Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
Introduction to Next-Generation Triazole Synthesis
The pharmaceutical industry continuously seeks robust and efficient pathways to construct privileged heterocyclic scaffolds that serve as the backbone for modern therapeutics. A significant breakthrough in this domain is documented in patent CN114920707B, which discloses a highly practical preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This specific class of molecules is not merely academic; it represents the core structural motif found in blockbuster drugs such as Sitagliptin, various anticonvulsants, Factor IXa inhibitors, and GlyT1 inhibitors, as illustrated by the diverse bioactive molecules shown below. The presence of the trifluoromethyl group is critical, as it significantly enhances the metabolic stability, lipophilicity, and binding affinity of the parent drug candidates, making the efficient construction of this scaffold a high-priority objective for process chemistry teams globally.
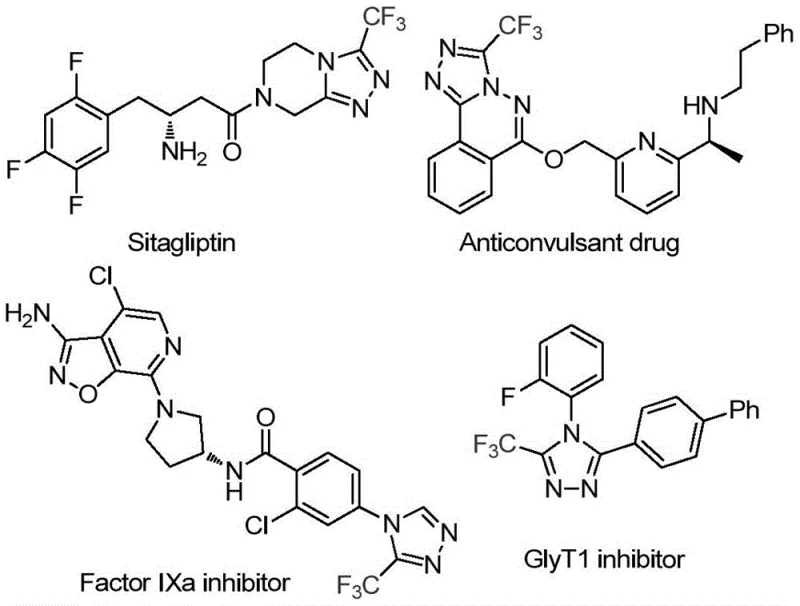
The innovation described in this patent moves beyond traditional synthetic limitations by utilizing N,N-dimethylformamide (DMF), a ubiquitous organic solvent, as the exclusive carbon source for the ring construction. This approach transforms a common commodity chemical into a valuable reactive synthon, thereby streamlining the synthetic route. For R&D directors and procurement specialists alike, this methodology offers a compelling value proposition: it merges reaction simplicity with high atom economy. By leveraging the dual functionality of DMF as both solvent and reactant, the process inherently reduces the number of unit operations and raw material inputs required, setting a new standard for the cost-effective manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant operational challenges that hinder large-scale adoption. Traditional routes often rely on the condensation of hydrazides with nitriles or amidines under harsh thermal conditions, frequently necessitating the use of expensive transition metal catalysts or toxic reagents to drive the cyclization forward. Furthermore, many existing protocols demand strictly anhydrous and anaerobic environments, requiring specialized equipment such as gloveboxes or Schlenk lines, which dramatically increases capital expenditure and operational overhead. The reliance on specialized C1 donors, distinct from the solvent system, adds another layer of complexity to the supply chain, introducing additional purification steps to remove unreacted starting materials and byproducts. These factors collectively result in prolonged lead times, elevated production costs, and a larger environmental footprint due to increased solvent consumption and waste generation, making conventional methods less attractive for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
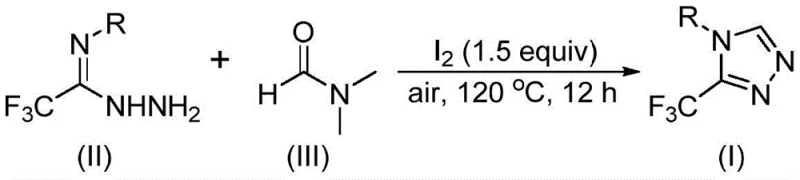
In stark contrast, the methodology outlined in patent CN114920707B presents a paradigm shift by employing a molecular iodine-promoted tandem cyclization that proceeds smoothly in an open-air atmosphere. This novel approach capitalizes on the intrinsic reactivity of DMF, activating its methyl or formyl groups to participate directly in the ring-closing event with trifluoroethyliminohydrazide substrates. The elimination of stringent inert gas requirements allows the reaction to be performed in standard glass-lined reactors, significantly lowering the barrier to entry for manufacturing facilities. Moreover, the use of molecular iodine as a promoter is advantageous due to its low cost, ease of handling, and high efficiency in facilitating the oxidative aromatization necessary to form the stable triazole ring. This streamlined protocol not only simplifies the reaction setup but also enhances the overall reaction efficiency, enabling the synthesis of a wide array of 4-substituted 3-trifluoromethyl-1,2,4-triazoles with excellent functional group tolerance, thereby addressing the critical need for versatile and scalable synthetic solutions in modern drug discovery.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic elegance of this transformation lies in the dual role of the DMF solvent, which can act as a carbon source via two distinct pathways depending on the specific activation mode induced by the iodine promoter. In one pathway, the formyl group of DMF undergoes a condensation reaction with the trifluoroethyliminohydrazide to generate a hydrazone intermediate, which subsequently experiences intramolecular cyclization accompanied by the elimination of dimethylamine to yield the target triazole. Alternatively, when the N-methyl group serves as the carbon donor, DMF first reacts with molecular iodine to form an amine salt species. Following the elimination of hydrogen iodide, this activated species undergoes nucleophilic addition with the hydrazide substrate. The subsequent elimination of N-methylformamide generates a reactive azadiene intermediate, which then undergoes intramolecular nucleophilic attack followed by oxidative aromatization to furnish the final 3-trifluoromethyl-1,2,4-triazole product. This mechanistic flexibility ensures high conversion rates across a broad substrate scope.
From an impurity control perspective, this mechanism offers distinct advantages for process chemists aiming for high-purity pharmaceutical intermediates. The byproducts generated during the cyclization, such as dimethylamine or N-methylformamide, are typically volatile or highly polar, facilitating their removal during the aqueous workup or silica gel purification stages. The use of iodine, while a strong oxidant, is selective enough to promote the desired aromatization without causing excessive degradation of sensitive functional groups on the aromatic ring, such as halogens or ethers. This selectivity is crucial for maintaining the integrity of complex drug-like molecules. Furthermore, the reaction conditions—specifically the temperature range of 110-130°C—are optimized to balance reaction kinetics with thermal stability, minimizing the formation of polymeric tars or decomposition products that often plague high-temperature heterocycle syntheses. Consequently, the resulting crude reaction mixtures are generally cleaner, reducing the burden on downstream purification processes and ensuring consistent quality for regulatory compliance.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and reproducibility. The protocol involves charging a reaction vessel, such as a 35mL Schlenk tube for screening or a larger reactor for scale-up, with the trifluoroethyliminohydrazide substrate and molecular iodine. The molar ratio is critical, with a preferred stoichiometry of 1:1.5 (substrate to iodine) ensuring complete promotion of the cyclization without excessive halogen waste. DMF is added in excess, serving as both the reaction medium and the carbon donor, typically at a concentration that ensures good solubility of the starting materials, approximately 5-8 mL per 1 mmol of substrate. The mixture is then heated to between 110°C and 130°C and stirred for 10 to 15 hours under an air atmosphere. Detailed standardized synthesis steps see the guide below.
- Charge a reaction vessel with molecular iodine (1.5 equivalents), trifluoroethyliminohydrazide substrate, and DMF solvent under an air atmosphere.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for a duration of 10 to 15 hours to ensure complete conversion.
- Upon completion, perform standard post-treatment including filtration, washing, and silica gel column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based synthetic route translates into tangible strategic benefits that extend far beyond the laboratory bench. The primary advantage lies in the drastic simplification of the raw material portfolio. By utilizing DMF, a solvent that is already stocked in virtually every chemical facility in massive quantities, the need to procure specialized, high-cost C1 building blocks is entirely eliminated. This consolidation of materials significantly reduces inventory complexity and mitigates the risk of supply disruptions associated with niche reagents. Furthermore, the operational simplicity of running the reaction under air rather than inert gas removes the dependency on nitrogen or argon infrastructure, lowering utility costs and increasing the throughput capacity of existing reactor farms. These factors combine to create a leaner, more resilient supply chain capable of responding rapidly to fluctuating market demands for key pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, driven primarily by the substitution of expensive reagents with commodity chemicals. Since DMF acts as both solvent and reactant, the effective cost of the carbon source is negligible compared to traditional methods that require dedicated formylation or methylation agents. Additionally, the use of molecular iodine as a promoter is far more cost-effective than employing precious metal catalysts like palladium or rhodium, which not only carry high upfront costs but also require expensive recovery and recycling processes to meet residual metal specifications in APIs. The simplified workup procedure, which avoids complex extractions or distillations in favor of straightforward filtration and chromatography, further reduces labor and energy consumption. Collectively, these efficiencies result in substantial cost savings per kilogram of produced intermediate, enhancing the overall margin profile for the final drug product.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount in the pharmaceutical sector, and this method bolsters reliability by relying on universally available starting materials. Trifluoroethyliminohydrazides can be readily synthesized from commercially available aromatic amines, triphenylphosphine, and hydrazine hydrate, all of which are produced on a multi-ton scale globally. This abundance ensures that raw material shortages are unlikely to bottleneck production. Moreover, the robustness of the reaction conditions—tolerating air and moisture—means that the process is less susceptible to failures caused by minor deviations in environmental controls or equipment integrity. This resilience allows for more predictable production schedules and shorter lead times for high-purity pharmaceutical intermediates, enabling manufacturers to maintain optimal inventory levels and meet just-in-time delivery commitments to downstream API producers without the buffer stocks typically required for more finicky chemistries.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often introduces unforeseen challenges, but the linear scalability of this iodine-promoted cyclization makes it an ideal candidate for commercial expansion. The absence of pyrophoric reagents or high-pressure requirements simplifies the engineering controls needed for large-scale reactors, reducing the capital investment required for plant modifications. From an environmental standpoint, the process aligns well with green chemistry principles by minimizing waste generation; the primary byproducts are small organic molecules that are easier to treat than heavy metal sludge. The high atom economy of incorporating the solvent into the product structure means less waste solvent needs to be disposed of or recycled. This reduced environmental burden facilitates easier regulatory approval and lowers the costs associated with waste management and emissions compliance, making the process sustainable for long-term industrial operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows or for procurement teams assessing the quality and consistency of the supplied intermediates.
Q: What are the key advantages of using DMF as a carbon source in this synthesis?
A: Using DMF eliminates the need for separate, expensive C1 building blocks, as the solvent acts dually as the reaction medium and the carbon donor, significantly simplifying the supply chain and reducing raw material costs.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, the patented method operates efficiently under standard air atmosphere without the need for rigorous anhydrous or oxygen-free environments, which drastically lowers operational complexity and equipment requirements for scale-up.
Q: What is the substrate scope for the R-group in the trifluoroethyliminohydrazide?
A: The method demonstrates broad compatibility with various substituted aryl groups, including those with electron-donating or electron-withdrawing substituents such as halogens, alkyls, alkoxy, and trifluoromethyl groups, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our technical team has thoroughly analyzed the potential of the DMF-based cyclization route described in CN114920707B and is fully prepared to leverage this technology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of 3-trifluoromethyl-1,2,4-triazole intermediates we produce, meeting the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to optimize your supply chain and reduce your overall cost of goods. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to reach out today to discuss your project specifics,索取 specific COA data, and review our comprehensive route feasibility assessments. Let us help you secure a reliable, cost-effective, and high-quality supply of these vital pharmaceutical building blocks for your next generation of therapeutics.
