Advanced Synthesis of N-N Axis Chiral Pyrrole Derivatives for Pharmaceutical Applications
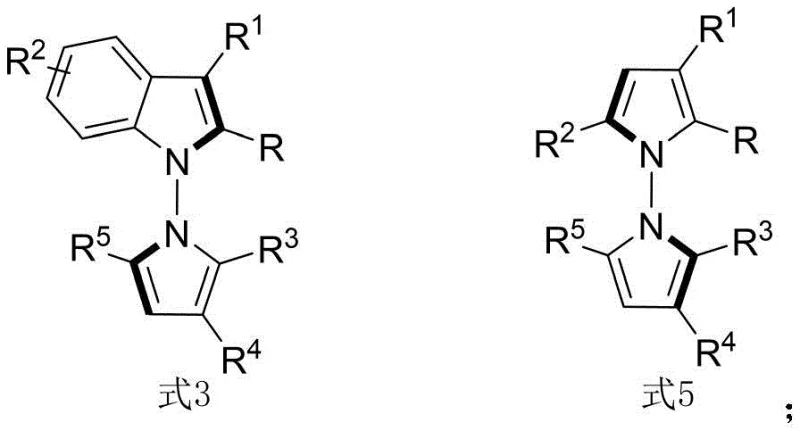
The pharmaceutical and fine chemical industries are constantly seeking novel scaffolds that offer unique biological profiles, and the recent disclosure in patent CN114524701B represents a significant breakthrough in this domain. This patent introduces a sophisticated methodology for the synthesis of N-N axis chiral pyrrole derivatives, a class of compounds that has historically been difficult to access with high stereochemical purity. The core innovation lies in the utilization of a chiral phosphoric acid catalyst to facilitate an in-situ ring formation strategy, effectively bridging the gap between simple amine precursors and complex, biologically active heterocycles. Unlike previous approaches that were restricted to narrow substrate scopes, this technology enables the construction of both N-N axis chiral indole-pyrrole and pyrrole-pyrrole derivatives with exceptional enantioselectivity. Furthermore, the biological evaluation data provided within the patent highlights the potent cytotoxic activity of these derivatives against QGP-1 tumor cells, positioning them as valuable candidates for oncology research and drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral compounds containing nitrogen-nitrogen bonds has been plagued by significant synthetic challenges that hindered their widespread application in medicinal chemistry. Conventional strategies primarily relied on dynamic kinetic resolution or desymmetrization reactions, which inherently limit the structural diversity of the accessible products. These traditional methods often require harsh reaction conditions, expensive transition metal catalysts, or complex multi-step sequences that result in poor atom economy and low overall yields. Moreover, the inability to effectively control the stereochemistry at the N-N axis frequently leads to racemic mixtures, necessitating difficult and costly separation processes that are impractical for large-scale manufacturing. The scarcity of efficient methods to construct these specific N-N chiral motifs has created a bottleneck in the development of new therapeutic agents that rely on this unique spatial arrangement for their biological efficacy.
The Novel Approach
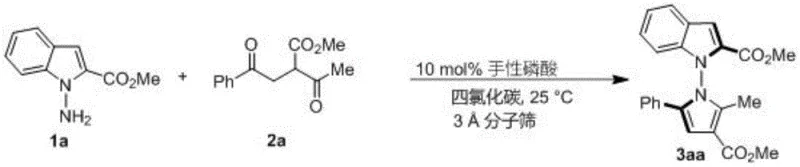
The methodology described in CN114524701B offers a transformative solution by employing a direct condensation and cyclization strategy driven by organocatalysis. By utilizing 1,4-diketone derivatives and indoleamines or pyrrole amines as readily available starting materials, this process bypasses the need for pre-functionalized chiral pools. The reaction proceeds smoothly in carbon tetrachloride at room temperature, facilitated by a chiral phosphoric acid catalyst and molecular sieves, which act as a water scavenger to drive the equilibrium forward. This approach not only simplifies the operational complexity but also dramatically improves the stereoselectivity, achieving enantiomeric excess (ee) values as high as 96% in optimized examples. The versatility of this method is further demonstrated by its tolerance to a wide range of substituents on both the amine and diketone components, allowing for the rapid generation of diverse libraries of chiral pyrroles for structure-activity relationship studies.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
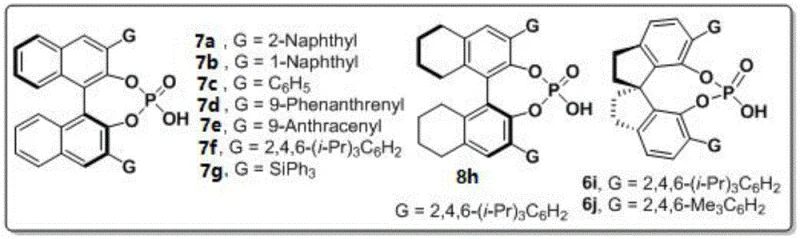
The success of this synthesis hinges on the precise activation mode of the chiral phosphoric acid catalyst, which serves as a bifunctional organocatalyst to orchestrate the stereochemical outcome of the reaction. The catalyst, particularly the spiro-binaphthyl derived phosphoric acid designated as compound 6j in the patent, likely operates through a dual hydrogen-bonding mechanism. It simultaneously activates the carbonyl groups of the 1,4-diketone electrophile and the amino group of the nucleophile, organizing them within a well-defined chiral pocket. This rigid chiral environment ensures that the cyclization occurs with high facial selectivity, effectively transferring the chirality from the catalyst backbone to the newly formed N-N axis of the product. The use of 3A molecular sieves is critical in this mechanism, as the removal of water generated during the condensation prevents the hydrolysis of the intermediate imines and shifts the reaction equilibrium towards the desired pyrrole product, thereby maximizing yield and preventing side reactions.
From an impurity control perspective, the mild nature of this organocatalytic process offers distinct advantages over metal-catalyzed alternatives. The absence of heavy metals eliminates the risk of metal contamination in the final API intermediate, a critical quality attribute for pharmaceutical manufacturing. Furthermore, the high regioselectivity observed in the cyclization minimizes the formation of structural isomers, simplifying the downstream purification process. The patent data indicates that varying the steric bulk of the substituents on the phosphoric acid catalyst (such as the 2,4,6-trimethylphenyl groups in catalyst 6j) fine-tunes the steric hindrance around the active site, which is essential for suppressing non-selective background reactions. This level of control ensures that the final product meets stringent purity specifications required for clinical applications, reducing the burden on quality control laboratories.
How to Synthesize N-N Axis Chiral Pyrrole Derivatives Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to specific protocol parameters regarding reagent stoichiometry and reaction monitoring. The process is designed to be robust, utilizing standard glassware and ambient temperature conditions, which lowers the barrier to entry for adoption. The key to success lies in the precise preparation of the reaction mixture, ensuring that the molecular sieves are activated and the solvent is anhydrous to maintain catalyst efficiency. While the general procedure is straightforward, attention to detail during the workup and purification stages is essential to preserve the optical purity of the sensitive chiral axis. For a comprehensive understanding of the standardized operating procedures, please refer to the detailed synthesis guide below.
- Prepare the reaction mixture by combining indoleamine or pyrrole amine with a 1,4-diketone derivative in carbon tetrachloride solvent.
- Add 3A molecular sieves as an additive and introduce 10 mol% of the optimal chiral phosphoric acid catalyst (such as compound 6j).
- Stir the reaction at 25 °C until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis technology presents a compelling value proposition centered on cost efficiency and supply reliability. The shift from complex multi-step syntheses or resolution processes to a direct, one-pot catalytic reaction fundamentally alters the cost structure of producing these high-value intermediates. By eliminating the need for expensive transition metals and reducing the number of unit operations, manufacturers can achieve substantial cost savings in raw material consumption and waste disposal. The use of commodity chemicals like carbon tetrachloride and commercially available amines further stabilizes the supply chain against volatility in specialty reagent markets. Additionally, the mild reaction conditions reduce energy consumption associated with heating or cooling, contributing to a lower carbon footprint and aligning with modern sustainability goals in chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive metal scavenging steps and rigorous testing for residual metals, which are significant cost drivers in pharmaceutical production. The high atom economy of the condensation reaction ensures that a larger proportion of the starting mass is converted into the final product, minimizing waste generation and lowering the cost per kilogram of the active ingredient. Furthermore, the ability to achieve high yields and enantioselectivity in a single step reduces the need for recrystallization or chiral chromatography, streamlining the production workflow and significantly lowering operational expenditures.
- Enhanced Supply Chain Reliability: The reliance on broadly available starting materials such as substituted indoles, pyrroles, and 1,4-diketones mitigates the risk of supply disruptions often associated with exotic or proprietary reagents. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process does not require specialized high-pressure reactors or cryogenic cooling systems that might create bottlenecks in a multipurpose facility. This flexibility ensures consistent delivery timelines for downstream customers, enhancing the overall reliability of the supply chain for critical oncology intermediates.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving basic filtration and concentration followed by standard column chromatography, facilitates easy scale-up from gram to ton quantities without significant process re-engineering. The use of molecular sieves as a benign additive avoids the generation of hazardous aqueous waste streams typical of acid-base workups, simplifying environmental compliance and wastewater treatment. This green chemistry profile not only reduces regulatory hurdles but also positions the manufacturer as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this method. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing portfolios and for procurement teams assessing the long-term viability of the supply source.
Q: What is the primary advantage of this new synthesis method over traditional dynamic kinetic resolution?
A: Unlike traditional methods limited to dynamic kinetic resolution and desymmetrization, this patent utilizes an in-situ ring formation strategy that significantly expands the structural diversity of N-N axis chiral pyrrole derivatives while achieving extremely high enantioselectivity.
Q: What biological activity do these N-N axis chiral pyrrole derivatives exhibit?
A: Biological activity tests indicate that these derivatives possess high sensitivity and strong cytotoxic activity specifically against QGP-1 pancreatic tumor cells, suggesting significant potential for oncology drug development.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates under mild and conventional conditions (room temperature, simple solvents) with high yields and stereoselectivity, making it highly suitable for cost-effective commercial scale-up without requiring extreme pressure or temperature equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Pyrrole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the N-N axis chiral pyrrole scaffold in next-generation drug discovery, particularly for oncology applications targeting pancreatic cancer. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications and high enantiomeric excess, guaranteeing that every batch meets the exacting standards required for pharmaceutical intermediates. We are committed to leveraging advanced organocatalytic technologies like the one described in CN114524701B to deliver high-quality solutions that accelerate your development timelines.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific project requirements. By engaging with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic benefits of switching to this catalytic method for your specific target molecules. We encourage you to contact our technical procurement team today to request specific COA data for our reference standards and to discuss route feasibility assessments tailored to your unique chemical challenges, ensuring a partnership built on scientific excellence and commercial reliability.
