Scalable Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazole Derivatives for Pharmaceutical Applications
Introduction to Next-Generation Imidazole Synthesis
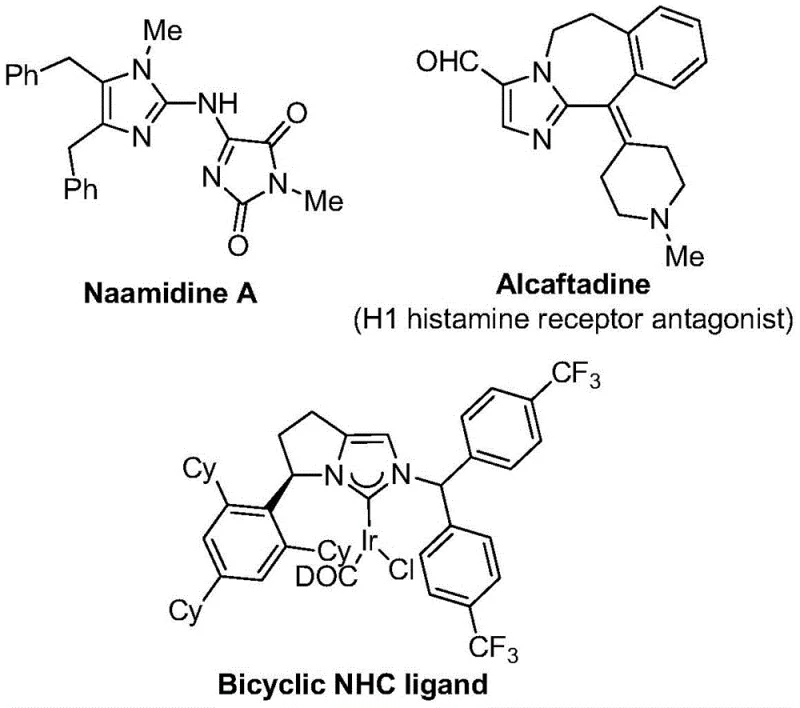
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds represents a cornerstone of modern medicinal chemistry, significantly enhancing the lipophilicity, metabolic stability, and bioavailability of drug candidates. As detailed in the groundbreaking patent CN111423381B, a novel preparation method for 2-trifluoromethyl substituted imidazole compounds has been developed that addresses critical limitations in existing synthetic methodologies. Imidazole derivatives are ubiquitous in biologically active molecules, serving as key structural motifs in various therapeutic agents and functional materials, as illustrated by the diverse molecular architectures found in current pharmacopeia.
This innovative approach leverages a transition metal palladium-catalyzed carbonylation series reaction, utilizing cheap and easily obtained starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts. The significance of this technology extends beyond academic interest; it provides a robust platform for the commercial scale-up of complex pharmaceutical intermediates. By operating under mild conditions at 30°C, the process minimizes energy consumption and safety risks associated with high-temperature reactions, positioning it as a highly attractive route for reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has relied heavily on the direct reaction of synthons with suitable substrates, often employing reagents like trifluorodiazoethane. While effective in specific contexts, these conventional methods suffer from significant drawbacks that hinder their widespread industrial adoption. Trifluorodiazoethane, for instance, is known to be potentially explosive and requires stringent safety protocols, increasing the operational complexity and cost of manufacturing. Furthermore, many traditional routes exhibit poor substrate compatibility, failing to tolerate sensitive functional groups that are frequently required in advanced drug discovery programs.
Additionally, the reliance on harsh reaction conditions or expensive, specialized catalysts in older methodologies often results in lower overall yields and difficult purification processes. The generation of hazardous byproducts and the need for specialized equipment to handle gaseous reagents further exacerbate the environmental and economic burden. For procurement managers and supply chain heads, these factors translate into higher costs, longer lead times, and increased supply chain vulnerability, making the search for alternative, safer synthetic routes a critical priority for the industry.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN111423381B introduces a streamlined, multicomponent reaction that utilizes stable and commercially available precursors. The core of this innovation lies in the use of trifluoroethylimidoyl chloride and diaryl iodonium salts, which are not only safer to handle but also offer superior designability for substrate modification. The reaction proceeds efficiently in common organic solvents like tetrahydrofuran, utilizing a palladium catalyst system that promotes the formation of the imidazole ring through a sophisticated carbonylation mechanism.

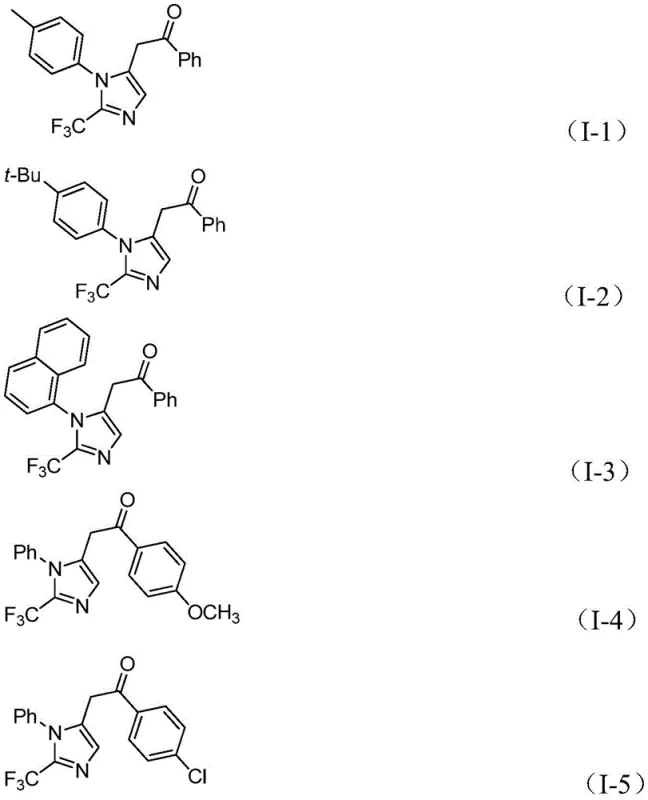
This novel approach achieves high reaction efficiency with excellent substrate compatibility, allowing for the introduction of diverse substituents such as methyl, tert-butyl, chlorine, bromine, and nitro groups without significant loss in yield. The ability to synthesize variously substituted imidazole compounds through simple substrate design broadens the utility of this method, enabling rapid access to diverse chemical libraries for drug screening. Moreover, the mild reaction temperature of 30°C and the use of formic acid/acetic anhydride as a carbon monoxide surrogate eliminate the need for high-pressure CO gas, drastically simplifying the reactor requirements and enhancing process safety.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, involving a cascade of well-defined steps that ensure high selectivity and yield. The reaction initiates with the formation of a trifluoroacetamidine compound via an intermolecular carbon-nitrogen bond promoted by the alkaline additive, followed by isomerization. Subsequently, the palladium catalyst activates the alkyne moiety of the propargylamine through palladation, generating a crucial alkenyl palladium intermediate. This intermediate undergoes further isomerization to form an alkyl palladium species, setting the stage for the pivotal carbonylation step.
Under the action of carbon monoxide released in situ from the formic acid and acetic anhydride mixture, the alkyl palladium intermediate converts into an acyl palladium intermediate. The cycle continues with the oxidative addition of the diaryl iodonium salt, generating a high-valent tetravalent palladium intermediate. Finally, reductive elimination occurs to release the final 2-trifluoromethyl-substituted imidazole product and regenerate the active palladium catalyst. This intricate cycle ensures that the reaction proceeds with high atom economy and minimal waste generation.
From an impurity control perspective, the mild conditions and specific catalytic cycle play a vital role in maintaining product purity. The low temperature prevents thermal degradation of sensitive functional groups, while the specific reactivity of the diaryl iodonium salt minimizes side reactions commonly associated with aryl halides. The use of sodium bicarbonate as a base effectively neutralizes acidic byproducts without promoting unwanted hydrolysis of the imidoyl chloride. This precise control over the reaction environment results in a clean crude product profile, significantly reducing the burden on downstream purification processes and ensuring that the final high-purity pharmaceutical intermediates meet stringent quality specifications required for GMP manufacturing.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot plant production. The protocol involves a straightforward one-pot procedure where all reagents, including the palladium catalyst, ligand, and additives, are combined in an organic solvent. The reaction is allowed to proceed for 16 to 24 hours at a constant temperature of 30°C, after which standard workup procedures such as filtration and silica gel mixing are employed. Detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid in an organic solvent such as tetrahydrofuran.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter and purify via column chromatography to isolate the target imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling strategic advantages that directly impact the bottom line and operational resilience. The shift from hazardous, specialized reagents to commodity chemicals like propargylamine and aromatic amines fundamentally alters the cost structure of production. By relying on raw materials that are widely available in the global chemical market, manufacturers can mitigate the risks associated with supply chain disruptions and price volatility often seen with exotic fluorinating agents.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide equipment and the use of mild reaction conditions significantly lower capital expenditure and energy costs. Furthermore, the high reaction efficiency and yield reduce the amount of raw material required per unit of product, leading to substantial cost savings in material procurement. The simplified post-treatment process, which avoids complex extraction or distillation steps, further reduces labor and utility costs, making the overall manufacturing process economically superior to traditional methods.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable starting materials such as trifluoroethylimidoyl chloride and diaryl iodonium salts ensures a consistent and reliable supply of inputs. Unlike gas-based reagents that require specialized logistics and storage infrastructure, these solid or liquid precursors can be sourced from multiple vendors globally. This diversification of the supply base enhances supply chain security, reducing lead time for high-purity pharmaceutical intermediates and ensuring continuous production schedules even during market fluctuations.
- Scalability and Environmental Compliance: The process has been demonstrated to be expandable to gram-level synthesis with consistent results, indicating a clear path toward kilogram and ton-scale production. The use of tetrahydrofuran as a solvent and the generation of minimal hazardous waste align with green chemistry principles, facilitating easier regulatory compliance. The absence of heavy metal contamination risks, thanks to the efficient catalytic cycle and purification methods, ensures that the final product meets the rigorous impurity profiles demanded by regulatory agencies for clinical and commercial use.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential partners and licensees.
Q: What are the primary advantages of using formic acid and acetic anhydride in this synthesis?
A: This combination serves as a safe and efficient carbon monoxide surrogate, eliminating the need for handling hazardous high-pressure CO gas while facilitating the carbonylation step essential for forming the imidazole core.
Q: How does this method improve substrate compatibility compared to traditional trifluoromethylation?
A: By utilizing stable trifluoroethylimidoyl chlorides and diaryl iodonium salts instead of explosive diazo compounds, the process tolerates a wide range of functional groups including halogens, nitro, and alkyl substituents without compromising yield.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the protocol operates at mild temperatures (30°C) using commercially available catalysts and solvents, and has been demonstrated to be expandable to gram-level synthesis, indicating strong potential for kilogram-to-ton scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed synthesis in accelerating drug discovery and development pipelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle fluorinated chemistry with the highest safety standards, and our rigorous QC labs enforce stringent purity specifications to guarantee the quality of every batch produced.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global pharmaceutical market.
