ranging from 70% to 84%) mean less raw material is wasted, directly improving the mass balance and reducing the cost of goods. The use of triethylamine as a low-cost reagent further depresses the overall material bill, making the production of these high-value intermediates financially attractive even at large commercial scales.
- Enhanced Supply Chain Reliability: Sourcing reliability is significantly improved because the key reagents are bulk commodities rather than specialized fine chemicals with long lead times. Ammonium iodide and DTBP are widely available from multiple global chemical distributors, mitigating the risk of single-source dependency. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, allowing for greater flexibility in vendor selection. This stability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream API manufacturers who cannot afford interruptions in their supply of critical intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns well with green chemistry principles by avoiding toxic heavy metals and minimizing waste generation. The solvent system is standard and recyclable, and the absence of metal residues simplifies wastewater treatment protocols. Scaling this reaction from gram to tonnage levels is feasible using standard stainless steel reactors capable of handling mild pressures and temperatures, without the need for specialized lined vessels required for corrosive metal catalysts. This ease of scale-up accelerates the timeline from process development to commercial launch, allowing companies to respond faster to market demands for new therapeutic agents based on this scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is vital for making informed decisions about process adoption and technology transfer.
Q: What are the primary advantages of this metal-free synthesis over traditional methods?
A: Unlike conventional methods that often produce regioisomer mixtures requiring complex purification, this patent describes a highly selective one-pot reaction that avoids transition metal catalysts, thereby eliminating heavy metal residue concerns and simplifying downstream processing.
Q: Can this process be scaled for industrial API intermediate production?
A: Yes, the process utilizes common solvents like toluene and inexpensive reagents such as triethylamine and ammonium iodide, operating at moderate temperatures (130°C), which makes it highly suitable for commercial scale-up without specialized high-pressure equipment.
Q: What is the typical yield range for these triazolopyrimidine derivatives?
A: According to the experimental data in patent CN112778317B, the reaction consistently achieves high yields ranging from 70% to 84% across various substrates including substituted benzaldehydes and heterocyclic aldehydes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable [1,2,4]Triazolo[1,5-a]pyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthetic route for the production of high-purity pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of [1,2,4]triazolo[1,5-a]pyrimidine meets the exacting standards required for global regulatory submissions. We are committed to leveraging this innovative chemistry to deliver cost-effective solutions that accelerate your drug development timelines.
We invite you to engage with our technical procurement team to discuss how this technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a partnership built on transparency, technical excellence, and mutual growth in the competitive pharmaceutical landscape.
