Advanced Palladium-Catalyzed Carbonylation for High-Purity 2-Trifluoromethyl Imidazole Manufacturing
Introduction to Next-Generation Imidazole Synthesis
The landscape of modern pharmaceutical development is increasingly defined by the incorporation of fluorinated heterocycles, which offer superior metabolic stability and bioavailability compared to their non-fluorinated counterparts. As highlighted in recent medicinal chemistry literature, imidazole scaffolds are ubiquitous in bioactive molecules, ranging from natural products like Naamidine A to synthetic drugs such as Alcaftadine, an H1 histamine receptor antagonist. The strategic introduction of a trifluoromethyl group at the 2-position of the imidazole ring further enhances these pharmacological properties, making 2-trifluoromethyl substituted imidazoles highly coveted building blocks for drug discovery. However, accessing these complex structures has historically been fraught with synthetic challenges.
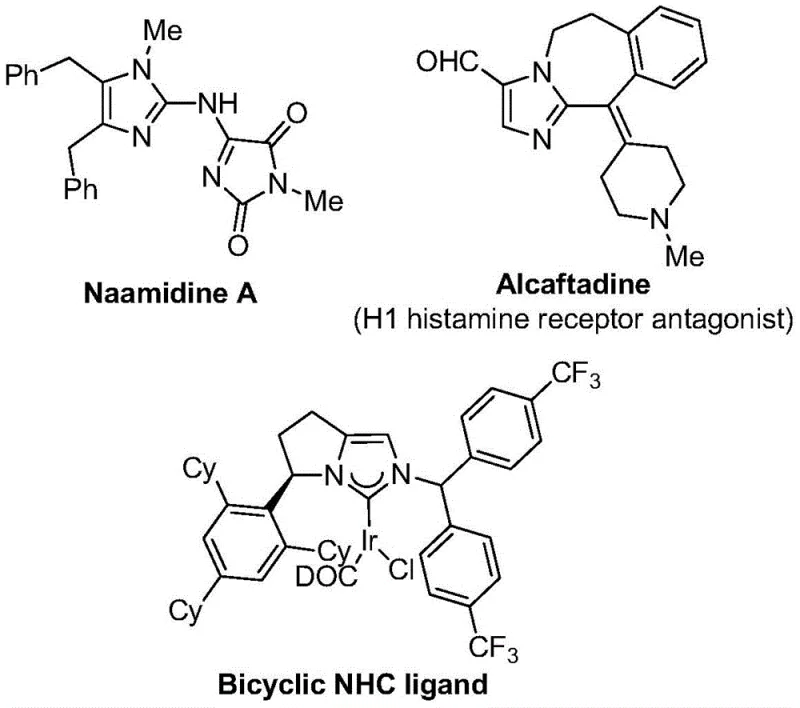
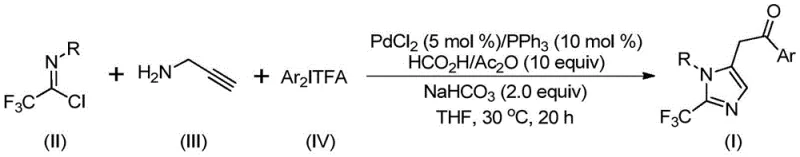
Addressing these challenges, the groundbreaking methodology detailed in Chinese Patent CN111423381B presents a transformative approach to constructing these valuable motifs. This patent discloses a robust preparation method that leverages a transition metal palladium-catalyzed carbonylation series reaction. By utilizing cheap and easily obtained starting materials—specifically trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts—this process achieves high reaction efficiency and exceptional substrate compatibility. For procurement managers and R&D directors seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant leap forward in accessing high-purity heterocyclic building blocks with streamlined operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has relied heavily on the direct reaction of specific trifluoromethyl synthons with suitable substrates. While effective in certain contexts, the commonly used synthons, such as trifluorodiazoethane and trifluoroethylimidoyl halides, present substantial limitations for industrial application. Trifluorodiazoethane, for instance, is inherently unstable and poses significant safety risks due to its explosive potential, necessitating specialized handling equipment and rigorous safety protocols that drive up operational costs. Furthermore, existing methods often suffer from narrow substrate scope, failing to accommodate diverse functional groups without compromising yield or selectivity. These constraints severely limit the designability of the final molecular architecture, forcing chemists to compromise on ideal substitution patterns during the lead optimization phase.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes a multicomponent coupling strategy that elegantly bypasses these historical bottlenecks. By employing trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts as the core reactants, the process enables the efficient construction of the imidazole core in a single pot. This approach not only utilizes readily available and inexpensive aromatic amines and alkynylamines but also exhibits remarkable tolerance for various functional groups, including methyl, tert-butyl, chlorine, bromine, and nitro substituents. The ability to synthesize diversified 2-trifluoromethyl imidazoles through simple substrate design significantly broadens the utility of this method, allowing for the rapid generation of compound libraries for biological screening. This flexibility is crucial for cost reduction in API manufacturing, as it allows for late-stage functionalization and modular assembly of complex drug candidates.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The success of this synthesis lies in its sophisticated yet operationally simple catalytic cycle, which orchestrates multiple bond-forming events with high precision. The reaction initiates with the formation of an intermolecular carbon-nitrogen bond promoted by the alkaline additive, yielding a trifluoroacetamidine intermediate. Following isomerization, the palladium catalyst engages the alkyne moiety of the propargylamine derivative, undergoing palladation to generate a key alkenyl palladium intermediate. This species subsequently isomerizes to a more stable alkyl palladium intermediate, setting the stage for the critical carbonylation step.
Uniquely, this process avoids the use of external carbon monoxide gas. Instead, it employs a mixture of formic acid and acetic anhydride as a safe and controllable carbon monoxide surrogate. Under the reaction conditions, this mixture releases CO in situ, which inserts into the palladium-carbon bond to form an acyl palladium intermediate. The cycle concludes with the oxidative addition of the diaryl iodonium salt to generate a transient tetravalent palladium species, followed by reductive elimination to release the final 2-trifluoromethyl-substituted imidazole product and regenerate the active catalyst. This mechanistic pathway ensures high atom economy and minimizes the formation of toxic byproducts, aligning with modern green chemistry principles essential for sustainable commercial scale-up of complex polymer additives and pharmaceutical intermediates.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized conditions outlined in the patent to ensure maximum yield and purity. The protocol is designed for operational ease, utilizing common organic solvents like tetrahydrofuran (THF) which effectively dissolve all reactants and promote high conversion rates. The reaction proceeds under remarkably mild thermal conditions, typically at 30°C, which significantly reduces energy consumption compared to traditional high-temperature reflux methods. Detailed standardized synthesis steps, including precise molar ratios and workup procedures, are provided in the technical guide below to facilitate immediate adoption by process chemists.
- Prepare the reaction mixture by adding palladium chloride (5 mol%), triphenylphosphine (10 mol%), sodium bicarbonate, acetic anhydride, and formic acid into an organic solvent such as THF.
- Introduce the substrates: trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt into the reaction vessel under stirring.
- Maintain the reaction at 30°C for 18 to 20 hours, then filter the mixture and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the transition to this novel synthetic route offers compelling economic and logistical benefits. The reliance on commodity chemicals such as formic acid, acetic anhydride, and sodium bicarbonate eliminates the dependency on specialized, high-cost reagents that are often subject to volatile market pricing and supply disruptions. Furthermore, the mild reaction temperature of 30°C drastically simplifies the engineering requirements for reactor vessels, removing the need for expensive heating jackets or high-pressure containment systems typically associated with carbonylation reactions. This translates directly into lower capital expenditure (CAPEX) for manufacturing facilities and reduced operational expenditure (OPEX) through lower energy usage.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive starting materials and a highly efficient catalytic system. By avoiding the use of hazardous gases like carbon monoxide and unstable diazo compounds, the method significantly reduces the costs associated with safety compliance, waste disposal, and specialized storage infrastructure. The high reaction efficiency, with yields reaching up to 97% for certain substrates, ensures minimal raw material waste, thereby optimizing the overall cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against variations in substrate electronics ensures a consistent supply of high-quality intermediates. Since the starting materials, including various substituted aromatic amines and aryl boronic acids (precursors to the iodonium salts), are widely available from global chemical suppliers, the risk of raw material shortages is minimized. This reliability is critical for maintaining uninterrupted production schedules and reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug development timelines are met without delay.
- Scalability and Environmental Compliance: The simplicity of the post-treatment procedure, which involves basic filtration and standard column chromatography, facilitates easy scale-up from gram to kilogram and eventually to ton-scale production. The use of THF as a solvent, which is easily recoverable and recyclable, combined with the absence of heavy metal waste streams typical of other transition metal catalyses, supports stringent environmental compliance standards. This eco-friendly profile not only mitigates regulatory risks but also enhances the corporate sustainability metrics of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical teams evaluating this technology for integration into their existing workflows.
Q: What is the primary advantage of using formic acid and acetic anhydride in this synthesis?
A: This combination serves as a safe and effective carbon monoxide surrogate, generating CO in situ under mild conditions (30°C), thereby eliminating the safety hazards associated with handling high-pressure CO gas cylinders.
Q: Does this method tolerate diverse functional groups on the aryl rings?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens, nitro, and trifluoromethyl substituents.
Q: Is the palladium catalyst loading economically viable for large-scale production?
A: The process utilizes a relatively low catalyst loading (5 mol% PdCl2), and since the starting materials are commercially available and inexpensive, the overall cost profile is favorable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results observed in the laboratory can be seamlessly translated to industrial reality. We are committed to delivering 2-trifluoromethyl imidazole intermediates with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch.
We invite pharmaceutical partners to leverage our technical expertise to optimize their supply chains. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next breakthrough therapy is built on a foundation of chemical excellence and supply chain security.
