Advanced Convergent Synthesis of Renin Inhibitor Intermediates for Commercial Scale-up
Introduction to Novel Renin Inhibitor Synthesis
The pharmaceutical industry continuously seeks more efficient pathways for producing complex active pharmaceutical ingredients (APIs), particularly for cardiovascular treatments like renin inhibitors. Patent CN101952242A introduces a groundbreaking convergent synthesis strategy for preparing 2(S),4(S),5(S),7(S)-2,7-dialkyl-4-hydroxy-5-amino-8-aryl-octamide derivatives, most notably the antihypertensive drug aliskiren. This technology addresses critical bottlenecks in traditional manufacturing by shifting the introduction of the essential nitrogen atom to an earlier stage in the synthetic sequence. By utilizing novel nitrogen-containing intermediates such as lactones of formula (8) and chiral aldehydes of formula (10), the process achieves superior stereochemical control at the C-5 position. This technical advancement is pivotal for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with consistent batch-to-batch reproducibility.
The significance of this patent lies in its ability to bypass the limitations of linear synthesis, where stereochemical integrity is often compromised during late-stage functionalization. The disclosed method leverages the inherent chirality of readily available starting materials to dictate the configuration of the final product. This approach not only streamlines the production workflow but also aligns with modern green chemistry principles by reducing the reliance on stoichiometric chiral auxiliaries. For procurement and supply chain leaders, understanding this underlying technology is essential for evaluating potential partners who can offer cost reduction in API manufacturing through process intensification and yield optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of renin inhibitors has relied heavily on linear strategies that construct the carbon backbone before introducing the critical amine functionality. As documented in prior art such as Sandham et al. (Tetrahedron Letters 2000), these conventional routes often involve coupling an aromatic moiety with an alcohol fragment, followed by a cumbersome substitution to install the nitrogen group. A major drawback of this approach is the poor diastereoselectivity observed during the coupling step, frequently resulting in a near 1:1 mixture of epimers (e.g., 55:45 ratio). Separating these closely related diastereomers requires extensive chromatography or crystallization steps, which drastically reduces overall yield and increases production costs. Furthermore, the use of stoichiometric chiral auxiliaries to induce asymmetry adds significant material costs and generates substantial chemical waste, posing challenges for environmental compliance and scalability.
The Novel Approach
In stark contrast, the methodology described in CN101952242A employs a convergent synthesis that couples a pre-functionalized nitrogen-containing building block with the aromatic component. This strategy ensures that the nitrogen atom is already present in the correct oxidation state and stereochemical environment before the final assembly of the molecule. The core innovation involves the use of chiral aldehydes (Formula 10) which are converted into stable lactone nitriles (Formula 8a) or protected cyanohydrins (Formula 7a). These intermediates serve as robust electrophiles or nucleophiles that can be coupled with organometallic reagents derived from the aromatic portion (Formula 2). 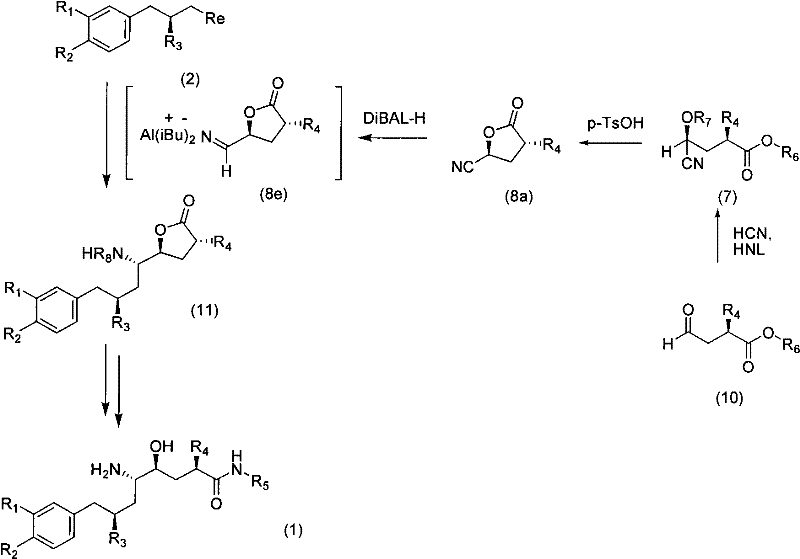 This convergence allows for independent optimization of the two main fragments, significantly enhancing process flexibility. The result is a synthetic route that delivers the desired 5(S) configuration with markedly improved selectivity, minimizing the need for difficult purification steps and enabling a more direct path to the final API.
This convergence allows for independent optimization of the two main fragments, significantly enhancing process flexibility. The result is a synthetic route that delivers the desired 5(S) configuration with markedly improved selectivity, minimizing the need for difficult purification steps and enabling a more direct path to the final API.
Mechanistic Insights into HNL-Catalyzed Stereoselective Synthesis
The heart of this novel synthetic pathway is the precise establishment of chirality at the C-2 and C-5 positions, achieved through a combination of biocatalysis and classical organic transformations. The process begins with the preparation of a chiral aldehyde (Formula 10), which serves as the foundational scaffold for the nitrogen-containing segment. This aldehyde undergoes a highly stereoselective cyanohydrin formation catalyzed by hydroxynitrile lyase (HNL). As detailed in the patent examples, reacting the aldehyde with HCN in the presence of (S)-HNL yields the corresponding hydroxynitrile with exceptional enantiomeric excess, often exceeding 94% ee. This enzymatic step is crucial as it sets the absolute configuration without the need for expensive metal-based chiral catalysts or resolution steps. Following cyanohydrin formation, the intermediate undergoes acid-catalyzed lactonization, typically using p-toluenesulfonic acid, to form the cyclic lactone nitrile (Formula 8a). 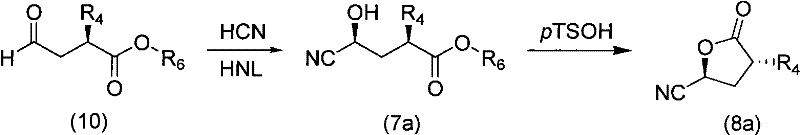 This cyclization not only protects the hydroxyl group but also locks the stereochemistry, preventing epimerization during subsequent handling.
This cyclization not only protects the hydroxyl group but also locks the stereochemistry, preventing epimerization during subsequent handling.
Impurity control is inherently built into this mechanism due to the high specificity of the enzymatic step and the thermodynamic stability of the lactone ring. The patent describes that even if a mixture of diastereomers is formed initially, the conditions can be tuned to favor the thermodynamically preferred isomer through epimerization or selective crystallization. Furthermore, the subsequent coupling of these nitrogen-rich intermediates with the aromatic Grignard or organolithium reagents proceeds with high chemoselectivity. The use of additives like Lewis acids or specific metal complexes (e.g., MnCl2, FeCl3) can further enhance the diastereoselectivity of the coupling reaction. This multi-layered approach to stereocontrol ensures that the final impurity profile is manageable and compliant with stringent regulatory standards for pharmaceutical intermediates, providing R&D directors with confidence in the robustness of the technology transfer.
How to Synthesize Aliskiren Intermediate Efficiently
Implementing this convergent synthesis requires careful attention to reaction conditions, particularly regarding temperature control and reagent stoichiometry during the organometallic coupling phase. The process generally involves three critical stages: the preparation of the chiral nitrogenous fragment, the generation of the aromatic organometallic species, and their subsequent union. Detailed operational parameters, such as the specific molar ratios of DiBAL-H for nitrile reduction or the exact temperature profiles for the Grignard formation, are essential for reproducing the high yields reported in the patent examples. For technical teams looking to adopt this methodology, it is recommended to follow the standardized protocols outlined in the experimental section of the source documentation to ensure optimal stereochemical outcomes. The detailed standardized synthesis steps are provided in the guide below.
- Preparation of the chiral aldehyde precursor (Formula 10) via oxidative cleavage or asymmetric hydrogenation to establish the C-2 stereocenter.
- Stereoselective introduction of the nitrogen functionality using HNL-catalyzed cyanohydrin formation followed by acid-catalyzed lactonization to form the key lactone nitrile (Formula 8a).
- Convergent coupling of the nitrogen-containing building block with the aromatic organometallic reagent (Formula 2) under controlled conditions to yield the final protected intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this advanced synthetic route offers profound strategic benefits for procurement and supply chain management, primarily driven by the simplification of the manufacturing process and the elimination of costly reagents. By shifting to a convergent strategy that utilizes enzymatic catalysis, manufacturers can achieve substantial cost savings through the reduction of raw material consumption and waste disposal fees. The removal of stoichiometric chiral auxiliaries, which are often expensive and difficult to recover, directly lowers the bill of materials. Additionally, the improved diastereoselectivity means that less material is lost to purification processes, effectively increasing the throughput of existing production assets without the need for capital-intensive expansion. This efficiency translates into a more competitive pricing structure for the final intermediate, allowing buyers to negotiate better terms with their reliable pharmaceutical intermediate supplier.
- Cost Reduction in Manufacturing: The process eliminates the need for stoichiometric amounts of expensive chiral resolving agents and reduces the number of isolation steps required. By achieving high enantiomeric excess early in the sequence via biocatalysis, the overall yield is significantly enhanced, leading to lower cost per kilogram of the final product. This efficiency is critical for maintaining margins in a competitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as the chiral aldehydes and aromatic chlorides, are derived from commodity chemicals with established global supply chains. This reduces the risk of supply disruptions associated with specialized, single-source reagents. Furthermore, the robustness of the enzymatic and coupling steps ensures consistent production schedules, minimizing lead time for high-purity pharmaceutical intermediates and allowing for better inventory planning.
- Scalability and Environmental Compliance: The use of enzymatic catalysis operates under mild conditions, reducing energy consumption and the generation of hazardous byproducts. This aligns with increasingly strict environmental regulations and corporate sustainability goals. The convergent nature of the synthesis also facilitates easier scale-up from pilot plant to commercial production, as the key coupling reactions are well-understood organometallic transformations that can be safely managed in large reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this convergent synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers below reflect the specific advantages of the nitrogen-first strategy and the role of biocatalysis in modern API production.
Q: How does this convergent synthesis improve stereocontrol compared to linear methods?
A: By introducing the nitrogen functionality early in the synthesis via a chiral aldehyde precursor, the method avoids the poor diastereoselectivity (often 55:45) associated with late-stage amination in conventional linear routes, ensuring higher purity of the desired 5(S) epimer.
Q: What are the key advantages of using enzymatic catalysis in this process?
A: The use of hydroxynitrile lyase (HNL) for cyanohydrin formation allows for high enantiomeric excess (>94% ee) under mild conditions, eliminating the need for stoichiometric chiral auxiliaries and reducing hazardous waste generation.
Q: Is this synthetic route suitable for large-scale manufacturing?
A: Yes, the convergent nature of the synthesis reduces the total number of steps and utilizes robust reactions like Grignard coupling and enzymatic catalysis, which are well-suited for scaling from kilogram to multi-ton production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aliskiren Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to maintain a competitive edge in the pharmaceutical sector. Our team of expert chemists has extensively evaluated the convergent synthesis pathway described in CN101952242A and possesses the technical capability to execute this complex chemistry with precision. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our facility is equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of renin inhibitor intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced manufacturing capability for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this convergent route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your development timeline. Let us help you optimize your supply chain and bring life-saving medications to market faster and more efficiently.
