Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Derivatives for Pharmaceutical Applications
Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Derivatives for Pharmaceutical Applications
The landscape of heterocyclic chemistry is continually evolving, driven by the demand for more efficient and environmentally benign synthetic routes for bioactive scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN113307790B, which discloses a robust preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. These nitrogen-containing heterocycles are pivotal structural motifs found in numerous drug intermediates and functional material molecules, including ligands for organic light-emitting diodes (OLEDs). The patent introduces a transformative oxidative cyclization strategy that bypasses the limitations of conventional multi-step syntheses. By leveraging a metal-free catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), this methodology offers a streamlined pathway to access complex triazole architectures. For R&D directors and procurement specialists alike, this innovation represents a critical opportunity to enhance the purity and cost-efficiency of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been fraught with inefficiencies that hinder large-scale commercial application. Traditional protocols typically rely on quinoline-2-carboxylic acid as the primary starting material, necessitating a cumbersome five-step reaction sequence to achieve the final target molecule. This linear approach not only consumes significant time and resources but also suffers from abysmal overall yields, often reported as low as 17%. Furthermore, these legacy methods frequently demand severe reaction conditions, including stringent temperature controls and the use of hazardous reagents, which escalate safety risks and waste disposal costs. The reliance on such inefficient pathways creates a bottleneck in the supply chain for reliable pharmaceutical intermediate suppliers, driving up the cost of goods sold (COGS) and limiting the availability of high-purity materials needed for downstream drug development.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113307790B utilizes readily available and inexpensive starting materials, specifically 2-methylquinoline and trifluoroacetohydrazide derivatives, to construct the triazole core in a single pot. This novel approach employs an oxidative cyclization mechanism promoted by a TBAI/TBHP system, effectively converting the methyl group of the quinoline into the necessary aldehyde intermediate in situ, which then condenses and cyclizes. As illustrated in the general reaction scheme below, this process eliminates the need for pre-functionalized carboxylic acids and avoids the use of toxic heavy metal catalysts entirely.
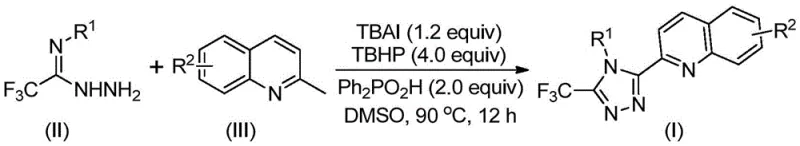
The operational simplicity is further enhanced by the fact that the reaction proceeds efficiently in common organic solvents like DMSO at moderate temperatures of 80-100°C. This shift from a five-step, low-yield process to a direct, high-yielding oxidative cyclization marks a substantial advancement in cost reduction in API manufacturing, providing a scalable solution for producing diverse triazole libraries.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction is believed to proceed through a radical-mediated pathway initiated by the interaction between tetrabutylammonium iodide and tert-butyl peroxide. Initially, the oxidant facilitates the conversion of the 2-methylquinoline substrate into a 2-quinolinecarbaldehyde intermediate. This aldehyde then undergoes a condensation reaction with the trifluoroacetohydrazide to form a dehydrated hydrazone intermediate. Subsequent oxidative iodination and intramolecular electrophilic substitution trigger the cyclization event, followed by aromatization to yield the final 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole. The presence of diphenylphosphoric acid acts as a crucial additive, likely stabilizing intermediates or facilitating proton transfer steps, thereby enhancing the overall reaction efficiency and yield.
From an impurity control perspective, the metal-free nature of this catalytic system is a distinct advantage. Traditional transition-metal catalyzed couplings often leave behind trace metal residues that are difficult to remove and can be toxic in pharmaceutical applications. By utilizing an organocatalytic iodine/peroxide system, the impurity profile is significantly simplified, primarily consisting of organic byproducts that are easier to separate via standard chromatographic techniques. This inherent cleanliness reduces the burden on downstream purification processes, ensuring that the final high-purity OLED material or pharmaceutical intermediate meets stringent regulatory specifications without the need for expensive metal scavenging resins.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
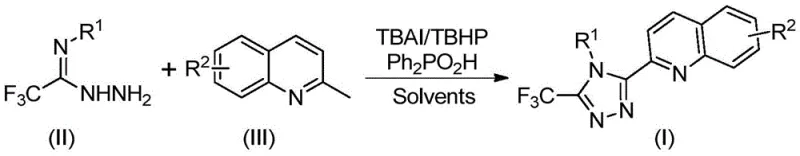
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield. The patent provides extensive experimental data across fifteen examples, demonstrating the robustness of the method across various electronic and steric environments. The standard protocol involves mixing the hydrazide, 2-methylquinoline, TBAI, TBHP, and diphenylphosphoric acid in DMSO, followed by heating. The versatility of the method allows for the introduction of diverse substituents (R1 and R2), including halogens, alkyl groups, and electron-withdrawing nitro groups, without compromising the reaction outcome. Below is the specific reaction setup used in the exemplary embodiments.
- Combine tetrabutylammonium iodide, tert-butyl peroxide aqueous solution, diphenylphosphoric acid, trifluoroacetohydrazide derivative, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for 8 to 14 hours to ensure complete conversion via oxidative cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product using column chromatography to isolate the high-purity triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of rare earth elements. Moreover, the use of commodity chemicals like 2-methylquinoline and tert-butyl peroxide ensures a stable and continuous supply of raw materials, reducing the lead time for high-purity pharmaceutical intermediates. The operational flexibility of the process, which tolerates ambient moisture and oxygen, lowers the barrier for entry for contract manufacturing organizations (CMOs) that may lack specialized inert atmosphere equipment.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by the drastic simplification of the synthetic route. By collapsing a five-step sequence into a single oxidative cyclization step, manufacturers can significantly reduce labor costs, solvent consumption, and energy usage associated with multiple isolation and purification stages. The avoidance of expensive heavy metal catalysts further contributes to substantial cost savings, as there is no need for costly metal removal technologies or validation testing for residual metals. Additionally, the high conversion rates observed in the patent examples minimize raw material waste, optimizing the atom economy of the process and lowering the overall cost of goods.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, bulk commodity feedstocks. Unlike specialized precursors that may have limited suppliers or long lead times, 2-methylquinoline and the requisite hydrazides are produced on a large industrial scale globally. This abundance ensures that production schedules are less likely to be disrupted by raw material shortages. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in environmental controls, leading to more consistent batch-to-batch quality and reliable delivery timelines for downstream customers.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this metal-free protocol aligns well with green chemistry principles. The absence of toxic heavy metals simplifies waste stream management and reduces the environmental footprint of the manufacturing process, facilitating easier regulatory compliance in jurisdictions with strict discharge limits. The reaction has been demonstrated to work effectively on gram scales with high yields, indicating strong potential for commercial scale-up of complex pharmaceutical intermediates. The straightforward workup procedure, involving filtration and column chromatography, is easily adaptable to larger reactor volumes, supporting the transition from pilot plant to multi-ton annual production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this synthesis route.
Q: What are the key advantages of this metal-free synthesis method over traditional routes?
A: Unlike traditional methods requiring quinoline-2-carboxylic acid and five reaction steps with only 17% yield, this novel approach utilizes cheap 2-methylquinoline and achieves yields up to 97% in a single pot without toxic heavy metal catalysts.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, a major operational advantage of this patented method is that it does not require anhydrous or oxygen-free conditions, significantly simplifying the equipment requirements and reducing operational costs for large-scale production.
Q: What is the substrate scope for the R1 and R2 groups in this triazole synthesis?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents such as methyl, methoxy, halogens (Cl, Br), nitro, and trifluoromethyl groups on both the aryl hydrazide and the quinoline ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting cutting-edge synthetic methodologies to drive innovation in the pharmaceutical and fine chemical sectors. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and assay. By integrating the efficient, metal-free oxidative cyclization technology described in CN113307790B into our portfolio, we can offer our partners a superior supply solution that balances performance with sustainability.
We invite global partners to collaborate with us to leverage this advanced manufacturing capability for their next-generation drug candidates or functional materials. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your project's bottom line while ensuring a secure and high-quality supply chain.
