Advanced Synthetic Route for Loxoprofen Sodium: Scalable Manufacturing & Cost Efficiency
Introduction to Advanced Loxoprofen Sodium Manufacturing
The pharmaceutical industry continuously seeks robust and economically viable pathways for the production of non-steroidal anti-inflammatory drugs (NSAIDs), with Loxoprofen Sodium standing out as a critical therapeutic agent. Patent CN101412670A discloses a highly efficient, eight-step synthetic methodology that transforms p-methylacetophenone into high-purity Loxoprofen Sodium through a sequence of reduction, activation, cyanation, hydrolysis, bromination, condensation, decarboxylation, and salt formation. This process is distinguished by its reliance on easily accessible raw materials and a unique operational stability that ensures high yields at every stage of the reaction sequence. Unlike traditional methods that often suffer from complex purification requirements or hazardous conditions, this novel approach emphasizes solvent recovery and reuse, directly addressing the growing demand for sustainable and cost-effective chemical manufacturing. The structural integrity and purity of the final product are rigorously maintained, making it suitable for direct formulation into pharmaceutical preparations.
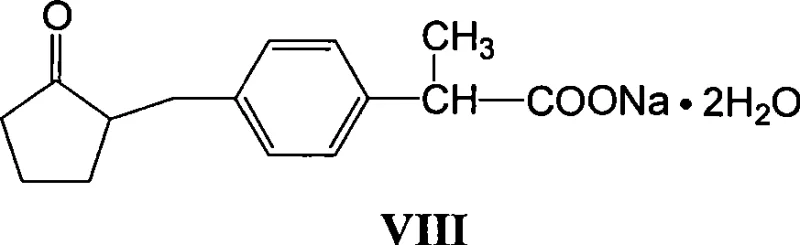
For global procurement teams and R&D directors, the significance of this patent lies in its ability to bypass the limitations of earlier synthetic strategies. By utilizing a linear progression from a simple aromatic ketone, the method minimizes the formation of difficult-to-separate impurities that typically plague multi-step syntheses. The detailed reaction conditions provided, ranging from mild reduction temperatures to controlled reflux parameters, offer a clear blueprint for commercial scale-up. This level of procedural clarity reduces the risk associated with technology transfer and ensures that the supply of this vital pharmaceutical intermediate remains consistent and reliable. Furthermore, the emphasis on recovering all organic solvents used in the synthesis process represents a substantial advancement in green chemistry principles, aligning modern manufacturing capabilities with stringent environmental compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-arylpropionic acids, the class to which Loxoprofen belongs, has been fraught with significant technical and economic challenges. Traditional approaches, such as the Friedel-Crafts alkylation using 2-chloro-2-methylthio propionic ester, are hindered by the difficulty in obtaining the alkylating reagent and the inherently high production costs associated with specialized precursors. Similarly, methods employing transition metal-catalyzed Grignard reactions require strictly anhydrous and anaerobic conditions, which demand expensive infrastructure and result in low yields due to unavoidable cross-coupling side reactions. Other routes, such as the Darzens reaction or those involving iodine rearrangement, introduce further complications including the use of highly basic sodium amide, long reaction times, and the generation of complex byproduct mixtures that are extremely difficult to purify. These legacy methods often rely on noble metal catalysts like palladium, which are not only costly but also present significant challenges in terms of recycling and residual metal removal, thereby restricting their suitability for large-scale industrial application.
The Novel Approach
In stark contrast, the methodology outlined in CN101412670A offers a streamlined and economically superior alternative that leverages the accessibility of p-methylacetophenone. This novel route eliminates the need for exotic reagents or harsh catalytic systems, instead utilizing standard organic transformations that are well-understood and easily controlled in a production environment. The process achieves high conversion rates through optimized stoichiometry and temperature control, such as the use of sodium borohydride for reduction under mild thermal conditions or phase-transfer catalysis for efficient cyanation. By avoiding the pitfalls of Grignard chemistry and expensive noble metals, this approach drastically simplifies the operational workflow and enhances the overall safety profile of the manufacturing plant. The ability to recover and reuse solvents like dichloromethane and toluene further amplifies the economic benefits, resulting in a process that is not only chemically robust but also financially competitive in the global market for NSAID intermediates.
Mechanistic Insights into Condensation and Decarboxylation
The core construction of the Loxoprofen skeleton occurs during the condensation step, where the brominated acid derivative reacts with ethyl 2-oxocyclopentanecarboxylate. This critical C-C bond-forming reaction is facilitated by a strong base in an organic solvent, promoting the nucleophilic attack of the enolate derived from the cyclopentanone ester onto the benzylic carbon of the bromide. The choice of base, ranging from inorganic hydroxides to alkoxides, and the precise control of reflux temperatures are paramount to driving this equilibrium towards the desired quaternary carbon center without inducing elimination side reactions. Following the condensation, the intermediate undergoes a sophisticated decarboxylation sequence under acidic reflux conditions using hydrobromic acid and acetic acid. This step serves a dual purpose: it hydrolyzes the ester functionality while simultaneously removing the carboxyl group introduced during the condensation, effectively unmasking the ketone moiety essential for the biological activity of the final drug substance.
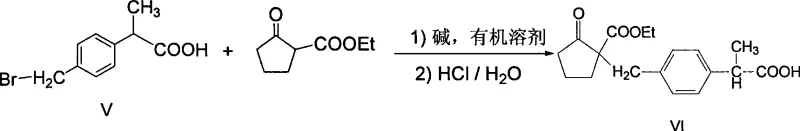
Understanding the mechanistic nuances of these steps is crucial for maintaining high purity levels, particularly regarding the control of stereoisomers and regioisomers. The acidic conditions employed during decarboxylation are carefully tuned to ensure complete conversion while minimizing the degradation of the sensitive cyclopentanone ring. The subsequent isolation of the free acid involves crystallization techniques that leverage specific solvent ratios, such as ethyl acetate and n-hexane, to exclude trace impurities and residual starting materials. This rigorous attention to detail in the reaction mechanism translates directly into a product with a clean impurity profile, which is a key requirement for regulatory approval in pharmaceutical manufacturing. The robustness of this chemical transformation ensures that the process can be scaled from laboratory benchtop to multi-ton production without compromising the structural integrity of the molecule.
How to Synthesize Loxoprofen Sodium Efficiently
The synthesis of Loxoprofen Sodium via this patented route involves a logical sequence of transformations that prioritize yield and operational simplicity. Beginning with the reduction of the ketone, the process moves through activation and cyanation to establish the propionic acid side chain, followed by functionalization of the aromatic ring to enable the final ring closure. Each step is designed to be telescoped or isolated with minimal loss of material, utilizing common laboratory unit operations such as extraction, washing, and distillation. The detailed protocol provided in the patent serves as a foundational guide for process chemists aiming to implement this technology, offering specific molar ratios and temperature ranges that have been empirically validated to maximize efficiency. For a comprehensive breakdown of the standardized operating procedures and safety protocols required for execution, please refer to the technical guide below.
- Reduction of p-methylacetophenone to the corresponding alcohol using sodium borohydride or catalytic hydrogenation.
- Activation of the alcohol via sulfonylation or halogenation followed by cyanation to form the nitrile intermediate.
- Hydrolysis of the nitrile to acid, followed by benzylic bromination and condensation with ethyl 2-oxocyclopentanecarboxylate.
- Final decarboxylation under acidic reflux conditions and subsequent salt formation with sodium hydroxide to yield Loxoprofen Sodium.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this synthetic route offers compelling advantages that directly impact the bottom line and supply chain resilience. The reliance on p-methylacetophenone as a starting material ensures a stable and abundant supply source, mitigating the risks associated with single-source or exotic raw materials that can disrupt production schedules. Furthermore, the elimination of precious metal catalysts removes a significant cost driver and simplifies the supply chain by reducing dependency on volatile commodity markets for metals like palladium. The process design inherently supports cost reduction through the systematic recovery and reuse of organic solvents, which constitutes a major portion of operating expenses in fine chemical manufacturing. This closed-loop solvent management system not only lowers material costs but also reduces the environmental footprint, aligning with corporate sustainability goals and regulatory requirements for waste minimization.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the avoidance of expensive reagents and the implementation of solvent recovery systems. By utilizing commodity chemicals like sodium borohydride and sodium cyanide instead of specialized organometallic reagents, the direct material costs are substantially lowered. Additionally, the high yield observed in each individual step cumulatively results in a superior overall process yield, meaning less raw material is wasted per kilogram of final product. The absence of complex purification steps, such as column chromatography, further reduces operational expenditures by allowing for simpler crystallization and filtration techniques that are easily scalable.
- Enhanced Supply Chain Reliability: The use of widely available reagents and standard reaction conditions ensures that the supply chain remains robust against external shocks. Since the process does not depend on custom-synthesized intermediates that may have long lead times, procurement teams can maintain leaner inventory levels while ensuring continuous production. The operational stability of the reaction steps, characterized by tolerance to minor variations in temperature and mixing, reduces the likelihood of batch failures that could otherwise cause significant delays. This reliability is critical for meeting the just-in-time delivery expectations of downstream pharmaceutical manufacturers who depend on a steady flow of high-quality intermediates.
- Scalability and Environmental Compliance: The pathway is explicitly designed for industrial scale-up, featuring reaction conditions that are safe and manageable in large reactors. The ability to recover and recycle solvents like dichloromethane and toluene dramatically reduces the volume of hazardous waste generated, simplifying compliance with environmental regulations. This green chemistry approach not only lowers disposal costs but also enhances the company's reputation as a responsible manufacturer. The straightforward workup procedures, involving phase separations and crystallizations, are inherently scalable, allowing for a seamless transition from pilot plant trials to full commercial production without the need for extensive re-engineering of the process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of Loxoprofen Sodium. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details helps stakeholders make informed decisions regarding process adoption and partnership opportunities. For more specific technical data or custom synthesis requirements, direct consultation with our technical team is recommended.
Q: What are the primary advantages of this synthetic route compared to traditional Grignard methods?
A: This route avoids the harsh anhydrous and anaerobic conditions required for Grignard reagents, significantly improving operational safety and reducing equipment costs while eliminating cross-coupling byproducts.
Q: How does the process address environmental and cost concerns regarding solvents?
A: The patent specifies that all solvents used throughout the synthesis, including dichloromethane, toluene, and alcohols, are recoverable and reusable, which drastically lowers waste disposal costs and raw material expenditure.
Q: Is the starting material p-methylacetophenone readily available for large-scale production?
A: Yes, p-methylacetophenone is a commodity chemical that is easy to obtain commercially, ensuring a stable supply chain and preventing bottlenecks associated with exotic or hard-to-synthesize starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Loxoprofen Sodium Supplier
At NINGBO INNO PHARMCHEM, we leverage deep expertise in process chemistry to deliver high-value pharmaceutical intermediates with unmatched consistency and quality. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Loxoprofen Sodium meets the highest international standards for safety and efficacy. Our commitment to excellence extends beyond mere compliance; we actively optimize processes to enhance yield and reduce environmental impact, providing our partners with a competitive edge in the global marketplace.
We invite you to collaborate with us to explore how this advanced synthetic route can benefit your supply chain and product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to demonstrate our capability to be your long-term strategic partner in the manufacture of complex organic intermediates.
