Revolutionizing Biphenyl Fused Ring Synthesis for Advanced OLED Material Manufacturing
Revolutionizing Biphenyl Fused Ring Synthesis for Advanced OLED Material Manufacturing
The landscape of organic synthesis for high-performance electronic materials is undergoing a significant transformation, driven by the urgent need for cost-effective and scalable manufacturing processes. A pivotal advancement in this domain is detailed in patent CN109678648B, which discloses a novel method for synthesizing fused ring compounds containing a biphenyl structure. This technology addresses the longstanding inefficiencies associated with traditional cyclization reactions, particularly the excessive consumption of precious metal catalysts. By leveraging a sophisticated non-aqueous diazotization protocol coupled with a low-temperature palladium-catalyzed coupling strategy, this invention achieves a dramatic reduction in catalyst loading while simultaneously enhancing reaction yields. For industry leaders seeking a reliable fused ring compound supplier, understanding the mechanistic nuances of this patent is essential, as it represents a paradigm shift from brute-force catalysis to precision chemical engineering.
The core innovation lies in the stabilization of the reactive diazo intermediate within a low-freezing-point solvent system. Unlike conventional aqueous methods that suffer from thermal instability and rapid catalyst deactivation, this approach maintains the integrity of the divalent palladium species throughout the reaction cycle. This not only ensures a more consistent product quality but also simplifies the downstream purification processes, which is a critical factor for manufacturers of high-purity OLED materials. The ability to produce complex biphenyl scaffolds with minimal metal contamination directly translates to improved device performance and longevity in final electronic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fused ring compounds containing biphenyl structures has been plagued by significant economic and technical hurdles. Traditional methodologies, such as the third method described in the background of the patent, rely heavily on the direct coupling of diazo groups with adjacent benzene rings under relatively harsh conditions. As illustrated in the reaction scheme below, these conventional routes typically necessitate reaction temperatures exceeding 60°C and require substantial quantities of divalent palladium catalysts, often ranging from 1% to 5% of the substrate mass.
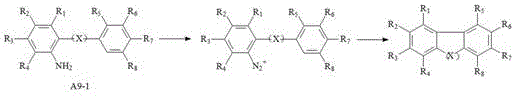
The reliance on such high catalyst loadings is primarily due to the rapid reduction of active divalent palladium to inactive zero-valent palladium colloids under acidic and high-temperature conditions. This deactivation phenomenon forces manufacturers to compensate with excess catalyst, driving up raw material costs significantly. Furthermore, the use of aqueous acidic media for diazotization introduces stability issues, leading to frequent side reactions and lower overall yields, often struggling to exceed 50% when non-palladium catalysts are attempted. These inefficiencies create a bottleneck for the commercial scale-up of complex polymer additives and electronic intermediates, where purity and cost are paramount.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN109678648B introduces a refined two-step process that elegantly circumvents these limitations. The novel approach begins with the preparation of a diazo compound in a non-aqueous phase using reagents such as tert-butyl nitrite or isoamyl nitrite. This initial step is conducted in a low-freezing-point solvent, which serves as the foundation for the subsequent low-temperature coupling reaction. By shifting the reaction environment away from aqueous acidity, the process inherently stabilizes the diazo functionality, preventing premature decomposition.
The true breakthrough occurs in the second step, where the coupling reaction is executed at cryogenic temperatures ranging from -20°C to -50°C. This低温 (low temperature) regime effectively suppresses the reduction of the palladium catalyst, allowing it to remain in its active divalent state for extended periods. Consequently, the required catalyst dosage plummets to a mere 0.05-0.2% of the substrate weight, representing a magnitude of improvement over prior art. This strategic manipulation of reaction thermodynamics not only slashes catalyst costs but also enhances the selectivity of the cyclization, resulting in superior yields and simplified waste management protocols.
Mechanistic Insights into Low-Temperature Palladium-Catalyzed Cyclization
To fully appreciate the technical depth of this invention, one must examine the specific mechanistic pathways enabled by the non-aqueous diazotization step. The process initiates with the conversion of an ortho-amino polyphenyl precursor into a diazonium species without the presence of water. As depicted in the reaction scheme below, the use of alkyl nitrites in solvents like methyl cyclopentyl ether or acetonitrile ensures that the resulting diazo compound remains soluble and stable, avoiding the precipitation issues common with diazonium fluoroborate salts.

Following the formation of the diazo intermediate, the reaction mixture is cooled to the critical range of -20°C to -50°C before the introduction of the palladium catalyst and an acid-binding agent. The inclusion of a base, such as triethylamine or potassium acetate, is crucial; it neutralizes any acidic byproducts generated during the coupling, maintaining a slightly alkaline environment that further protects the divalent palladium from reduction. This precise control over the reaction milieu allows the intramolecular coupling to proceed smoothly, forming the new carbon-carbon bond that closes the fused ring system. The result is a highly efficient transformation that maximizes atom economy while minimizing the formation of metallic impurities.
Furthermore, the impurity profile of the resulting product is significantly cleaner compared to traditional methods. In conventional high-temperature processes, the degradation of the catalyst often leads to the formation of palladium black and various organometallic byproducts that are difficult to remove. By maintaining the catalyst in a homogeneous, active state at low temperatures, this novel method reduces the generation of such intractable residues. This aspect is particularly vital for R&D directors focused on reducing lead time for high-purity fused ring compounds, as it reduces the burden on downstream purification steps like chromatography or extensive recrystallization.
How to Synthesize Fused Ring Compounds Efficiently
The practical implementation of this synthesis route involves a carefully orchestrated sequence of temperature controls and reagent additions. Operators must first establish an inert atmosphere, typically using nitrogen, to prevent oxidation of sensitive intermediates. The diazotization step requires heating to moderate temperatures (10°C to 100°C depending on the specific reagent) to ensure complete conversion of the amine, followed by a rapid cooldown to sub-zero temperatures for the coupling phase. Detailed standard operating procedures regarding specific stoichiometry, stirring rates, and quenching protocols are essential for reproducibility.
- Prepare a diazo compound mixture by reacting a polyphenyl precursor containing ortho-amino groups with a non-aqueous diazotization reagent (e.g., tert-butyl nitrite) in a low-freezing-point solvent at elevated temperatures.
- Cool the reaction mixture to a strictly controlled low temperature range of -20°C to -50°C to stabilize the diazo intermediate and prevent premature decomposition.
- Introduce an ultra-low loading of divalent palladium catalyst (0.05-0.2% wt) and an acid-binding agent to facilitate the intramolecular coupling reaction, yielding the target fused ring structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the implications of adopting this patented technology extend far beyond the laboratory bench. The primary value proposition lies in the drastic optimization of the cost structure associated with catalyst consumption. Palladium is a precious metal with volatile pricing, and reducing its usage from percentage levels to parts-per-thousand levels represents a substantial direct cost saving. This reduction eliminates the need for expensive catalyst recovery systems that are often required when high loadings are used, thereby simplifying the overall production infrastructure.
- Cost Reduction in Manufacturing: The most immediate financial benefit is derived from the ultra-low catalyst loading of 0.05-0.2%. In traditional processes where catalyst loading exceeds 1%, the cost of the metal itself can constitute a significant portion of the bill of materials. By minimizing this input, manufacturers can achieve a leaner cost profile without compromising on reaction efficiency. Additionally, the use of common, low-freezing-point solvents like acetonitrile and tetrahydrofuran ensures that raw material sourcing remains stable and economical, avoiding the need for exotic or hazardous reagents.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to a more resilient supply chain. Because the reaction conditions are milder and the catalyst is more stable, the risk of batch failure due to thermal runaway or catalyst deactivation is significantly mitigated. This reliability ensures consistent delivery schedules for downstream clients in the OLED and liquid crystal industries. Furthermore, the simplified purification process means that production cycles can be shortened, allowing for faster turnover and improved responsiveness to market demand fluctuations.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers distinct advantages. The reduction in heavy metal usage aligns with increasingly stringent global regulations regarding metal residues in fine chemicals. Lower metal content in the crude product reduces the load on wastewater treatment facilities and minimizes the generation of hazardous solid waste. This eco-friendly profile facilitates easier regulatory approval for new facilities and supports the long-term sustainability goals of modern chemical enterprises aiming for green manufacturing standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a clear picture of the operational benefits.
Q: How does this patent reduce palladium catalyst consumption compared to traditional methods?
A: Traditional methods often require over 1% palladium loading due to catalyst deactivation at higher temperatures. This patented process utilizes a low-temperature protocol (-20°C to -50°C) and non-aqueous conditions, which stabilizes the divalent palladium species, allowing effective catalysis at loadings as low as 0.05-0.2%.
Q: What are the primary applications for these biphenyl fused ring compounds?
A: These compounds are critical building blocks for liquid crystal materials and organic electroluminescent (OLED) materials. They are also widely utilized as intermediates in the synthesis of complex agrochemicals and pharmaceutical agents requiring rigid biphenyl scaffolds.
Q: Why is the non-aqueous diazotization step critical for yield improvement?
A: Conventional aqueous diazotization often leads to side reactions and instability of the diazonium salt. By employing non-aqueous reagents like tert-butyl nitrite in low-freezing-point solvents, the process minimizes hydrolysis and thermal decomposition, significantly boosting the conversion rate to the target fused ring product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fused Ring Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate low-temperature protocols described in CN109678648B can be executed with precision on an industrial scale. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch adheres to the highest standards required for electronic and pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this low-palladium route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to drive innovation and efficiency in your supply chain.
