Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Intermediates for Global Pharmaceutical Supply Chains
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Intermediates for Global Pharmaceutical Supply Chains
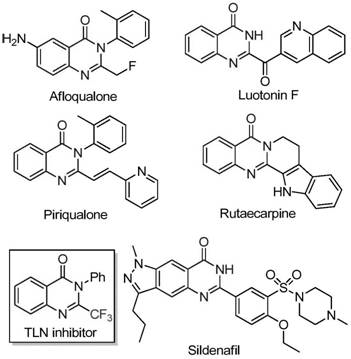
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. Quinazolinones are a privileged scaffold in medicinal chemistry, found in numerous bioactive molecules exhibiting anti-cancer, anticonvulsant, and anti-inflammatory properties. The strategic introduction of a trifluoromethyl group into this heterocyclic core further enhances the pharmacokinetic profile of potential drug candidates by improving metabolic stability and lipophilicity. This patent presents a robust methodology that addresses long-standing challenges in synthesizing these valuable structures, offering a pathway that is not only chemically elegant but also commercially viable for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functionalities has relied heavily on cyclization reactions involving synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with substrates like anthranilamide or isatoic anhydride. While effective in academic settings, these traditional methodologies suffer from significant drawbacks when evaluated through the lens of industrial manufacturing. The reaction conditions are often severe, requiring harsh reagents that pose safety risks and complicate waste management. Furthermore, the starting materials, particularly specialized trifluoromethyl synthons, can be prohibitively expensive and difficult to source in bulk quantities. These factors collectively result in low overall yields and narrow substrate scopes, limiting the ability of process chemists to rapidly generate diverse libraries of analogs for structure-activity relationship (SAR) studies or to scale up production for clinical trials without incurring excessive costs.
The Novel Approach
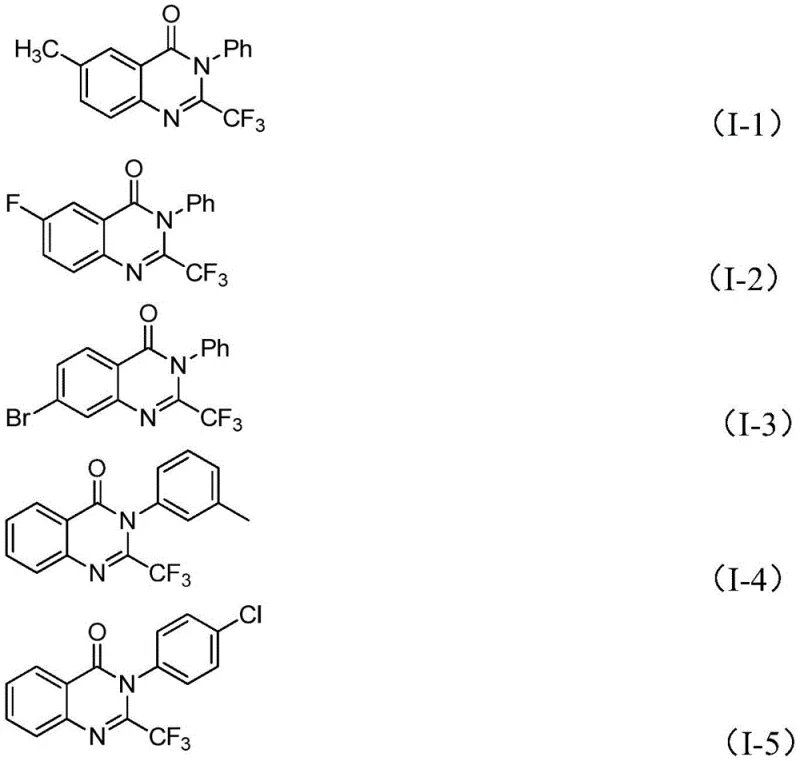
In stark contrast to these legacy methods, the technology described in patent CN111675662B introduces a paradigm shift by utilizing readily available trifluoroethylimidoyl chloride and isatin as the primary building blocks. This innovative route leverages a series of cyclization reactions catalyzed by cheap metal iron, specifically ferric chloride, which dramatically lowers the barrier to entry for production. The process demonstrates exceptional functional group tolerance, allowing for the synthesis of a wide array of derivatives with substituents at various positions on the aromatic rings. By shifting away from expensive precious metal catalysts or hazardous anhydrides, this method not only simplifies the operational procedure but also significantly broadens the applicability of the synthesis. The ability to access complex 2-trifluoromethyl substituted quinazolinones through such a direct and efficient pathway represents a major advancement for reliable pharmaceutical intermediate supplier networks seeking to optimize their portfolios.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the unique catalytic cycle facilitated by ferric chloride (FeCl3) in conjunction with sodium hydride (NaH). The reaction initiates with the formation of carbon-nitrogen bonds between the trifluoroethylimidoyl chloride and the isatin substrate, promoted by the base. This initial step generates a trifluoroacetamidine intermediate, which serves as the precursor for the subsequent ring closure. The presence of the iron catalyst is crucial for the next phase, where it mediates a decarbonylation and cyclization sequence. This iron-catalyzed isomerization effectively rearranges the molecular skeleton to form the stable quinazolinone core while retaining the critical trifluoromethyl group at the 2-position. The mechanistic elegance of this transformation ensures high atom economy and minimizes the formation of unwanted byproducts, which is a key consideration for maintaining high purity standards in API manufacturing.
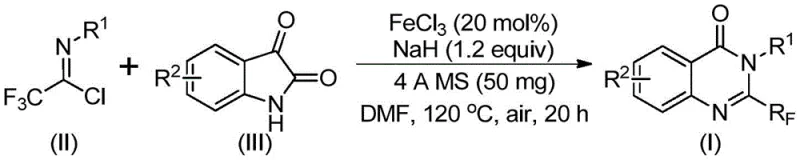
From an impurity control perspective, the use of 4A molecular sieves in the reaction mixture plays a pivotal role in driving the equilibrium towards the desired product by sequestering moisture, which could otherwise hydrolyze the sensitive imidoyl chloride starting material. The reaction conditions, typically involving a two-stage temperature profile starting at 40°C and ramping to 120°C in a polar aprotic solvent like DMF, are optimized to balance reaction kinetics with thermal stability. This controlled environment prevents the degradation of the trifluoromethyl moiety and ensures that the final crude product contains a minimal impurity profile. For R&D directors, understanding this mechanism highlights the robustness of the process; the tolerance for various substituents (R1 and R2 groups) means that a single platform technology can be adapted to produce a vast library of high-purity pharmaceutical intermediates without needing to re-optimize the entire synthetic route for each new analog.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves a straightforward mixing of reagents followed by a controlled heating sequence, eliminating the need for complex addition protocols or inert atmosphere techniques beyond standard Schlenk line practices. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and temporal parameters required to achieve optimal conversion. This simplicity is a direct result of the robust nature of the iron catalyst system, which remains active and selective even under aerobic conditions, further simplifying the engineering requirements for the reaction vessel.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C for 18-20 hours under air.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iron-catalyzed methodology offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of the raw material supply chain. By utilizing isatin and trifluoroethylimidoyl chloride, which are commodity chemicals or easily synthesized from abundant aromatic amines, manufacturers can mitigate the risks associated with sourcing exotic or monopolized reagents. This shift ensures a more resilient supply chain, reducing the likelihood of production delays caused by raw material shortages. Furthermore, the elimination of expensive transition metal catalysts, such as palladium or rhodium, which are often subject to volatile market pricing and geopolitical supply constraints, leads to substantial cost savings in the overall bill of materials.
- Cost Reduction in Manufacturing: The economic impact of replacing precious metal catalysts with ferric chloride cannot be overstated. Iron is one of the most abundant and inexpensive metals on earth, and its use here eliminates the need for costly catalyst recovery systems or extensive heavy metal scavenging steps during downstream processing. This reduction in catalyst cost, combined with the high yields reported in the patent data, translates directly to a lower cost of goods sold (COGS). Additionally, the simplified post-treatment procedure, which relies on standard filtration and column chromatography rather than complex distillation or crystallization sequences, reduces energy consumption and labor hours, further driving down the operational expenditure associated with cost reduction in API manufacturing.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials like isatin and simple aromatic amines ensures that the supply chain remains robust against market fluctuations. Unlike specialized fluorinating agents that may have limited suppliers, the precursors for this reaction are produced globally at massive scales for various industries. This abundance guarantees consistent availability and competitive pricing, allowing procurement teams to negotiate better terms and secure long-term contracts. The stability of the supply chain is further reinforced by the reaction's tolerance to air and moisture, which reduces the logistical burden of transporting and storing sensitive reagents under strictly anhydrous conditions, thereby minimizing the risk of spoilage and waste.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns perfectly with the principles of green chemistry. The use of non-toxic iron catalysts reduces the heavy metal load in wastewater, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. The high atom economy of the cyclization reaction means less waste generation per kilogram of product, contributing to a smaller environmental footprint. Moreover, the demonstrated scalability of the reaction, capable of moving from milligram to gram scales without loss of efficiency, indicates a clear path to commercial scale-up of complex pharmaceutical intermediates. This scalability ensures that as demand for the final drug product grows, the supply of the intermediate can be ramped up seamlessly without requiring entirely new process development campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for your specific production needs. Understanding these details is crucial for making informed decisions about process integration and supplier qualification.
Q: What are the key advantages of this iron-catalyzed method over traditional synthesis?
A: This method utilizes inexpensive iron catalysts and readily available isatin substrates, avoiding the severe conditions and expensive reagents like trifluoroacetic anhydride often required in conventional routes.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and provides possibilities for industrial scale application due to its simple operation and high functional group tolerance.
Q: What is the role of the trifluoromethyl group in these compounds?
A: The introduction of the trifluoromethyl group significantly improves electronegativity, bioavailability, metabolic stability, and lipophilicity of the target molecule, enhancing its potential as a pharmaceutical agent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner who can translate cutting-edge patent technologies into commercial reality. Our team of expert process chemists has extensively evaluated the iron-catalyzed route described in CN111675662B and confirmed its potential for delivering high-quality intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move smoothly from clinical phases to full-scale market launch. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, meeting the exacting standards required by global regulatory bodies for pharmaceutical ingredients.
We invite you to collaborate with us to leverage this efficient synthesis for your drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss your project needs,索取 specific COA data for our existing inventory, and review our comprehensive route feasibility assessments. Let us help you secure a competitive advantage through superior chemistry and reliable supply chain execution.
