Advanced Synthesis of Paxlovid Intermediate Compound I for Commercial Scale-up
Advanced Synthesis of Paxlovid Intermediate Compound I for Commercial Scale-up
The global pharmaceutical landscape has been profoundly impacted by the development of antiviral therapeutics, specifically regarding the synthesis of key intermediates for drugs like Paxlovid. Patent CN114133350B, published on May 23, 2023, discloses a groundbreaking preparation method for (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid methyl ester hydrochloride, widely known as Compound I (CAS: 565456-77-1). This compound serves as a critical chiral building block for PF07321332, the active protease inhibitor component of Paxlovid. The structural relationship between the final drug substance and this pivotal intermediate is illustrated below, highlighting the complexity of the bicyclic core that must be constructed with high stereochemical fidelity.
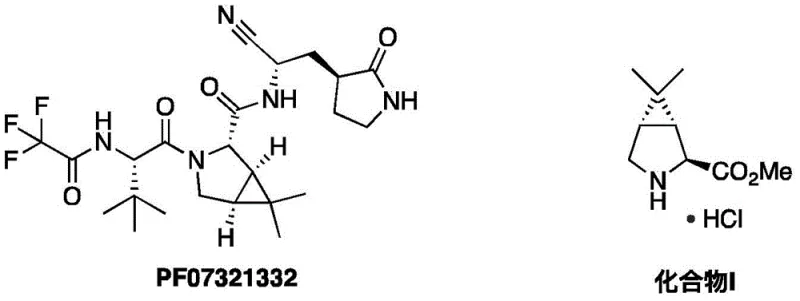
For R&D directors and process chemists, the significance of this patent lies in its departure from traditional, hazardous synthetic pathways. By leveraging a chiral pool starting material, N-Boc-trans-4-hydroxy-L-proline methyl ester, the inventors have devised a route that circumvents the need for late-stage resolution or dangerous cyanide additions. This innovation not only streamlines the manufacturing process but also aligns with modern green chemistry principles, offering a robust solution for reliable pharmaceutical intermediate suppliers aiming to secure the supply chain for next-generation antiviral medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of Compound I was plagued by significant operational hazards and inefficiencies, as documented in earlier patents such as WO2004113295A1 and WO2007075790A1. The legacy routes often commenced from less optimal starting materials like first ethyl chrysanthemate or caronic anhydride, necessitating long, multi-step sequences involving harsh oxidative cleavage with potassium permanganate and risky reductions using lithium aluminum hydride. Furthermore, these conventional methods frequently relied on the use of extremely toxic reagents such as potassium cyanide (KCN) and trimethylsilyl cyanide (TMSCN) to introduce the necessary nitrogen functionality or carbon framework. The reliance on heavy metal oxidants like silver nitrate and the requirement for chemical resolution steps further exacerbated the environmental burden, resulting in low total yields and prohibitive comprehensive production costs that hindered scalable manufacturing.
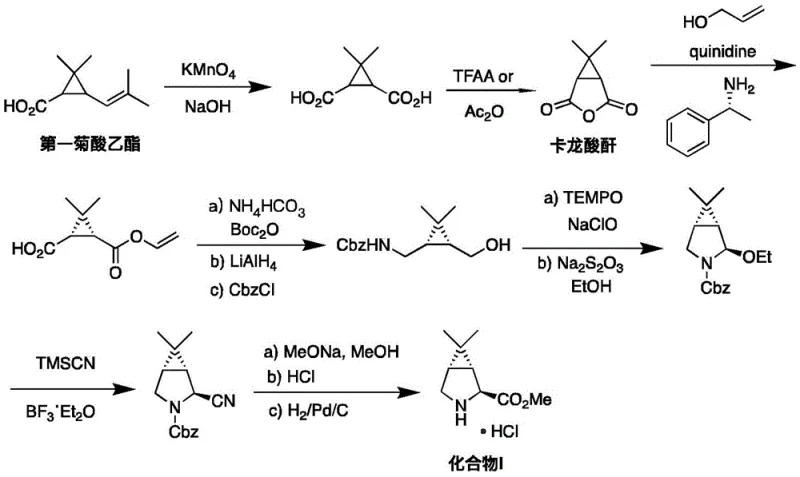
The Novel Approach
In stark contrast, the methodology described in CN114133350B introduces a streamlined, three-step strategy that fundamentally reshapes the production logic for cost reduction in API manufacturing. The new route initiates with the readily available and inexpensive N-Boc-trans-4-hydroxy-L-proline methyl ester (Compound II), effectively bypassing the need for complex ring constructions from scratch. The core innovation involves a sophisticated elimination reaction to generate a reactive alkene intermediate (Compound IV), followed by a highly stereoselective cyclopropanation using a phosphine ylide reagent. This approach completely eliminates the use of cyanide sources and strong reducing agents like LiAlH4, thereby drastically simplifying the safety protocols required for industrial operation. The final step involves a straightforward deprotection to yield the target hydrochloride salt, ensuring a process that is not only shorter but also significantly more amenable to commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Phosphine Ylide-Mediated Cyclopropanation
The heart of this synthetic breakthrough lies in the mechanistic elegance of the cyclopropanation step, which constructs the strained bicyclic [3.1.0] hexane system with precise stereocontrol. The process begins with the activation of the hydroxyl group in Compound II via sulfonylation using reagents such as methanesulfonyl chloride or p-toluenesulfonyl chloride. This creates a good leaving group, which is subsequently eliminated under basic conditions (using bases like potassium tert-butoxide or DBU) to form the unsaturated Compound IV. This alkene serves as the dipolarophile or acceptor in the subsequent cyclopropanation. The generation of the phosphine ylide reagent in situ, typically from isopropyl triphenylphosphine bromide or iodide and an organolithium base like n-butyllithium at temperatures ranging from -80°C to 50°C, allows for a controlled addition across the double bond. This reaction mechanism ensures the formation of the cis-fused cyclopropane ring system essential for the biological activity of the final drug, maintaining the chirality inherited from the proline starting material without erosion.
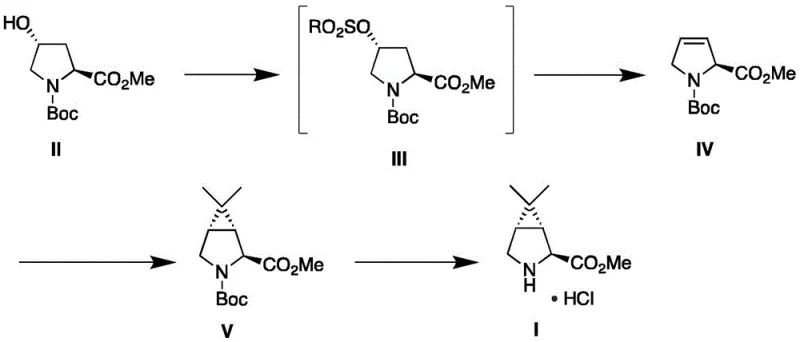
From an impurity control perspective, this mechanism offers distinct advantages over oxidative or cyanide-based routes. By avoiding radical oxidation steps or nucleophilic attacks by cyanide ions, the formation of regio-isomers and over-oxidized byproducts is minimized. The use of a chiral pool precursor means that the stereochemistry at the 2-position is locked early in the synthesis, and the cyclopropanation proceeds with high diastereoselectivity due to the steric influence of the existing chiral centers and the Boc protecting group. This inherent selectivity reduces the burden on downstream purification, allowing for the production of high-purity pharmaceutical intermediates with a cleaner impurity profile, which is a critical parameter for regulatory approval and patient safety.
How to Synthesize (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid methyl ester hydrochloride Efficiently
The execution of this synthesis requires careful attention to reaction conditions, particularly during the generation of the phosphine ylide and the subsequent cyclization. The patent details specific molar ratios and temperature controls to maximize yield and purity, emphasizing the importance of anhydrous conditions during the organolithium step. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for process implementation.
- React N-Boc-trans-4-hydroxy-L-proline methyl ester with sulfonyl chloride followed by base-mediated elimination to form the alkene intermediate.
- Perform cyclopropanation using a phosphine ylide reagent generated in situ from isopropyl triphenylphosphine halide and organolithium.
- Remove the Boc protecting group using a hydrogen chloride solution to yield the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates directly into tangible operational benefits and risk mitigation. The shift away from hazardous reagents and complex multi-step sequences addresses several critical pain points in the global supply of antiviral intermediates. By simplifying the chemical architecture of the process, manufacturers can achieve greater consistency in batch-to-batch quality while simultaneously reducing the logistical complexities associated with handling dangerous goods.
- Cost Reduction in Manufacturing: The economic impact of this route is driven primarily by the elimination of expensive and hazardous reagents. By removing the need for lithium aluminum hydride, silver nitrate, and cyanide salts, the process significantly lowers raw material costs and, more importantly, reduces the expenditure on specialized waste treatment and safety infrastructure. The use of cheap, commercially available starting materials like N-Boc-trans-4-hydroxy-L-proline methyl ester further drives down the cost of goods sold, making the final intermediate more competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the regulatory restrictions surrounding toxic chemicals. Since this new method avoids controlled substances like cyanides and highly reactive pyrophoric reagents where possible, it faces fewer regulatory hurdles and shipping restrictions. This ease of logistics ensures a more stable supply chain, reducing lead time for high-purity pharmaceutical intermediates and minimizing the risk of production stoppages due to raw material shortages or compliance issues.
- Scalability and Environmental Compliance: The reduction in reaction steps from lengthy sequences to a concise three-step process inherently improves scalability. Fewer unit operations mean less equipment occupancy time and lower energy consumption per kilogram of product. Furthermore, the drastic reduction in three-waste generation (wastewater, waste gas, and solid waste) aligns with increasingly stringent environmental regulations, facilitating easier permitting for commercial scale-up and enhancing the sustainability profile of the manufacturing site.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new preparation method is essential for stakeholders evaluating its potential for integration into their supply networks. The following questions address common inquiries regarding the safety, scalability, and chemical robustness of the process described in patent CN114133350B, providing clarity on its industrial viability.
Q: What are the safety advantages of this new synthesis route compared to prior art?
A: This novel route eliminates the use of highly toxic reagents such as potassium cyanide (KCN), trimethylsilyl cyanide (TMSCN), and lithium aluminum hydride (LiAlH4), significantly improving operational safety and reducing hazardous waste treatment costs.
Q: How does the chiral integrity of the molecule get maintained?
A: The process utilizes N-Boc-trans-4-hydroxy-L-proline methyl ester as a chiral pool starting material, ensuring high enantioselectivity throughout the elimination and cyclopropanation steps without the need for complex resolution processes.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method features short reaction steps, mild conditions, and avoids dangerous oxidation or reduction processes, making it highly scalable and cost-effective for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Compound I Supplier
As the demand for antiviral therapeutics continues to evolve, securing a stable source of high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep technical expertise to master complex synthetic challenges like the one presented in this patent. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global pharmaceutical partners. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Compound I meets the exacting standards required for API synthesis.
We invite you to collaborate with us to optimize your supply chain for Paxlovid intermediates. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value for your organization.
