Scalable Metal-Free Synthesis of Pyrimidoindazole Compounds for Pharma Applications
Introduction to Advanced Pyrimidoindazole Synthesis
The pharmaceutical industry continuously seeks robust and efficient pathways for constructing complex heterocyclic scaffolds, particularly those exhibiting potent biological activity. A significant breakthrough in this domain is detailed in patent CN109912606B, which discloses a novel synthetic method for pyrimidoindazole compounds. These derivatives are increasingly recognized as potential protein kinase inhibitors with therapeutic applications ranging from senile dementia and viral infections to autoimmune diseases and antitumor treatments. The core innovation lies in a transition-metal-free, one-pot cascade reaction that directly couples aromatic aldehydes with 3-aminoindazoles. Unlike traditional multi-step syntheses that often suffer from resource waste and environmental hazards, this methodology employs inexpensive reagents such as triethylamine and ammonium iodide under mild oxidative conditions. This approach not only streamlines the production workflow but also aligns with modern green chemistry principles, offering a compelling solution for the commercial scale-up of complex pharmaceutical intermediates.
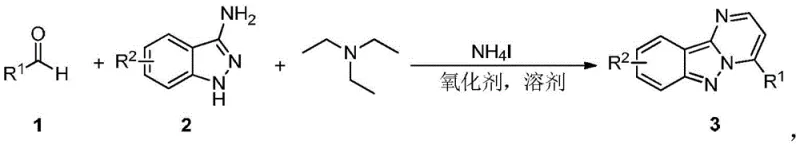
The strategic value of this technology extends beyond mere academic interest; it addresses critical pain points in supply chain stability and cost management for active pharmaceutical ingredient (API) manufacturing. By eliminating the need for precious metal catalysts like palladium or copper, which are prone to price volatility and strict regulatory limits on residual metals in final drug products, this process offers a cleaner and more economically viable alternative. The reaction operates effectively at temperatures between 110-150°C using common solvents such as chlorobenzene, toluene, or even greener options like air as the terminal oxidant. For R&D directors and procurement managers alike, adopting such a streamlined protocol means reducing the complexity of purification steps and minimizing the environmental footprint associated with waste disposal, thereby enhancing the overall sustainability profile of the manufacturing process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrimidoindazole core has relied on cumbersome methodologies that pose significant challenges for large-scale production. Traditional routes often involve the condensation of 3-aminoindazole with 1,3-dicarbonyl compounds or propiolates, or alternatively, Grieco condensation reactions requiring alkenes or alkynes. These legacy methods are frequently plagued by the necessity of using corrosive and toxic reagents, which demand specialized handling equipment and rigorous safety protocols. Furthermore, the reliance on expensive transition metal catalysts introduces a substantial cost burden and creates a bottleneck in the purification stage, where removing trace metal residues to meet pharmacopeial standards can be technically demanding and yield-reducing. The multi-step nature of these conventional syntheses inevitably leads to lower overall yields due to cumulative losses at each isolation stage, alongside the generation of significant chemical waste that complicates environmental compliance and increases operational expenditures.
The Novel Approach
In stark contrast, the methodology described in CN109912606B revolutionizes the synthesis landscape by introducing a direct, one-pot series reaction that bypasses the need for intermediate isolation. This novel approach utilizes a simple combination of aromatic aldehydes, 3-aminoindazoles, and triethylamine in the presence of ammonium iodide and an oxidant. The reaction conditions are remarkably mild, typically proceeding at 110-150°C, and the system is versatile enough to accommodate various oxidants including di-tert-butyl peroxide, DMSO, or even molecular oxygen from the air. This simplicity translates directly into operational efficiency, as the entire transformation occurs in a single vessel, drastically reducing labor time and solvent consumption. For a reliable pharmaceutical intermediate supplier, this means the ability to offer high-purity products with a significantly reduced lead time, as the complex work-up procedures associated with multi-step syntheses are rendered obsolete by this elegant cascade design.
Mechanistic Insights into Metal-Free Oxidative Cyclization
The mechanistic elegance of this synthesis lies in its ability to forge multiple bonds without the aid of transition metals. The reaction initiates with the activation of the aromatic aldehyde and the 3-aminoindazole by the ammonium iodide and the base, triethylamine. Under oxidative conditions, likely involving radical intermediates generated by the peroxide or oxygen, the system facilitates a cascade cyclization. The iodide species plays a crucial catalytic role, potentially mediating the oxidation steps required to aromatize the final pyrimidoindazole ring system. This metal-free pathway ensures that the final product is free from heavy metal contamination, a critical quality attribute for any candidate intended for clinical development. The robustness of this mechanism is evidenced by its tolerance to a wide array of functional groups, allowing for the late-stage diversification of the scaffold without compromising the integrity of sensitive moieties.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed counterparts. In traditional cross-coupling reactions, side products often arise from homocoupling of the starting materials or incomplete conversion due to catalyst deactivation. Here, the use of stoichiometric or excess oxidants drives the reaction to completion, while the absence of metal ligands eliminates a whole class of potential organometallic impurities. The purification process is consequently simplified to standard aqueous work-ups and silica gel chromatography, yielding products with high purity profiles as confirmed by NMR and HRMS data in the patent examples. This level of control is essential for maintaining stringent purity specifications required by global regulatory bodies, ensuring that the supply of these intermediates remains consistent and reliable for downstream drug synthesis.
How to Synthesize Pyrimidoindazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the molar ratios and reaction parameters outlined in the patent data. The standard protocol involves dissolving the aromatic aldehyde and 3-aminoindazole in a solvent like chlorobenzene, followed by the addition of triethylamine and ammonium iodide. The mixture is then heated, typically in an oil bath, with the option to run the reaction under an open atmosphere if air is used as the oxidant, or in a sealed tube for peroxide-based systems. The simplicity of the setup allows for easy monitoring and control, making it accessible for process chemists looking to optimize cost reduction in API manufacturing. While the specific stoichiometry may vary slightly depending on the electronic nature of the substrates, the general procedure remains consistent across a broad range of derivatives.
- Dissolve aromatic aldehyde, 3-aminoindazole, and triethylamine in a suitable solvent such as chlorobenzene or toluene.
- Add ammonium iodide (NH4I) and an oxidant (e.g., DTBP, DMSO, or air) to the reaction mixture.
- Heat the mixture to 110-150°C for approximately 12 hours, then quench and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend far beyond the laboratory bench. The primary advantage is the drastic simplification of the raw material portfolio. By utilizing commodity chemicals like benzaldehydes and simple amines, the supply chain becomes less vulnerable to the geopolitical and logistical disruptions often associated with specialized catalysts or exotic reagents. Furthermore, the elimination of transition metals removes the need for expensive scavenging resins or complex filtration steps, directly lowering the cost of goods sold (COGS). The ability to use air as an oxidant in certain variations of this process further underscores its economic and environmental efficiency, reducing the dependency on hazardous chemical oxidants and simplifying waste treatment protocols.
- Cost Reduction in Manufacturing: The transition-metal-free nature of this process eliminates the substantial cost burden associated with purchasing and recovering precious metal catalysts such as palladium or rhodium. Additionally, the one-pot design minimizes solvent usage and labor hours by removing intermediate isolation steps, leading to substantial cost savings in utility and waste disposal. The use of inexpensive reagents like triethylamine and ammonium iodide further drives down the raw material costs, making the production of high-value pyrimidoindazoles economically feasible on a multi-ton scale.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, primarily substituted benzaldehydes and aminoindazoles, are widely available from multiple global suppliers, ensuring a robust and resilient supply chain. This diversity in sourcing mitigates the risk of single-supplier dependency and allows for flexible procurement strategies. Moreover, the mild reaction conditions reduce the wear and tear on reactor equipment, extending asset life and minimizing unplanned downtime, which is critical for maintaining consistent delivery schedules to pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently in standard reaction vessels without the need for specialized high-pressure or cryogenic equipment. The reduced generation of hazardous waste, particularly heavy metal sludge, simplifies compliance with increasingly stringent environmental regulations. This green chemistry profile not only lowers the cost of environmental management but also enhances the corporate sustainability image, a factor of growing importance to stakeholders and end-users in the healthcare sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrimidoindazole synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is vital for R&D teams evaluating route feasibility and for procurement professionals assessing the long-term viability of this supply source.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the patented method (CN109912606B) specifically utilizes a transition-metal-free approach, relying on ammonium iodide and organic oxidants, which significantly reduces raw material costs and eliminates heavy metal residue concerns.
Q: What is the substrate scope for this reaction?
A: The method demonstrates excellent tolerance for various substituents, including phenyl, naphthyl, pyridyl, and thienyl groups, as well as electron-donating and electron-withdrawing groups like halogens, nitro, and trifluoromethyl.
Q: Can this process be scaled for industrial production?
A: Yes, the process is designed for scalability with simple operation, mild reaction conditions (110-150°C), and the ability to use air as a green oxidant, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidoindazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free synthesis described in CN109912606B for the production of high-purity pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of pyrimidoindazole compound meets the exacting standards required for clinical and commercial applications. We are equipped to handle the specific nuances of this oxidative cyclization, optimizing parameters to maximize yield and minimize impurities.
We invite you to collaborate with us to leverage this advanced synthetic technology for your drug development pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data for our existing inventory or to discuss route feasibility assessments for novel derivatives. By partnering with NINGBO INNO PHARMCHEM, you secure a supply chain that is not only cost-effective and reliable but also aligned with the future of sustainable chemical manufacturing.
