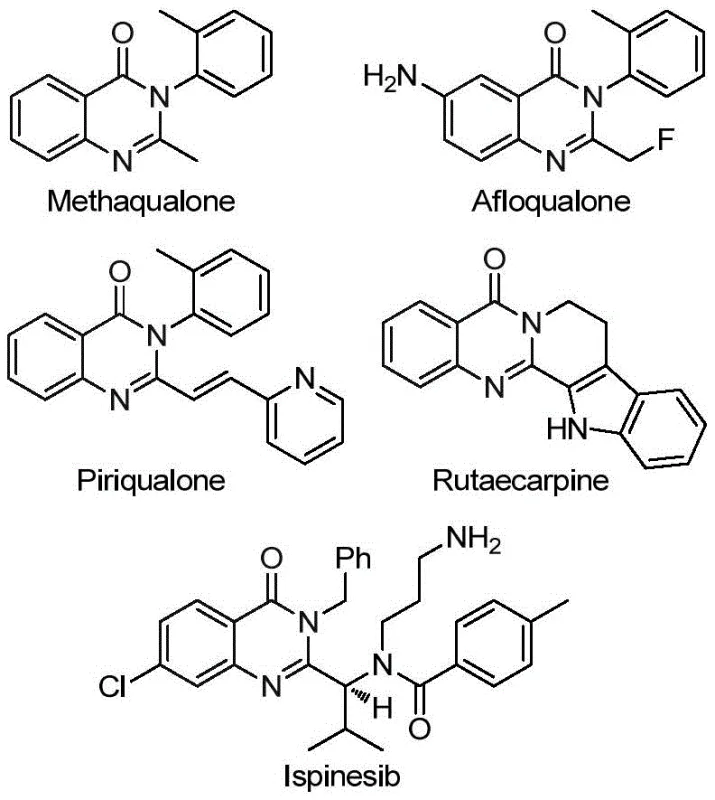
Advanced Palladium-Catalyzed Carbonylation for High-Purity Quinazolinone Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic methodologies to access nitrogen-containing heterocycles, particularly quinazolinones, which serve as privileged scaffolds in medicinal chemistry. Patent CN113045503A discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing critical bottlenecks in current manufacturing processes. This innovation leverages a transition metal palladium-catalyzed carbonylation tandem reaction, utilizing cheap and easily obtainable trifluoroethylimidoyl chloride and amines as starting materials. The introduction of the trifluoromethyl group is strategically significant, as it enhances key physicochemical properties of drug candidates, including metabolic stability, lipophilicity, and bioavailability. By employing TFBen as a solid carbon monoxide surrogate, this process circumvents the safety hazards associated with traditional high-pressure CO gas usage, offering a safer and more operationally simple pathway for producing high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl substituted quinazolinones has been plagued by significant operational and economic challenges that hinder large-scale adoption. Conventional routes often rely on the cyclization of anthranilamide with ethyl trifluoroacetate or trifluoroacetic anhydride, reactions that typically require harsh conditions and generate substantial acidic waste. Alternative methods involving isatoic anhydride or T3P-promoted tandem reactions frequently suffer from narrow substrate scopes, limiting the structural diversity accessible to medicinal chemists. Furthermore, many existing protocols necessitate the use of unstable intermediates or expensive activating agents, driving up the cost of goods sold (COGS) and complicating supply chain logistics. The reliance on gaseous carbon monoxide in some carbonylation approaches introduces severe safety risks and requires specialized high-pressure equipment, creating barriers for facilities lacking such infrastructure.
The Novel Approach
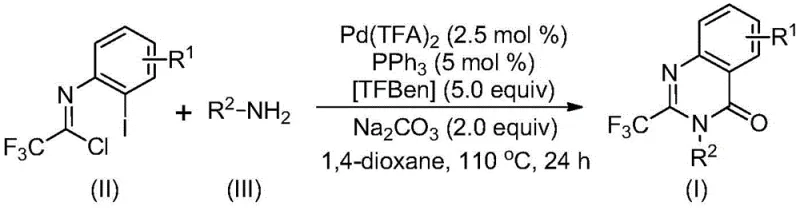
The methodology described in the patent represents a paradigm shift by utilizing a palladium-catalyzed system that operates under relatively mild thermal conditions at 110°C. Instead of hazardous gas cylinders, the process employs TFBen, a solid benzoate derivative that releases carbon monoxide in situ upon heating, ensuring a controlled and safe reaction environment. This approach utilizes readily available trifluoroethylimidoyl chlorides and a wide variety of amines, significantly broadening the chemical space available for exploration. The reaction exhibits remarkable efficiency, with experimental data showing high conversion rates and yields across diverse substrates, including those with electron-withdrawing and electron-donating groups. This versatility allows for the rapid generation of compound libraries for drug discovery while maintaining a streamlined workflow suitable for industrial scale-up.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The catalytic cycle begins with an alkali-promoted intermolecular carbon-nitrogen bond coupling between the amine and the imidoyl chloride, likely forming a trifluoroacetamidine derivative intermediate. Subsequently, the palladium catalyst, generated from palladium trifluoroacetate and triphenylphosphine, undergoes oxidative insertion into the carbon-iodine bond of the aromatic ring to form a divalent palladium species. As the temperature rises, the TFBen additive decomposes to release carbon monoxide, which then inserts into the carbon-palladium bond to generate an acyl palladium intermediate. This step is crucial as it builds the carbonyl functionality directly into the heterocyclic core without external gas feed. The presence of base facilitates the formation of a palladium-nitrogen bond, leading to a seven-membered ring palladium intermediate, which finally undergoes reductive elimination to release the desired 2-trifluoromethyl-substituted quinazolinone product and regenerate the active catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over traditional acid-mediated cyclizations. The use of a defined palladium catalyst system with specific ligands ensures high regioselectivity, minimizing the formation of isomeric byproducts that are difficult to separate. The mild reaction conditions prevent the degradation of sensitive functional groups that might occur under the strongly acidic or high-temperature conditions of older methods. Furthermore, the in situ generation of CO from TFBen avoids the local concentration spikes of carbon monoxide that can lead to side reactions in gas-phase carbonylations. The post-treatment process is equally straightforward, involving simple filtration and silica gel column chromatography, which effectively removes palladium residues and organic impurities, ensuring the final product meets stringent purity specifications required for pharmaceutical applications.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
To implement this synthesis effectively, precise control over stoichiometry and reaction parameters is essential to maximize yield and minimize catalyst loading. The protocol recommends a molar ratio of trifluoroethylimidoyl chloride to amine to palladium catalyst of approximately 1:2.5:0.025, ensuring the amine is in excess to drive the initial coupling step to completion. The reaction is conducted in an aprotic solvent such as 1,4-dioxane, which provides optimal solubility for all reagents and stabilizes the catalytic intermediates. Maintaining the temperature at 110°C for a duration of 16 to 30 hours allows sufficient time for the slow carbonylation and cyclization steps to proceed without decomposing the product. Detailed standardized operating procedures for this synthesis, including specific workup and purification guidelines, are provided in the technical documentation below.
- Combine palladium trifluoroacetate, triphenylphosphine, sodium carbonate, TFBen, and organic solvent in a reaction vessel.
- Add trifluoroethylimidoyl chloride and the corresponding amine substrate to the mixture under stirring.
- Heat the reaction mixture to 110°C for 16-30 hours, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, this novel synthetic route presents a compelling value proposition by addressing key pain points related to raw material availability and process safety. The shift from hazardous gaseous reagents to stable solid surrogates like TFBen drastically simplifies logistics, eliminating the need for specialized gas handling infrastructure and reducing regulatory compliance burdens associated with toxic gas storage. The starting materials, specifically the trifluoroethylimidoyl chlorides and various amines, are commercially available and inexpensive, which translates to a significantly reduced cost of raw materials compared to proprietary or complex precursors used in legacy methods. Additionally, the high atom economy and efficient catalyst turnover suggest a substantial reduction in waste generation, aligning with modern green chemistry initiatives and lowering disposal costs.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and expensive activating agents leads to drastic capital expenditure savings and lower operational costs. By using a solid CO source, the process avoids the premium pricing and safety costs associated with handling carbon monoxide gas, while the high experimental yields reported in the patent indicate minimal material loss during production. The use of common solvents like dioxane and standard purification techniques further ensures that the process remains economically viable for large-scale manufacturing without requiring exotic reagents.
- Enhanced Supply Chain Reliability: The broad substrate tolerance of this method means that supply chain disruptions for specific precursors can be mitigated by switching to alternative amines or substituted imidoyl chlorides without re-optimizing the entire process. Since the core reagents are commodity chemicals rather than custom-synthesized intermediates, lead times for raw material procurement are significantly shortened. This flexibility ensures continuous production capability even when specific niche chemicals face global shortages, providing a robust buffer against market volatility.
- Scalability and Environmental Compliance: The reaction has been demonstrated to work efficiently on gram scales with potential for expansion to multi-kilogram batches, supported by the simplicity of the workup procedure which involves basic filtration and chromatography. The avoidance of harsh acids and the use of a closed system for CO generation minimize the release of volatile organic compounds and acidic fumes, facilitating easier adherence to environmental protection regulations. This cleaner profile reduces the burden on wastewater treatment facilities and lowers the overall environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this palladium-catalyzed technology. These answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity for R&D teams evaluating this route for their own pipelines. Understanding these nuances is critical for assessing the feasibility of adopting this method for specific target molecules.
Q: What is the carbon monoxide source in this novel synthesis method?
A: The method utilizes TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide substitute, which releases CO in situ under heating, eliminating the need for hazardous high-pressure CO gas cylinders.
Q: What is the substrate compatibility of this palladium-catalyzed reaction?
A: The reaction demonstrates excellent substrate tolerance, accommodating various substituents on the aryl ring (such as F, Cl, Br, Me, CF3) and diverse amine structures including alkyl, cycloalkyl, benzyl, and substituted aryl amines.
Q: Can this method be applied to the synthesis of complex drug molecules?
A: Yes, the patent explicitly demonstrates the successful application of this methodology in the efficient, high-yield total synthesis of the bioactive natural product Rutaecarpine through a three-step sequence.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
As the demand for fluorinated heterocycles continues to surge in oncology and anti-inflammatory drug development, securing a partner with deep technical expertise in C-H activation and carbonylation chemistry is paramount. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions with rigorous QC labs dedicated to maintaining stringent purity specifications, guaranteeing that every batch of intermediate meets the highest quality standards required by global regulatory bodies.
We invite you to leverage our technical capabilities to optimize your supply chain and reduce time-to-market for your critical drug candidates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis platforms can deliver superior value and reliability for your organization.
