Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-Up
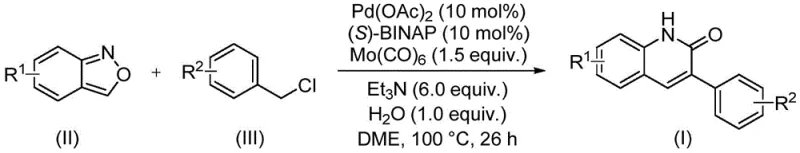
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN113045489B, which discloses a highly efficient preparation method for 3-arylquinolin-2(1H)-one derivatives. This technology leverages a palladium-catalyzed aminocarbonylation strategy that transforms simple benzisoxazoles and benzyl chlorides into complex quinolinone structures. For R&D directors and procurement managers alike, this innovation represents a paradigm shift away from traditional, hazardous carbonylation methods towards a safer, more atom-economical process. The ability to utilize benzisoxazole as a dual nitrogen and carbon source not only streamlines the synthetic pathway but also drastically reduces the logistical burden associated with handling gaseous carbon monoxide, thereby enhancing overall operational safety and cost-efficiency in API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)-one derivatives has relied heavily on classical methodologies such as the Vilsmeier-Haack, Knorr, and Friedlander reactions. While these methods are well-documented in academic literature, they often suffer from significant drawbacks when applied to modern industrial settings. Traditional routes frequently require harsh reaction conditions, including strong acids or bases, elevated temperatures, and prolonged reaction times that can degrade sensitive functional groups. Furthermore, many conventional approaches involve multi-step sequences with low overall yields, necessitating extensive purification protocols that generate substantial chemical waste. The reliance on external carbon monoxide sources for carbonylation reactions introduces severe safety hazards and requires specialized high-pressure equipment, which increases capital expenditure and complicates regulatory compliance. These limitations collectively hinder the rapid scale-up and cost-effective production of high-purity pharmaceutical intermediates required for next-generation therapeutics.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach described in the patent utilizes a sophisticated palladium-catalyzed system that operates under remarkably mild and controlled conditions. By employing benzisoxazole as an internal source of both nitrogen and the formyl group, the reaction eliminates the need for external CO gas, fundamentally changing the safety profile of the synthesis. The process integrates molybdenum hexacarbonyl as a solid CO surrogate, which releases carbon monoxide in situ under thermal conditions, ensuring a steady and safe supply of the carbonyl species directly within the reaction mixture. This methodology allows for the direct coupling of readily available benzyl chlorides with benzisoxazoles in a single pot, significantly reducing the number of unit operations. The result is a streamlined workflow that offers superior functional group tolerance, enabling the synthesis of diverse derivatives with electron-donating or electron-withdrawing substituents without compromising yield or purity.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The core of this transformative synthesis lies in its intricate catalytic cycle, which orchestrates the activation of inert bonds and the precise assembly of the quinolinone core. The reaction initiates with the oxidative addition of the benzyl chloride substrate to the active palladium(0) species, generated in situ from palladium acetate and the chiral ligand (S)-BINAP. This step forms a benzyl-palladium(II) complex, which is subsequently intercepted by carbon monoxide released from the thermal decomposition of molybdenum hexacarbonyl. The resulting acyl-palladium intermediate is then poised for nucleophilic attack. Crucially, the benzisoxazole ring undergoes a reductive cleavage or activation facilitated by the metal center, providing the necessary nitrogen nucleophile and the carbon framework for ring closure. This tandem sequence of carbonylation and cyclization occurs seamlessly within the coordination sphere of the palladium catalyst, driven by the thermodynamic stability of the final aromatic quinolinone system.
From an impurity control perspective, this mechanism offers distinct advantages over random condensation reactions. The specificity of the palladium catalyst ensures that the carbonylation occurs selectively at the benzylic position, minimizing side reactions such as homocoupling of the benzyl chloride or polymerization of the intermediates. The use of triethylamine as a base serves to neutralize the hydrochloric acid byproduct generated during the oxidative addition, preventing acid-catalyzed degradation of the sensitive benzisoxazole ring or the final product. Additionally, the inclusion of a stoichiometric amount of water appears to play a critical role in facilitating the hydrolysis or proton transfer steps necessary for the final aromatization and catalyst turnover. This precise control over the reaction environment results in a clean crude profile, simplifying downstream purification and ensuring that the final API intermediates meet stringent quality specifications regarding residual metals and organic impurities.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction parameters to maximize yield and reproducibility. The protocol dictates a specific molar ratio of catalyst to substrate, typically utilizing 10 mol% of palladium acetate and 10 mol% of the (S)-BINAP ligand to ensure sufficient catalytic activity throughout the 26-hour reaction period. The choice of solvent, ethylene glycol dimethyl ether (DME), is critical for solubilizing both the organic substrates and the inorganic additives while maintaining thermal stability at the operating temperature of 100°C. Operators must ensure that the reaction vessel is properly sealed to prevent solvent loss and maintain the integrity of the in situ generated carbon monoxide atmosphere. Following the reaction, the workup procedure involves a straightforward filtration to remove insoluble metal residues and salts, followed by adsorption onto silica gel for final purification via column chromatography, yielding the target compound in high purity suitable for further biological evaluation or drug development.
- Charge a reactor with palladium acetate, (S)-BINAP ligand, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and the specific benzyl chloride substrate in DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers compelling economic and logistical benefits that directly impact the bottom line. The primary driver of cost reduction is the substitution of hazardous and expensive gaseous carbon monoxide with solid molybdenum hexacarbonyl and inexpensive benzisoxazole feedstocks. This shift not only lowers the raw material costs but also eliminates the need for specialized high-pressure reactors and the associated safety infrastructure, leading to substantial capital expenditure savings. Furthermore, the high functional group tolerance of the reaction means that a single standardized protocol can be used to manufacture a wide library of derivatives, reducing the need for custom process development for each new analog. This versatility allows for greater agility in responding to market demands and accelerates the timeline from discovery to commercial supply, providing a significant competitive advantage in the fast-paced pharmaceutical sector.
- Cost Reduction in Manufacturing: The elimination of external carbon monoxide cylinders and high-pressure equipment drastically reduces operational overhead and safety compliance costs. By utilizing cheap and widely available starting materials like benzyl chlorides and benzisoxazoles, the overall cost of goods sold (COGS) is significantly optimized. The high yields reported across various substrates minimize waste generation and maximize the throughput of valuable intermediates, ensuring that every kilogram of raw material contributes effectively to the final product output without the need for costly recycling loops or complex waste treatment procedures.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as benzyl chlorides and palladium acetate ensures a stable and resilient supply chain, as these materials are sourced from multiple global suppliers with established logistics networks. Unlike specialized reagents that may face shortages or long lead times, the precursors for this synthesis are produced at scale for various industrial applications, guaranteeing consistent availability. This reliability mitigates the risk of production delays due to raw material scarcity, allowing manufacturers to maintain continuous production schedules and meet tight delivery deadlines for their downstream pharmaceutical clients without interruption.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, transitioning smoothly from gram-scale laboratory experiments to multi-kilogram pilot runs and eventually to ton-scale commercial production. The use of a closed system with solid CO surrogates minimizes volatile organic compound (VOC) emissions and eliminates the risk of toxic gas leaks, aligning perfectly with increasingly stringent environmental regulations. The simplified workup procedure reduces solvent consumption and waste generation, supporting green chemistry initiatives and lowering the environmental footprint of the manufacturing process, which is a key metric for modern sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on the practical aspects of adopting this method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the robustness of the chemistry and its alignment with industry standards for quality and safety.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl (C1) source, eliminating the need for separate toxic carbon monoxide gas cylinders and simplifying the reaction setup significantly.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process utilizes commercially available starting materials like benzyl chlorides and operates under standard heating conditions (100°C), making it highly scalable for industrial production without requiring specialized high-pressure equipment.
Q: What is the typical yield range for these derivatives?
A: According to the patent data, the reaction demonstrates high efficiency with isolated yields ranging from 68% to 97% across various substrates, indicating robust functional group tolerance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the palladium-catalyzed aminocarbonylation described in CN113045489B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle sensitive organometallic reactions with the highest safety standards, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 3-arylquinolin-2(1H)-one meets the exacting requirements of global regulatory bodies. We are committed to delivering high-quality pharmaceutical intermediates that empower your drug discovery and development programs.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us be your partner in turning complex chemical challenges into commercial successes.
