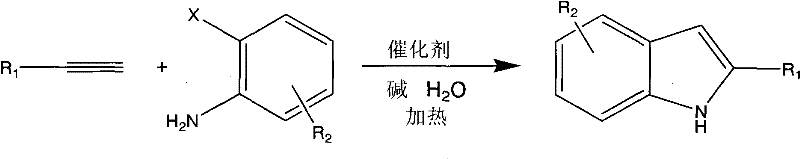
Revolutionizing Indole Production: Green Aqueous Phase Catalysis for Commercial Scale-up
The chemical industry is currently witnessing a paradigm shift towards sustainable manufacturing processes, driven by stringent environmental regulations and the economic necessity of reducing waste. Patent CN101906061A introduces a groundbreaking methodology for the synthesis of indole derivatives, a class of compounds fundamental to the pharmaceutical and agrochemical sectors. This patent details a novel approach utilizing a water-soluble coordination compound as a catalyst to facilitate the one-pot reaction between o-haloanilines and terminal alkynes in a pure aqueous phase. Unlike traditional methods that rely heavily on volatile organic compounds (VOCs), this technology leverages the unique properties of water to create an environmentally benign reaction medium. The significance of this innovation lies not only in its ecological footprint but also in its operational simplicity and high efficiency, offering a compelling value proposition for manufacturers seeking to optimize their production lines for complex heterocyclic compounds.
Furthermore, the adoption of this aqueous-phase catalysis represents a strategic move towards green chemistry principles, addressing the growing demand for cleaner synthetic routes in the production of high-value fine chemicals. By eliminating the need for hazardous organic solvents, the process inherently reduces the burden on waste treatment facilities and minimizes the risk of workplace exposure to toxic vapors. For R&D directors and process engineers, this patent provides a robust framework for developing scalable syntheses that maintain high purity standards while adhering to increasingly rigorous safety protocols. The ability to perform these transformations in water without compromising yield or selectivity marks a substantial advancement over legacy technologies, positioning adopters of this method at the forefront of sustainable chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of indole compounds has been plagued by significant technical and environmental challenges associated with the use of organic solvents. Traditional synthetic routes often necessitate the use of expensive, volatile, and toxic organic media to dissolve reactants and facilitate catalytic cycles. These solvents not only increase the raw material costs but also introduce severe safety hazards due to their flammability and potential for causing chronic health issues among operators. Moreover, the separation and purification steps in organic solvent-based systems are energy-intensive, requiring complex distillation setups to recover and recycle the solvents, which inevitably leads to material loss and increased carbon emissions. The reliance on precious metal catalysts in some conventional methods further exacerbates cost issues, as these metals are not only expensive to procure but also difficult to remove completely from the final product, posing risks for pharmaceutical applications where heavy metal residues are strictly regulated.
The Novel Approach
In stark contrast to these legacy issues, the method disclosed in CN101906061A utilizes a specialized water-soluble copper complex to drive the cyclization reaction in pure water. This approach fundamentally alters the reaction landscape by replacing hazardous organic media with the safest and most abundant solvent available. The use of water simplifies the workup procedure significantly, as the organic products can often be separated via simple extraction or filtration, bypassing the need for energy-heavy distillation. Additionally, the catalyst system described is designed to be highly active under relatively mild conditions, avoiding the extreme temperatures and pressures often required in older industrial processes. This transition to an aqueous system not only mitigates environmental pollution but also streamlines the entire manufacturing workflow, resulting in a process that is safer, cheaper, and more efficient.
Mechanistic Insights into Water-Soluble Copper Complex Catalysis
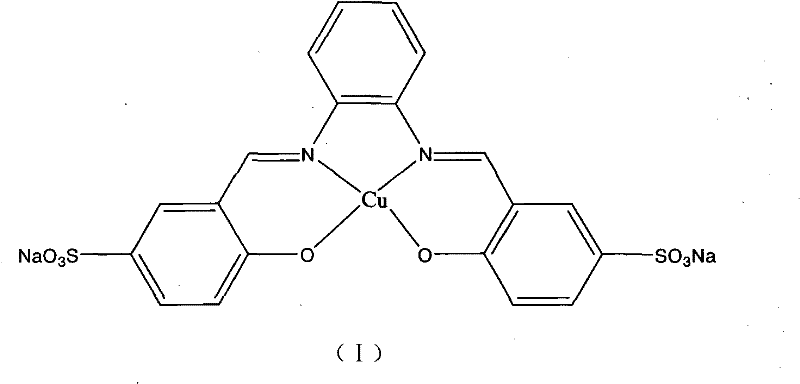
The core of this technological breakthrough lies in the specific design of the catalyst, a water-soluble metallic copper complex designated as structure (I) in the patent documentation. This complex features a ligand system modified with sulfonate groups, which impart high solubility in aqueous environments while maintaining the coordination geometry necessary for catalytic activity. The mechanism involves the activation of the terminal alkyne by the copper center, followed by a coupling reaction with the o-haloaniline substrate. The presence of the base, typically sodium hydroxide or potassium hydroxide, facilitates the deprotonation steps required for the cyclization to proceed. The unique electronic properties of the ligand stabilize the copper species in the water phase, preventing precipitation and ensuring a homogeneous catalytic cycle that maximizes contact between the reactants and the active sites. This homogeneity is crucial for achieving the high conversion rates and selectivity observed in the experimental examples provided in the patent.
Beyond the primary catalytic cycle, the system exhibits remarkable tolerance towards a wide array of functional groups, which is a critical factor for the synthesis of diverse pharmaceutical intermediates. The patent data indicates that substituents such as nitro, methoxy, methyl, and halogen groups on both the aniline and alkyne rings do not inhibit the reaction, allowing for the direct synthesis of complex substituted indoles without the need for protecting group strategies. This functional group compatibility simplifies the synthetic route considerably, reducing the number of steps and the overall consumption of reagents. Furthermore, the impurity profile of the resulting products is notably clean, with the patent reporting single major products in most cases. This high selectivity minimizes the formation of by-products, thereby reducing the complexity of downstream purification and ensuring that the final material meets the stringent quality specifications required for active pharmaceutical ingredients (APIs) and advanced fine chemicals.
How to Synthesize Indole Derivatives Efficiently
The practical implementation of this synthesis route is straightforward and amenable to standard chemical processing equipment. The general procedure involves charging a reaction vessel with the o-haloaniline substrate, the terminal alkyne, the water-soluble copper catalyst, and a suitable base in pure water. The mixture is then heated to a temperature range of 60°C to 130°C, depending on the specific reactivity of the substrates, and maintained for a period of 4 to 30 hours. Upon completion, the reaction mixture is cooled, and the product is isolated, often through simple extraction with an organic solvent like ethyl acetate or by filtration if the product precipitates. The detailed standardized synthesis steps, including specific molar ratios and purification techniques for various derivatives, are outlined in the guide below.
- Mix o-haloaniline and terminal alkyne substrates in pure water with a water-soluble copper complex catalyst.
- Add an inorganic base such as sodium hydroxide or potassium hydroxide to the reaction mixture.
- Heat the reaction mixture to temperatures between 60°C and 130°C for 4 to 30 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this aqueous-phase technology offers tangible economic and logistical benefits that extend beyond mere regulatory compliance. The elimination of bulk organic solvents from the reaction medium drastically reduces the volume of hazardous materials that need to be stored, transported, and handled on-site. This reduction in hazardous inventory lowers insurance premiums and minimizes the risk of supply chain disruptions caused by solvent shortages or transportation restrictions. Furthermore, the simplified workup process reduces the consumption of utilities such as steam and cooling water, leading to substantial operational cost savings over the lifecycle of the production facility. The robustness of the catalyst system also implies longer campaign lengths and reduced downtime for cleaning and maintenance, enhancing overall asset utilization.
- Cost Reduction in Manufacturing: The replacement of expensive organic solvents with water results in a direct and significant decrease in raw material costs. Additionally, the avoidance of complex solvent recovery systems reduces capital expenditure on equipment and lowers energy consumption associated with distillation processes. The high yields and selectivity reported in the patent minimize waste generation, further driving down the cost per kilogram of the final product. By eliminating the need for precious metal catalysts in favor of a more abundant copper-based system, the process also mitigates the financial volatility associated with noble metal markets.
- Enhanced Supply Chain Reliability: Utilizing water as the primary solvent removes the dependency on petrochemical-derived organic solvents, whose supply chains can be susceptible to geopolitical instability and price fluctuations. The starting materials, o-haloanilines and terminal alkynes, are widely available commodity chemicals, ensuring a stable and continuous supply of feedstocks. The simplified logistics of handling non-flammable aqueous mixtures also streamline the transportation of intermediates between different stages of production or to external partners, reducing lead times and improving responsiveness to market demand.
- Scalability and Environmental Compliance: The inherent safety of the aqueous system facilitates easier scale-up from laboratory to commercial production without the need for extensive hazard reassessments required for large-scale organic solvent operations. The process aligns perfectly with green chemistry initiatives, helping companies meet their sustainability goals and regulatory obligations regarding VOC emissions and wastewater discharge. The reduced environmental footprint enhances the corporate brand image and ensures long-term operational viability in regions with strict environmental enforcement, securing the supply chain against future regulatory shocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous-phase indole synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on the process capabilities and limitations. Understanding these details is essential for making informed decisions about adopting this method for large-scale manufacturing.
Q: What are the primary advantages of using water as a solvent for indole synthesis?
A: Using water eliminates the need for expensive and volatile organic solvents, significantly reducing environmental pollution and operational hazards while simplifying product separation and purification processes.
Q: Does this catalytic method tolerate various functional groups on the substrates?
A: Yes, the method demonstrates excellent functional group tolerance, accommodating substituents such as chloro, bromo, nitro, methoxy, and methyl groups on both the aniline and alkyne components without requiring protection steps.
Q: How does the water-soluble copper catalyst improve process safety?
A: The catalyst operates effectively in an aqueous environment, removing the flammability and explosion risks associated with organic solvents, thereby enhancing overall plant safety and reducing insurance and compliance costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the aqueous-phase catalytic technology described in CN101906061A for the production of high-quality indole derivatives. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory method into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this green chemistry approach are fully realized at an industrial level. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify the absence of heavy metal residues and organic impurities.
We invite global pharmaceutical and fine chemical companies to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact us today to request specific COA data for our indole intermediates and to discuss route feasibility assessments for your target molecules. Let us help you optimize your supply chain and achieve your sustainability goals through the adoption of cutting-edge catalytic technologies.
