Scalable Synthesis of 2-Trifluoromethyl Quinazolinones via Iron Catalysis for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. This technology addresses critical bottlenecks in the synthesis of these privileged scaffolds, which are ubiquitous in drug molecules exhibiting anti-cancer, anticonvulsant, and anti-inflammatory activities. By leveraging a cost-effective iron-catalyzed system, this invention offers a transformative approach for the commercial scale-up of complex pharmaceutical intermediates, providing a reliable pathway for manufacturers aiming to optimize their supply chains for high-value API precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones functionalized with trifluoromethyl groups has relied heavily on cyclization reactions involving synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with anthranilamides or isatoic anhydrides. While chemically feasible, these traditional routes suffer from severe limitations that hinder industrial adoption. The reaction conditions are often excessively harsh, requiring strong acids or bases and elevated temperatures that can degrade sensitive functional groups. Furthermore, the starting materials, particularly specialized trifluoromethyl synthons, are frequently expensive and difficult to source in bulk quantities, leading to inflated production costs. Perhaps most critically for process chemists, these legacy methods often exhibit narrow substrate scope and inconsistent yields, making them unreliable for the cost reduction in pharmaceutical intermediate manufacturing that modern generic and innovator drug companies demand.
The Novel Approach
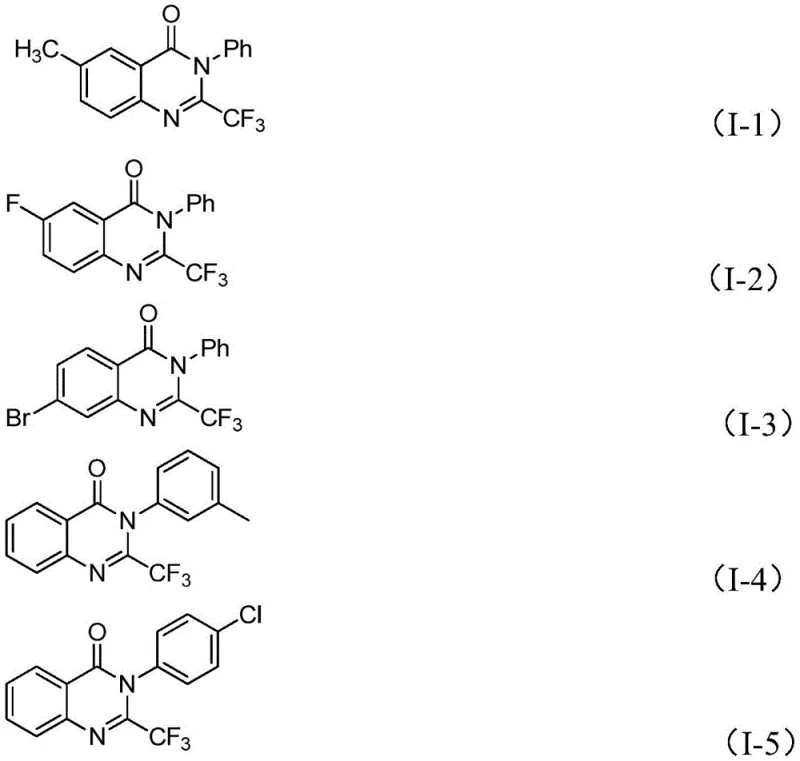
In stark contrast, the methodology described in the patent utilizes readily available trifluoroethylimidoyl chloride and isatin derivatives as starting materials, catalyzed by inexpensive ferric chloride. This new route fundamentally shifts the economic and operational landscape of quinazolinone production. The reaction proceeds through a tandem sequence involving base-promoted carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization. This strategy not only utilizes cheaper raw materials but also demonstrates exceptional functional group tolerance, accommodating a wide array of substituents including halogens, alkyls, and nitro groups. The ability to synthesize diverse analogs like those shown below ensures that R&D teams can rapidly access varied chemical space for SAR studies without being constrained by synthetic feasibility.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the efficient utilization of iron catalysis to drive the cyclization process. The reaction mechanism initiates with the interaction between the trifluoroethylimidoyl chloride and the isatin substrate in the presence of sodium hydride. This base-promoted step facilitates the formation of a key trifluoroacetamidine intermediate. Subsequently, the ferric chloride catalyst plays a pivotal role in mediating the decarbonylation and subsequent intramolecular cyclization. This iron-mediated pathway is distinct from traditional transition metal catalysis as it avoids the use of toxic and costly precious metals, aligning with green chemistry principles while maintaining high reaction efficiency. The catalytic cycle is robust enough to proceed even under air atmosphere, simplifying the operational requirements for large-scale reactors.
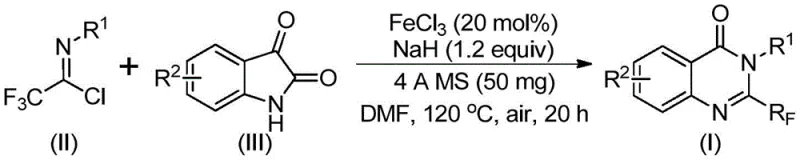
From an impurity control perspective, this mechanism offers significant advantages. The specificity of the iron-catalyzed decarbonylation minimizes the formation of side products often associated with non-selective thermal cyclizations. The use of 4A molecular sieves in the reaction mixture further enhances purity by scavenging moisture, which could otherwise hydrolyze the sensitive imidoyl chloride starting material. This results in a cleaner crude reaction profile, reducing the burden on downstream purification processes such as column chromatography or recrystallization. For a reliable pharmaceutical intermediate supplier, this translates to higher overall throughput and consistent quality, ensuring that the final high-purity pharmaceutical intermediates meet the rigorous specifications required for GMP manufacturing.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The experimental protocol outlined in the patent provides a clear roadmap for executing this synthesis with high reproducibility. The process involves a two-stage heating profile, starting at a moderate temperature to establish the intermediate, followed by a higher temperature phase to drive the cyclization to completion. This controlled thermal progression is key to maximizing yield while minimizing decomposition. Detailed standardized operating procedures for this synthesis are essential for technology transfer and scale-up activities.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in anhydrous DMF solvent.
- Stir the reaction mixture at 40°C for 8-10 hours to facilitate initial bond formation, then increase temperature to 120°C.
- Maintain reaction at 120°C for 18-20 hours under air atmosphere, followed by filtration, silica gel mixing, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical curiosity. The shift from precious metal catalysts to abundant iron salts represents a direct intervention in the cost structure of the molecule. By eliminating the dependency on volatile commodity markets for palladium or rhodium, manufacturers can stabilize their cost of goods sold (COGS) and protect margins against fluctuating metal prices. Furthermore, the removal of heavy metal catalysts simplifies the purification train, potentially reducing the number of processing steps and the consumption of solvents and adsorbents required for metal scavenging.
- Cost Reduction in Manufacturing: The utilization of ferric chloride as a catalyst provides a substantial economic advantage due to its negligible cost compared to traditional noble metal catalysts. Additionally, the starting materials, specifically isatin derivatives and trifluoroethylimidoyl chlorides, are commodity chemicals available from multiple global suppliers, fostering a competitive sourcing environment that drives down input costs. The high atom economy of the cyclization step further contributes to waste reduction, lowering disposal costs and enhancing the overall sustainability profile of the manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of this reaction conditions, specifically its tolerance to air and moisture (with molecular sieves), reduces the risk of batch failures due to minor environmental excursions. This reliability is crucial for maintaining continuous supply lines. Moreover, the broad substrate scope means that a single manufacturing platform can be adapted to produce a library of related analogs, allowing supply chain planners to respond agilely to changing demand for different API variants without needing to validate entirely new processes for each derivative.
- Scalability and Environmental Compliance: The protocol has been demonstrated to work effectively on gram scales with potential for seamless expansion to multi-kilogram and ton-level production. The absence of toxic heavy metals simplifies regulatory compliance regarding residual metal limits in the final drug substance. This ease of compliance accelerates the regulatory filing timeline and reduces the analytical burden on quality control laboratories, facilitating faster time-to-market for new drug candidates utilizing this scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed synthesis. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: What are the advantages of using FeCl3 over precious metal catalysts for quinazolinone synthesis?
A: Using ferric chloride eliminates the need for expensive palladium or rhodium catalysts, significantly reducing raw material costs and simplifying the removal of heavy metal residues to meet stringent pharmaceutical purity standards.
Q: What is the substrate scope for this trifluoromethylation reaction?
A: The method demonstrates excellent functional group tolerance, successfully accommodating substrates with electron-donating groups (methyl, methoxy) and electron-withdrawing groups (halogens like F, Cl, Br, and nitro groups) at various positions on the aromatic rings.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the protocol utilizes cheap, commercially available starting materials like isatin and operates under relatively mild conditions without requiring inert gas protection (air atmosphere), making it highly amenable to kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has thoroughly analyzed the potential of the FeCl3-catalyzed cyclization method described in CN111675662B. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest industry standards.
We invite you to collaborate with us to leverage this advanced technology for your specific pipeline needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for both cost and continuity in the competitive pharmaceutical landscape.
