Advanced Palladium-Catalyzed Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-One Production
Introduction to Next-Generation Pyrrol-2-One Synthesis
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a critical pharmacophore found in a diverse array of bioactive natural products and therapeutic agents. As illustrated in the provided structural analysis, this core skeleton is integral to molecules such as Althiomycin, known for its significant antibacterial properties, Glimepiride, a potent hypoglycemic agent used in diabetes management, and Isomalyngamide A, which exhibits promising anticancer activity. 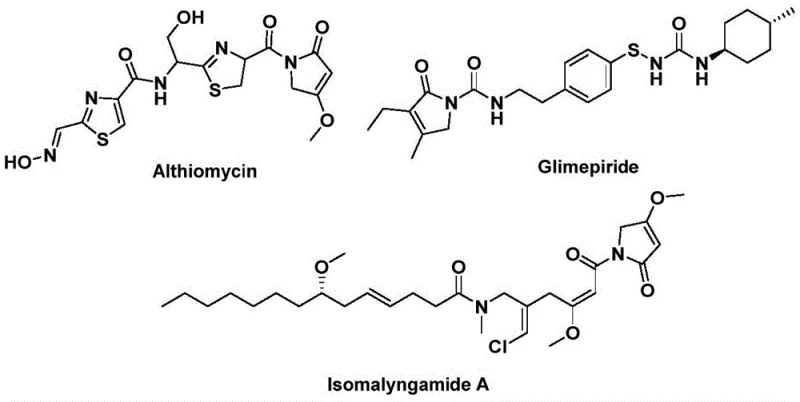 . Despite the high value of these compounds, traditional synthetic routes often suffer from complexity, low atom economy, and the requirement for hazardous reagents. Addressing these challenges, the groundbreaking methodology detailed in patent CN112694430B introduces a robust, palladium-catalyzed bis-carbonylation strategy. This innovation allows for the direct assembly of the pyrrol-2-one framework from simple, commercially abundant precursors, marking a significant leap forward in the efficient manufacturing of high-purity pharmaceutical intermediates.
. Despite the high value of these compounds, traditional synthetic routes often suffer from complexity, low atom economy, and the requirement for hazardous reagents. Addressing these challenges, the groundbreaking methodology detailed in patent CN112694430B introduces a robust, palladium-catalyzed bis-carbonylation strategy. This innovation allows for the direct assembly of the pyrrol-2-one framework from simple, commercially abundant precursors, marking a significant leap forward in the efficient manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-dihydro-2H-pyrrol-2-one ring system has relied on multi-step sequences that often involve harsh reaction conditions and expensive, specialized reagents. Conventional approaches may require the pre-functionalization of substrates, protection-deprotection strategies, or the use of toxic gaseous carbon monoxide under high pressure, which poses significant safety risks and engineering challenges in a production environment. Furthermore, many existing methods exhibit poor substrate tolerance, failing to accommodate diverse functional groups such as halogens or electron-withdrawing substituents without significant loss in yield. These limitations not only inflate the cost of goods sold (COGS) but also extend the lead time for process development, creating bottlenecks for reliable pharmaceutical intermediate suppliers aiming to bring new drugs to market rapidly.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a streamlined, one-pot tandem reaction that merges propargylamine derivatives with benzyl chloride equivalents. This method leverages a solid carbon monoxide substitute, specifically 1,3,5-tricarboxylic acid phenol ester (TFBen), which safely releases CO in situ, thereby eliminating the need for high-pressure gas equipment. The reaction demonstrates exceptional versatility, successfully accommodating a wide range of substituents on both the amine and the benzyl chloride components. As demonstrated by the successful synthesis of derivatives I-1 through I-5, the process tolerates methoxy, fluoro, chloro, trifluoromethyl, and methyl groups with impressive efficiency. 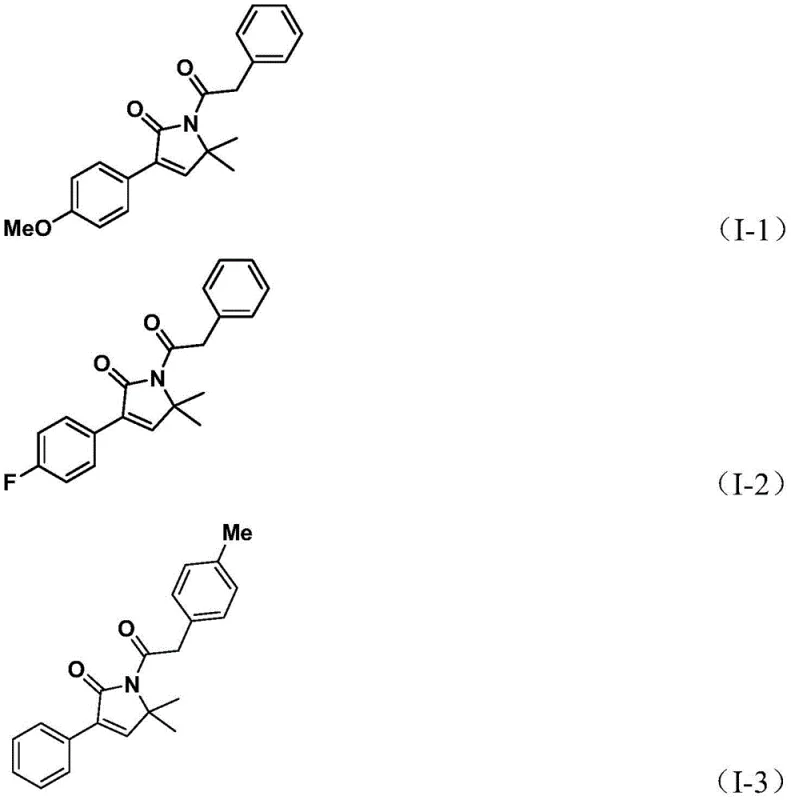 . This broad compatibility ensures that the method can be adapted for the cost reduction in API manufacturing across a vast chemical space, providing a flexible platform for generating diverse libraries of bioactive candidates.
. This broad compatibility ensures that the method can be adapted for the cost reduction in API manufacturing across a vast chemical space, providing a flexible platform for generating diverse libraries of bioactive candidates.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The efficacy of this transformation relies on a sophisticated catalytic cycle driven by palladium. The reaction initiates with the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzylpalladium intermediate. Subsequently, carbon monoxide, liberated from the phenol 1,3,5-tricarboxylate source, inserts into this organometallic species to form an acylpalladium complex. This acyl species then undergoes nucleophilic attack by the propargylamine, facilitating the formation of a five-membered ring palladium intermediate. The uniqueness of this mechanism lies in the second carbonylation event; another molecule of carbon monoxide inserts into the ring system to expand it transiently before reductive elimination occurs. 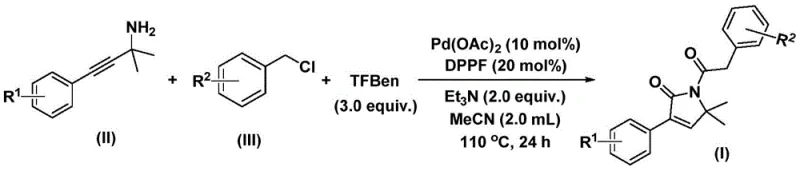 . This precise sequence of insertions and eliminations ensures the formation of the desired 1,5-dihydro-2H-pyrrol-2-one scaffold with high regioselectivity. Understanding this mechanism is crucial for R&D directors, as it highlights the importance of ligand selection (DPPF) and base strength (triethylamine) in stabilizing the intermediates and driving the reaction to completion.
. This precise sequence of insertions and eliminations ensures the formation of the desired 1,5-dihydro-2H-pyrrol-2-one scaffold with high regioselectivity. Understanding this mechanism is crucial for R&D directors, as it highlights the importance of ligand selection (DPPF) and base strength (triethylamine) in stabilizing the intermediates and driving the reaction to completion.
From an impurity control perspective, the use of a solid CO surrogate significantly reduces the risk of side reactions associated with uncontrolled CO pressure, such as over-carbonylation or polymerization. The mild reaction temperature of 110°C further minimizes thermal degradation of sensitive functional groups. The protocol specifies a molar ratio of catalyst to ligand to CO source that optimizes turnover frequency while keeping metal loading low (10 mol% Pd), which simplifies downstream purification. For procurement teams, this mechanistic clarity translates to predictable batch-to-batch consistency and reduced waste generation, aligning with modern green chemistry principles and stringent environmental compliance standards required in fine chemical production.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
The operational simplicity of this patented method makes it highly attractive for immediate adoption in pilot and production scales. The procedure involves charging a reactor with palladium acetate, the DPPF ligand, triethylamine, the CO source, and the respective substrates in acetonitrile. The mixture is then heated to 110°C for 24 hours, after which standard workup procedures involving filtration and silica gel treatment yield the crude product. Purification is achieved via standard column chromatography, a technique well-established in industrial settings. The detailed standardized synthesis steps, including specific molar ratios and troubleshooting tips for scaling, are outlined below to assist technical teams in replicating these high-yield results.
- Combine palladium catalyst, ligand, carbon monoxide substitute, base, propargylamine, and benzyl chloride in an organic solvent.
- Heat the reaction mixture to 100-120°C and maintain stirring for 24-48 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the shift towards this palladium-catalyzed methodology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the sourcing of raw materials; benzyl chlorides and propargylamines are commodity chemicals available from multiple global vendors, ensuring supply continuity and mitigating the risk of single-source dependency. Furthermore, the elimination of gaseous carbon monoxide cylinders removes a major logistical and safety hurdle, reducing the need for specialized storage infrastructure and hazardous material handling certifications. This simplification of the input supply chain directly contributes to enhanced supply chain reliability, allowing manufacturers to maintain consistent production schedules even during periods of raw material volatility.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the convergence of several factors. By utilizing a one-pot synthesis, the method eliminates the need for intermediate isolation and purification steps, which are typically the most labor-intensive and solvent-consuming parts of chemical manufacturing. The use of acetonitrile as a solvent, which is easily recoverable and recyclable, further drives down operational expenditures. Additionally, the high reaction efficiency reported in the patent, with yields reaching up to 92% for certain substrates, means that less raw material is wasted, maximizing the output per kilogram of input. This efficiency, combined with the avoidance of expensive, custom-synthesized precursors, results in substantial cost savings that can be passed down the value chain.
- Enhanced Supply Chain Reliability: In the context of global pharmaceutical supply chains, resilience is key. This synthesis route relies on robust, shelf-stable reagents that do not require cryogenic storage or inert atmosphere shipping, unlike many organometallic reagents used in alternative methods. The tolerance for various functional groups means that a single manufacturing line can be adapted to produce a wide portfolio of derivatives simply by swapping the starting benzyl chloride or amine. This flexibility allows suppliers to respond rapidly to changing market demands or custom orders from R&D partners, significantly reducing lead time for high-purity pharmaceutical intermediates and ensuring that critical drug development timelines are met without delay.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but the parameters defined in this patent are inherently scalable. The reaction temperature of 110°C is easily achievable in standard stainless steel reactors, and the use of a solid CO source avoids the mass transfer limitations associated with gas-liquid reactions in large vessels. From an environmental standpoint, the atom economy of the reaction is favorable, and the waste profile is cleaner compared to traditional cyclization methods that might generate stoichiometric amounts of heavy metal waste or toxic byproducts. This alignment with green chemistry metrics facilitates easier regulatory approval and supports the sustainability goals of modern chemical enterprises, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled answers to common inquiries regarding the process specifics and its applicability. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, ensuring that stakeholders have accurate information for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis?
A: This method utilizes readily available starting materials like benzyl chloride and propargylamine, operates under relatively mild conditions (110°C), and achieves high yields (up to 92%) in a single step, significantly simplifying the process compared to multi-step traditional routes.
Q: Is this process suitable for large-scale manufacturing of API intermediates?
A: Yes, the protocol uses commercially available catalysts and solvents (acetonitrile) and demonstrates excellent substrate compatibility with various functional groups (halogens, alkyl, alkoxy), making it highly adaptable for commercial scale-up.
Q: What is the role of 1,3,5-tricarboxylic acid phenol ester in the reaction?
A: It serves as a solid carbon monoxide substitute (CO source), releasing CO in situ to facilitate the bis-carbonylation process without the need for handling hazardous gaseous carbon monoxide directly.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN112694430B for accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop success to industrial reality is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay.
We invite pharmaceutical partners and contract research organizations to collaborate with us on leveraging this efficient synthesis route for their specific projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a secure and cost-effective supply of critical building blocks for your pipeline.
