Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Iodine-Promoted Cyclization for Global Pharmaceutical Supply Chains
The pharmaceutical and agrochemical industries continuously seek robust synthetic routes for fluorinated heterocycles, particularly 1,2,4-triazoles, due to their prevalence in bioactive molecules. As illustrated in the structural diversity of modern therapeutics, the 3-trifluoromethyl-1,2,4-triazole scaffold serves as a critical pharmacophore in potent agents such as Factor IXa inhibitors and GlyT1 inhibitors, where the trifluoromethyl group significantly enhances metabolic stability and lipophilicity. 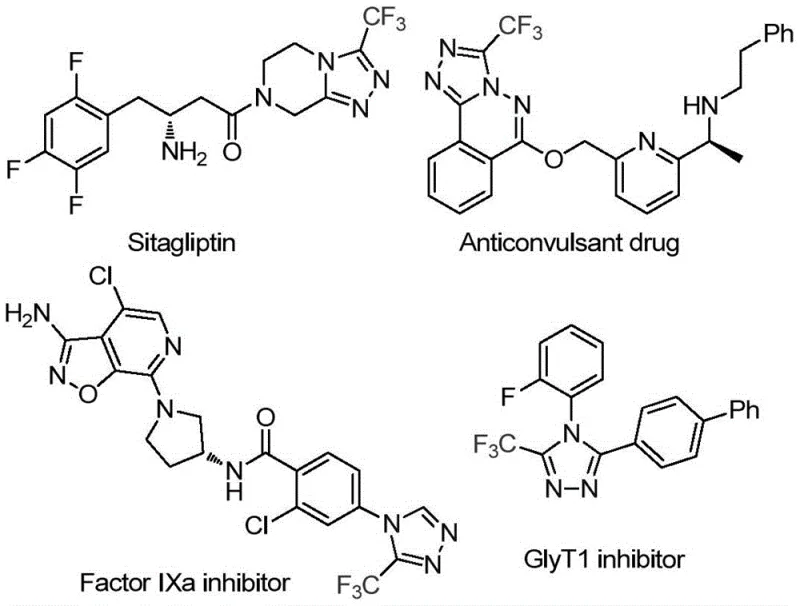 Patent CN114920707B introduces a groundbreaking preparation method that addresses long-standing inefficiencies in constructing these valuable cores. By leveraging dimethylformamide (DMF) not merely as a solvent but as an active carbon synthon, this technology enables the direct assembly of the triazole ring from readily available trifluoroethyliminohydrazide precursors. This approach represents a paradigm shift for reliable pharmaceutical intermediate suppliers aiming to streamline the production of complex nitrogen-containing heterocycles without compromising on purity or yield.
Patent CN114920707B introduces a groundbreaking preparation method that addresses long-standing inefficiencies in constructing these valuable cores. By leveraging dimethylformamide (DMF) not merely as a solvent but as an active carbon synthon, this technology enables the direct assembly of the triazole ring from readily available trifluoroethyliminohydrazide precursors. This approach represents a paradigm shift for reliable pharmaceutical intermediate suppliers aiming to streamline the production of complex nitrogen-containing heterocycles without compromising on purity or yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic strategies for accessing 3-trifluoromethyl-1,2,4-triazoles often suffer from significant operational complexities that hinder large-scale manufacturing. Conventional routes frequently necessitate the use of expensive transition metal catalysts, stringent anhydrous conditions, and inert atmospheres to prevent catalyst deactivation or side reactions. Furthermore, many existing methods rely on specialized one-carbon donors that are costly, hazardous, or difficult to handle on a multi-kilogram scale. The requirement for multiple steps, coupled with the need for rigorous exclusion of moisture and oxygen, drastically increases the capital expenditure for reactor setups and extends the overall production lead time. These factors collectively contribute to higher manufacturing costs and supply chain vulnerabilities, making it challenging for procurement teams to secure cost reduction in API manufacturing while maintaining consistent quality standards.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN114920707B offers a streamlined, atom-economical solution that utilizes molecular iodine as a promoter and DMF as a dual-purpose reagent. This innovative route eliminates the dependency on precious metal catalysts and allows the reaction to proceed efficiently under ambient air conditions. The ability of DMF to serve as both the reaction medium and the carbon source simplifies the reagent profile, reducing the number of raw materials that need to be sourced, qualified, and stored. By operating at moderate temperatures between 110°C and 130°C, the process ensures high reaction efficiency while minimizing energy consumption. This simplification of reaction conditions directly translates to enhanced process safety and operability, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates where operational simplicity is paramount for supply chain reliability.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic elegance of this transformation lies in the activation of the stable DMF molecule by molecular iodine, which facilitates the cleavage of carbon-nitrogen bonds to generate reactive intermediates. 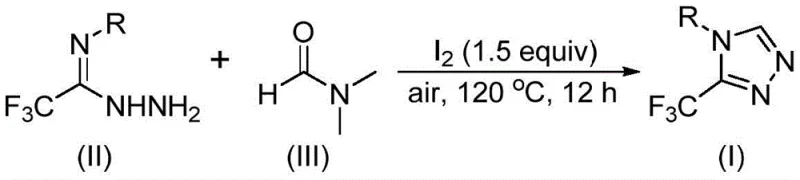 Detailed analysis suggests two potential pathways depending on which fragment of the DMF molecule participates: the formyl group can condense with the hydrazide to form a hydrazone followed by cyclization, or the N-methyl group can undergo iodination and subsequent nucleophilic attack. In both scenarios, the iodine species acts as a Lewis acid or oxidant to drive the tandem cyclization and oxidative aromatization steps necessary to form the stable triazole ring. This dual-pathway capability ensures that the reaction remains robust even with slight variations in substrate electronics, providing a wide functional group tolerance that is crucial for medicinal chemistry applications.
Detailed analysis suggests two potential pathways depending on which fragment of the DMF molecule participates: the formyl group can condense with the hydrazide to form a hydrazone followed by cyclization, or the N-methyl group can undergo iodination and subsequent nucleophilic attack. In both scenarios, the iodine species acts as a Lewis acid or oxidant to drive the tandem cyclization and oxidative aromatization steps necessary to form the stable triazole ring. This dual-pathway capability ensures that the reaction remains robust even with slight variations in substrate electronics, providing a wide functional group tolerance that is crucial for medicinal chemistry applications.
From an impurity control perspective, the use of iodine as a promoter rather than a stoichiometric oxidant helps minimize the formation of over-oxidized byproducts or polymeric tars that often plague radical-mediated cyclizations. The reaction generates dimethylamine or N-methylformamide as benign byproducts, which are easily removed during the aqueous workup or purification stages. This clean reaction profile is essential for R&D directors focused on purity specifications, as it reduces the burden on downstream purification processes like column chromatography or recrystallization. The mechanistic clarity allows process chemists to fine-tune parameters such as iodine loading (optimized at 1.5 equivalents) and reaction time (10-15 hours) to maximize conversion while minimizing the generation of hard-to-remove impurities, ensuring the final high-purity pharmaceutical intermediates meet stringent regulatory requirements.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of execution in standard laboratory and pilot plant settings. The protocol involves simply mixing the trifluoroethyliminohydrazide substrate with molecular iodine in DMF, followed by heating under air. This straightforward procedure removes the need for specialized glovebox techniques or Schlenk lines, significantly lowering the barrier to entry for contract manufacturing organizations. The detailed standardized synthetic steps, including precise stoichiometry and workup procedures, are outlined below to guide process development teams in replicating these results effectively.
- Combine trifluoroethyliminohydrazide, molecular iodine (1.5 equivalents), and DMF solvent in a reaction vessel.
- Heat the mixture to 110-130°C and maintain reaction for 10-15 hours under air atmosphere without inert gas protection.
- Upon completion, perform post-treatment including filtration, washing, and column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based synthesis strategy offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of expensive transition metal catalysts removes a major cost driver and simplifies the supply chain by reducing reliance on volatile precious metal markets. Additionally, the use of DMF, a commodity chemical available in bulk quantities globally, ensures raw material security and price stability. The operational robustness of the method, which tolerates air and moisture, reduces the risk of batch failures due to environmental excursions, thereby enhancing overall supply chain reliability and consistency of delivery.
- Cost Reduction in Manufacturing: The dual role of DMF as both solvent and reactant significantly reduces the material bill of materials (BOM) by eliminating the need for separate, often expensive, one-carbon building blocks. Furthermore, the absence of precious metal catalysts negates the costs associated with metal scavenging and residual metal testing, which are critical quality attributes for pharmaceutical ingredients. This streamlined reagent profile leads to substantial cost savings in raw material procurement and waste disposal, directly impacting the bottom line for large-scale production campaigns.
- Enhanced Supply Chain Reliability: By removing the requirement for inert atmosphere conditions, the process can be executed in standard glass-lined or stainless steel reactors without the need for complex nitrogen blanketing systems. This flexibility allows for greater manufacturing agility and reduces the downtime associated with reactor preparation and purging. The use of commercially available starting materials with broad market availability ensures that production schedules are not disrupted by raw material shortages, providing a stable and predictable supply of critical intermediates to downstream customers.
- Scalability and Environmental Compliance: The reaction generates minimal hazardous waste compared to traditional methods that might utilize toxic solvents or generate heavy metal contamination. The benign nature of the byproducts simplifies effluent treatment and aligns with increasingly stringent environmental regulations. The simplicity of the workup, involving basic filtration and chromatography, facilitates easy scale-up from gram to ton scales, ensuring that the process remains efficient and manageable even as production volumes increase to meet global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical teams evaluating this route for potential integration into their manufacturing portfolios.
Q: Why is DMF critical in this synthesis beyond being a solvent?
A: In this novel pathway described in patent CN114920707B, DMF acts dually as the reaction medium and the essential carbon source, providing either the formyl or methyl group required for ring construction, which eliminates the need for separate expensive carbon donors.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, a significant advantage of this iodine-promoted method is its operational simplicity; it proceeds efficiently under standard air atmosphere and does not demand rigorous anhydrous or oxygen-free environments, greatly simplifying equipment requirements.
Q: What is the substrate scope for the R group in the triazole product?
A: The method demonstrates broad applicability, successfully accommodating various substituted aryl groups including those with electron-donating or withdrawing substituents such as halogens, alkyls, alkoxy, and trifluoromethyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are successfully translated into viable industrial processes. We are committed to delivering high-purity intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in handling fluorinated compounds and iodine-mediated reactions positions us as a preferred partner for complex organic synthesis projects.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthetic technology for your pipeline projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your drug development timelines while optimizing your overall production costs.
