Revolutionizing Folic Acid Production: A Safe, High-Yield Heteropoly Acid Catalytic Route for Global API Manufacturers
Revolutionizing Folic Acid Production: A Safe, High-Yield Heteropoly Acid Catalytic Route for Global API Manufacturers
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the production of essential vitamins and active pharmaceutical ingredients. A groundbreaking advancement in this domain is detailed in patent CN110105362B, which introduces a safe and green synthesis method for folic acid (Vitamin B9) catalyzed by heteropoly acids. This technology represents a paradigm shift from traditional, hazard-laden chlorination processes to a streamlined, one-pot condensation strategy. By leveraging the unique bifunctional properties of heteropoly acids—specifically phosphorus-containing variants like H4Cs2[PMo11V1O40]—this method achieves exceptional conversion rates while drastically reducing the environmental footprint. For global supply chain leaders and R&D directors, this innovation offers a compelling solution to the longstanding challenges of purity control and process safety in vitamin manufacturing.
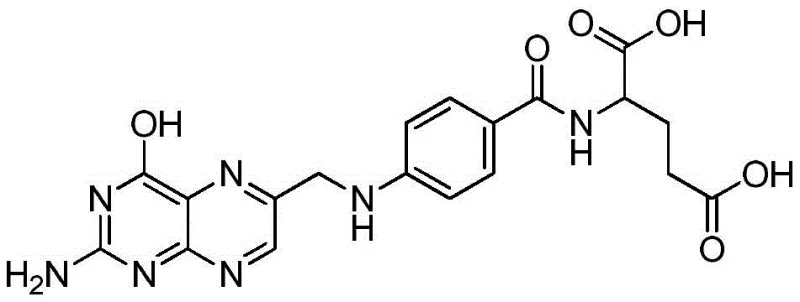
Folic acid, chemically known as N-[4-[(2-amino-4-oxo-1,4-dihydro-6-pteridinyl)methylamino]benzoyl]-L-glutamic acid, is a critical nutrient for cell growth and reproduction. The structural complexity of this molecule has historically necessitated multi-step syntheses involving hazardous reagents. The new methodology described in the patent simplifies this architecture by directly constructing the key pteridine-benzoyl linkage through a catalytic oxidative condensation. This approach not only enhances the atom economy of the reaction but also ensures that the final product meets the stringent purity specifications required for pharmaceutical-grade applications, positioning it as a superior alternative for reliable API intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of folic acid has been plagued by significant safety and environmental drawbacks, primarily stemming from the reliance on halogenated intermediates. The most common legacy route involves the synthesis of trichloroacetone via the chlorination of acetone, a process that requires the handling of elemental chlorine gas. As illustrated in the traditional reaction pathways, this step is inherently dangerous due to the exothermic nature of chlorination and the high toxicity of chlorine, which poses severe risks of leakage and explosion. Furthermore, the purification of trichloroacetone is notoriously difficult, often resulting in a mixture of dichloro and tetrachloro byproducts that compromise the quality of the subsequent cyclization steps.
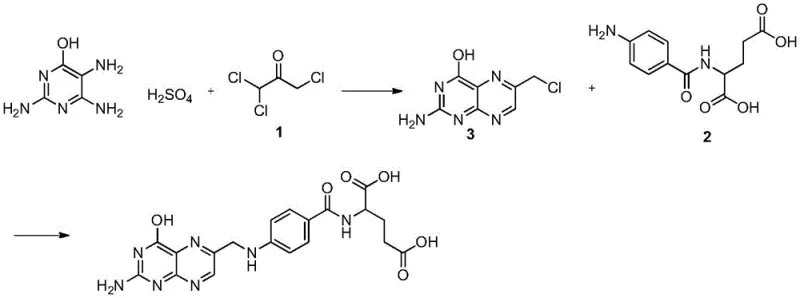
Beyond safety concerns, the conventional trichloroacetone route suffers from poor atom efficiency and substantial waste generation. The absorption of hydrogen chloride tail gas generates high-salinity wastewater that is complex and costly to treat, creating a significant burden on environmental compliance systems. Alternative routes, such as those utilizing dibromoacrolein or tetramethoxy-2-propanol, attempt to mitigate some purity issues but introduce new problems regarding raw material costs and overall yield. For instance, the dibromoacrolein pathway, while capable of producing high-purity products, involves expensive bromine reagents and suffers from low total yields (around 36.9%), making it economically unviable for large-scale commercial production. These cumulative inefficiencies highlight the urgent need for a greener, more cost-effective manufacturing strategy.
The Novel Approach
The innovative process disclosed in patent CN110105362B circumvents these historical bottlenecks by employing a direct condensation of acrolein, 2,5,6-triamino-4-hydroxypyrimidine, and p-aminobenzoate. This novel approach eliminates the need for pre-synthesized halogenated intermediates entirely, replacing them with readily available and inexpensive acrolein. The reaction is driven by a heteropoly acid catalyst, which facilitates the formation of the critical methylene bridge between the pyrimidine and benzoyl moieties in a single, efficient step. This "one-pot" strategy significantly shortens the synthetic timeline, reducing the number of unit operations and minimizing the potential for impurity accumulation that typically occurs during intermediate isolation and purification stages.
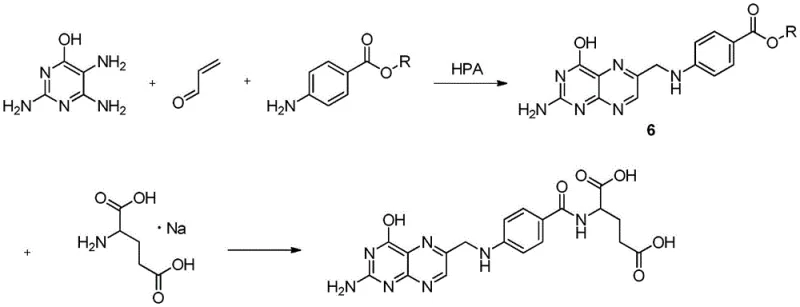
Moreover, the new method demonstrates remarkable versatility regarding reaction conditions and substrate scope. The patent specifies that various esters of p-aminobenzoic acid (methyl, ethyl, propyl, etc.) can be utilized effectively, allowing manufacturers to optimize based on local raw material availability and cost. The reaction proceeds under mild thermal conditions (30-60°C), which contrasts sharply with the harsh conditions often required in traditional halogenation chemistries. This mildness not only enhances operational safety but also preserves the integrity of sensitive functional groups, leading to a cleaner reaction profile. The result is a light yellow solid intermediate (Intermediate 6) that can be isolated with high purity (>98%) through simple filtration and decolorization, setting the stage for a highly efficient final coupling with L-sodium glutamate.
Mechanistic Insights into Heteropoly Acid-Catalyzed Condensation
The efficacy of this synthesis hinges on the unique physicochemical properties of heteropoly acids (HPAs). These compounds, characterized by their Keggin or Dawson structures, function as bifunctional catalysts possessing both strong Brønsted acidity and redox capabilities. In the context of folic acid synthesis, the HPA acts as a proton donor to activate the carbonyl group of acrolein, facilitating a nucleophilic attack by the amino groups of the triaminopyrimidine. Simultaneously, the redox-active metal centers (such as Vanadium and Molybdenum within the catalyst structure) assist in the oxidative aromatization required to form the stable pteridine ring system. This dual functionality allows the reaction to proceed rapidly and completely without the need for additional oxidants or harsh acidic media that could degrade the product.
From an impurity control perspective, the mechanism offers distinct advantages over free acid catalysis. The quasi-liquid phase behavior of HPAs ensures a homogeneous distribution of active sites, which minimizes localized hot spots that often lead to polymerization or charring of organic substrates. Furthermore, the specific stoichiometry of the catalyst, such as H4Cs2[PMo11V1O40], can be tuned to maximize selectivity for the desired 6-substituted pteridine isomer. Experimental data from the patent indicates that optimizing the molar ratio of the catalyst to substrates (e.g., 0.01 to 0.3 equivalents) is critical for suppressing side reactions. This precise control over the catalytic cycle ensures that the final crude product exhibits purity levels exceeding 99.1%, drastically reducing the burden on downstream purification processes.
How to Synthesize Folic Acid Efficiently
The implementation of this heteropoly acid-catalyzed route offers a straightforward pathway for manufacturing facilities aiming to upgrade their production capabilities. The process is divided into two primary stages: the formation of the pteridine-benzoyl intermediate and the subsequent amidation with glutamic acid. The simplicity of the workup procedures—primarily involving filtration, washing, and crystallization—makes this method particularly attractive for plants looking to reduce solvent consumption and waste disposal costs. The following guide outlines the critical operational parameters derived from the patent examples to ensure optimal yield and quality.
- Condense 2,5,6-triamino-4-hydroxypyrimidine, p-aminobenzoate, and acrolein in an alcohol solvent using a phosphorus-containing heteropoly acid catalyst at 30-60°C to form Intermediate 6.
- Isolate Intermediate 6 via filtration, washing, and activated carbon decolorization to achieve high purity (>98%).
- React Intermediate 6 with L-sodium glutamate in an alcohol-water mixture at 60-120°C, followed by cooling and crystallization to obtain pure Folic Acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this HPA-catalyzed process translates into tangible strategic benefits beyond mere technical performance. The elimination of chlorine gas and bromine reagents removes a major category of hazardous material from the supply chain, thereby simplifying logistics, storage, and regulatory compliance. Traditional chlorination processes often require specialized infrastructure and rigorous safety protocols that inflate operational expenditures. By switching to a system based on acrolein and solid acid catalysts, manufacturers can significantly reduce their exposure to volatile raw material markets and the high costs associated with transporting and storing toxic gases.
- Cost Reduction in Manufacturing: The economic impact of this new route is profound, driven primarily by the simplification of the process flow. By consolidating multiple steps into a single condensation reaction, the method reduces labor hours, energy consumption for heating and cooling, and the volume of solvents required for intermediate purifications. The high yield of the intermediate (consistently above 92%) means that less raw material is wasted, directly lowering the cost of goods sold (COGS). Additionally, the avoidance of expensive halogenated reagents and the ability to use commodity alcohols as solvents further contribute to substantial cost savings in folic acid manufacturing.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the scarcity or regulatory restrictions on key precursors. Acrolein and p-aminobenzoates are widely produced commodity chemicals with stable global supply chains, unlike specialized halogenated intermediates which may be subject to production quotas or environmental shutdowns. The robustness of the HPA catalyst, which can be recovered and potentially reused, adds another layer of security against raw material price fluctuations. This reliability ensures that production schedules can be maintained without the frequent interruptions caused by the sourcing difficulties inherent in older, more complex synthetic routes.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the ability to demonstrate a "green" manufacturing process is a significant competitive advantage. This method generates significantly less saline wastewater compared to the hydrochloric acid byproducts of the trichloroacetone route. The absence of heavy metal catalysts or toxic halogens simplifies effluent treatment, allowing facilities to meet discharge standards with lower capital investment in waste processing equipment. Furthermore, the mild reaction conditions and straightforward scale-up profile make it easier to expand production capacity from pilot scales to multi-ton commercial batches without encountering the non-linear engineering challenges typical of hazardous exothermic reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this heteropoly acid-catalyzed synthesis. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio.
Q: How does the heteropoly acid catalyst improve safety compared to traditional chlorination methods?
A: Traditional methods rely on hazardous chlorine gas and trichloroacetone, posing significant explosion and toxicity risks. The heteropoly acid method utilizes acrolein and a solid acid catalyst, eliminating the need for toxic halogenation reagents and reducing process safety hazards significantly.
Q: What yields and purity levels can be expected from this new synthetic route?
A: The patent data indicates that the intermediate synthesis achieves yields between 92.8% and 96.2% with purity exceeding 98%. The final folic acid product demonstrates yields up to 95.1% with purity levels reaching 99.8%, surpassing many conventional multi-step processes.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process is designed for scalability. It uses readily available raw materials like acrolein and avoids complex purification steps associated with halogenated byproducts. The mild reaction conditions (30-60°C) and simplified workup make it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Folic Acid Supplier
The technological advancements detailed in patent CN110105362B underscore the immense potential for optimizing folic acid production through green chemistry principles. At NINGBO INNO PHARMCHEM, we recognize that adopting such innovative pathways requires a partner with deep technical expertise and a commitment to quality. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of folic acid or intermediate meets the highest international pharmacopeial standards.
We invite forward-thinking pharmaceutical companies and bulk buyers to collaborate with us on leveraging this superior synthetic route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing you to validate how this green, high-yield process can enhance your supply chain resilience and drive down long-term production costs.
