Scalable Production of 2-Trifluoromethyl Quinazolinones Using Earth-Abundant Iron Catalysts
Scalable Production of 2-Trifluoromethyl Quinazolinones Using Earth-Abundant Iron Catalysts
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to access complex heterocyclic scaffolds, particularly those containing fluorine motifs which are critical for enhancing metabolic stability and bioavailability in drug candidates. A recent technological breakthrough detailed in patent CN111675662B introduces a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds, a structural core found in numerous bioactive molecules ranging from anticancer agents to antifungal drugs. This innovation leverages an iron-catalyzed cyclization strategy that circumvents the limitations of traditional synthetic routes, offering a robust platform for the manufacturing of high-value pharmaceutical intermediates. By utilizing readily available starting materials such as isatin and trifluoroethylimidoyl chloride, this method addresses key pain points in process chemistry, including cost, safety, and scalability, making it an attractive option for commercial production.
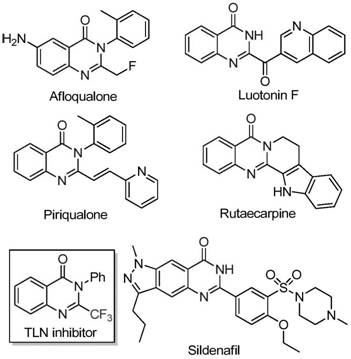
Quinazolinone compounds represent a privileged class of nitrogen-containing fused ring six-membered heterocycles that are ubiquitous in medicinal chemistry due to their diverse biological activities. As illustrated in the structural diversity of known drugs, these scaffolds exhibit potent anti-cancer, anticonvulsant, anti-inflammatory, and antimalarial properties. The strategic introduction of a trifluoromethyl group into these heterocyclic systems further amplifies their pharmacological potential by significantly improving electronegativity, lipophilicity, and metabolic resistance. However, accessing these specific trifluoromethyl-substituted architectures has historically been challenging, often requiring multi-step sequences or expensive reagents. The new methodology described in the patent provides a direct and efficient entry point to these valuable chemical spaces, enabling the rapid generation of libraries for drug discovery and the streamlined production of active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functional groups has relied heavily on the cyclization of specific synthons with substrates like anthranilamide, anthranilic acid, or isatoic anhydride. While effective in academic settings, these conventional approaches suffer from several critical drawbacks that hinder their industrial applicability. Commonly used trifluoromethylating agents such as trifluoroacetic anhydride and ethyl trifluoroacetate are often expensive and can generate significant amounts of hazardous waste. Furthermore, these reactions frequently necessitate severe reaction conditions, including high temperatures or strong acidic/basic environments, which can lead to poor functional group tolerance and low overall yields. The narrow substrate scope of these traditional methods limits the ability of chemists to explore diverse chemical space, forcing reliance on costly protecting group strategies or specialized starting materials that are not commercially viable for large-scale manufacturing.
The Novel Approach
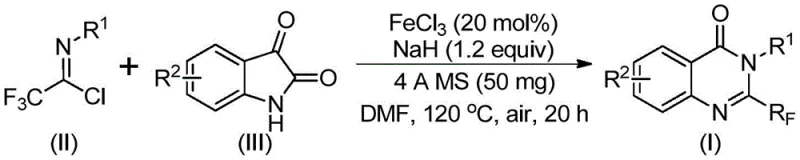
In stark contrast to these legacy methods, the invention disclosed in CN111675662B utilizes a remarkably simple yet powerful strategy involving the reaction of trifluoroethylimidoyl chloride with isatin derivatives. This novel approach employs ferric chloride (FeCl3) as a catalyst, an earth-abundant and inexpensive metal salt, in conjunction with sodium hydride as a base. The reaction proceeds through a cascade sequence that efficiently constructs the quinazolinone core while installing the trifluoromethyl group in a single operational step. This method boasts excellent functional group tolerance, allowing for the incorporation of various substituents such as alkyl, halogen, and methoxy groups on the aromatic rings without compromising yield. The simplicity of the operation, combined with the use of cheap and easily obtained initial raw materials, represents a paradigm shift in how these complex heterocycles can be manufactured, offering a clear path toward cost reduction and process intensification.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The success of this transformation lies in the unique mechanistic pathway facilitated by the iron catalyst and the basic conditions. The reaction initiates with an alkali-promoted formation of carbon-nitrogen bonds between the trifluoroethylimidoyl chloride and the isatin substrate. This initial coupling generates a trifluoroacetamidine intermediate, which serves as the precursor for the subsequent cyclization event. The presence of ferric chloride is crucial at this stage, as it catalyzes a decarbonylation and cyclization sequence that drives the isomerization of the intermediate into the final stable 2-trifluoromethyl-substituted quinazolinone structure. This iron-mediated process avoids the need for stoichiometric amounts of toxic reagents or harsh oxidants, relying instead on the redox properties of the iron center to facilitate bond rearrangement. The mechanism ensures high atom economy and minimizes the formation of side products, which is essential for maintaining high purity standards required in pharmaceutical manufacturing.
Furthermore, the reaction conditions are optimized to maximize conversion while minimizing degradation. The use of 4A molecular sieves plays a pivotal role in scavenging trace moisture, which is critical given the sensitivity of sodium hydride and the imidoyl chloride to hydrolysis. The reaction typically proceeds in two stages: an initial period at 40°C to allow for the formation of the intermediate, followed by heating to 120°C to drive the cyclization to completion. This controlled thermal profile prevents the decomposition of sensitive functional groups and ensures that the reaction reaches full conversion within a reasonable timeframe of 24 to 48 hours. The robustness of this mechanistic pathway allows for a wide range of substrates to be processed with consistent results, demonstrating the versatility of the iron catalytic system in handling diverse electronic and steric environments.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot plant equipment. The process involves mixing the key reagents—isatin, trifluoroethylimidoyl chloride, ferric chloride, and sodium hydride—in a polar aprotic solvent such as DMF. The addition of molecular sieves ensures anhydrous conditions, which is vital for the stability of the reactive intermediates. Following the reaction, the workup procedure is equally simple, involving filtration to remove solids and standard column chromatography for purification. This ease of execution makes the method highly accessible for process chemists looking to optimize production workflows. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then increase temperature to 120°C and continue reacting for 18-20 hours.
- Upon completion, filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits that directly address the priorities of procurement managers and supply chain directors. The shift from expensive precious metal catalysts or specialized fluorinating agents to commodity chemicals like ferric chloride and isatin drastically reduces the raw material cost base. This cost efficiency is compounded by the high yields reported across a broad substrate scope, which minimizes waste and maximizes the output per batch. For organizations focused on cost reduction in pharmaceutical intermediate manufacturing, this route presents a compelling alternative to legacy processes that are often burdened by low efficiency and high reagent costs. The ability to source starting materials from established global supply chains further enhances the reliability of production schedules.
- Cost Reduction in Manufacturing: The replacement of costly catalysts and reagents with inexpensive iron salts and readily available organic precursors leads to a significant decrease in the overall cost of goods sold. By eliminating the need for expensive transition metals and complex purification steps associated with their removal, the process streamlines the manufacturing workflow. This economic advantage is further amplified by the high conversion rates observed, which reduce the volume of raw materials required to produce a fixed amount of product, thereby optimizing resource utilization and lowering the environmental footprint of the synthesis.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as isatin and ferric chloride ensures a stable and resilient supply chain. Unlike specialized reagents that may be subject to shortages or long lead times, these materials are produced in large volumes by multiple suppliers globally. This abundance mitigates the risk of production delays caused by raw material scarcity. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, providing greater flexibility in sourcing and reducing the need for stringent incoming quality control measures that can slow down operations.
- Scalability and Environmental Compliance: The simplicity of the reaction setup and the use of common solvents like DMF facilitate easy scale-up from gram to kilogram and potentially ton scales. The absence of highly toxic or hazardous reagents simplifies waste management and aligns with increasingly strict environmental regulations. The process generates fewer byproducts and avoids the use of heavy metals that require complex disposal protocols, making it a more sustainable choice for modern chemical manufacturing. This scalability ensures that the method can meet growing market demands without the need for extensive process re-engineering or capital investment in specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 2-trifluoromethyl quinazolinones. These answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing reliable guidance for R&D and procurement teams evaluating this technology. Understanding these details is crucial for assessing the feasibility of integrating this method into existing production pipelines.
Q: What are the primary advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers a significant cost advantage as it is an earth-abundant, inexpensive metal compared to precious metal catalysts. Additionally, it demonstrates high catalytic efficiency and excellent functional group tolerance, allowing for a broader substrate scope without requiring harsh reaction conditions often associated with traditional methods.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent explicitly states that the method is simple to operate, uses readily available raw materials, and has been demonstrated to expand to the gram level, providing a strong possibility for industrial scale application due to the robustness of the reaction conditions.
Q: What is the role of 4A molecular sieves in this reaction?
A: 4A molecular sieves are added to the reaction mixture to act as a desiccant, effectively removing trace water that could interfere with the sodium hydride base or the sensitive imidoyl chloride intermediate, thereby ensuring higher reaction yields and purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed synthesis route for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical building blocks. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest industry standards. We are committed to leveraging innovative technologies like the one described in CN111675662B to drive efficiency and value for our global partners.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your supply chain and reduce costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the marketplace through superior chemical manufacturing solutions.
