Advanced One-Pot Synthesis of Oxazolidine-Protected Aminodiols for Scalable Florfenicol Production
The veterinary pharmaceutical landscape is constantly evolving, driven by the need for more efficient and cost-effective manufacturing processes for critical antibiotics like Florfenicol. Patent CN101300237A introduces a transformative methodology for preparing oxazolidine-protected aminodiol compounds, which serve as pivotal intermediates in the synthesis of this broad-spectrum antibiotic. This technology addresses long-standing challenges in the production of Florfenicol, specifically targeting the inefficiencies associated with handling unstable amine intermediates. By leveraging a telescoped synthetic route, the patent outlines a process that not only enhances chemical yield but also streamlines the operational workflow for industrial-scale production. As a reliable veterinary pharmaceutical intermediate supplier, understanding these mechanistic advancements is crucial for optimizing supply chains and ensuring the consistent availability of high-quality active ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
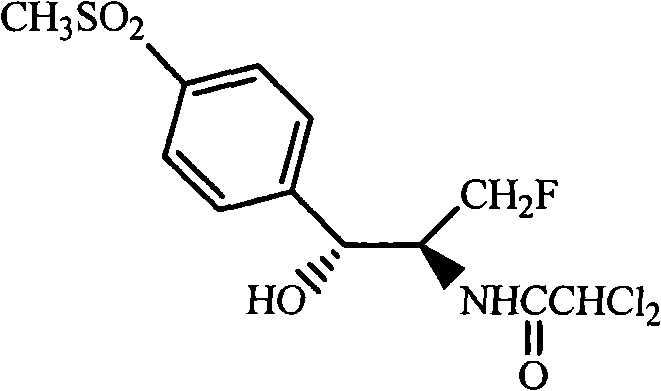
Historically, the synthesis of Florfenicol intermediates has been plagued by the difficulties associated with the aminodiol free base, often referred to as Formula III in prior art such as US Patent 5,663,361. This specific aminodiol compound possesses amphoteric properties, making its isolation and purification exceptionally challenging and costly. The zwitterionic nature of the molecule leads to poor solubility characteristics in many standard organic solvents, necessitating complex work-up procedures that often result in significant material loss. Furthermore, the instability of the free amine requires stringent handling conditions to prevent degradation, which adds layers of complexity to the manufacturing process. These factors collectively contribute to higher production costs and extended lead times, creating bottlenecks for manufacturers aiming to scale up production efficiently.
The Novel Approach
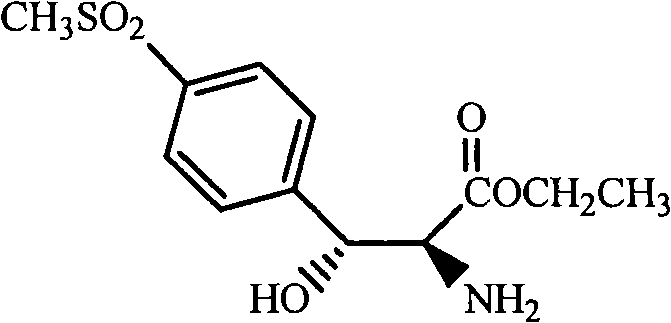
The innovative strategy disclosed in CN101300237A circumvents these issues by utilizing stable ester precursors, specifically compounds of Formula IV or VI, as the starting materials. Instead of isolating the problematic free aminodiol, the process generates it in situ through a reduction step and immediately subjects it to protection and acylation within the same reaction vessel. This telescoped approach effectively masks the reactive amine functionality as a stable oxazolidine ring, thereby eliminating the need for the difficult isolation step entirely. By starting with commercially available and stable esters, the process ensures a more robust and predictable reaction profile. This shift from isolating unstable intermediates to a continuous flow of transformations represents a significant leap forward in process chemistry, offering a pathway to substantial cost reduction in florfenicol manufacturing.
Mechanistic Insights into Telescoped Reduction and Protection
The core of this synthetic advancement lies in the precise orchestration of reduction, cyclization, and acylation reactions. The process initiates with the reduction of the ester precursor (Formula VI) using a hydride reducing agent. In alcoholic solvents such as methanol or ethanol, potassium borohydride (KBH4) is the preferred reductant, typically employed in a molar ratio ranging from 1:1 to 2:1 relative to the substrate. The reaction is conducted at moderate temperatures, generally between 30°C and 80°C, to ensure quantitative conversion to the aminodiol (Formula VII) without inducing side reactions. For anhydrous conditions, lithium aluminum hydride (LiAlH4) in tetrahydrofuran serves as a viable alternative, providing flexibility depending on the specific substrate requirements and facility capabilities.
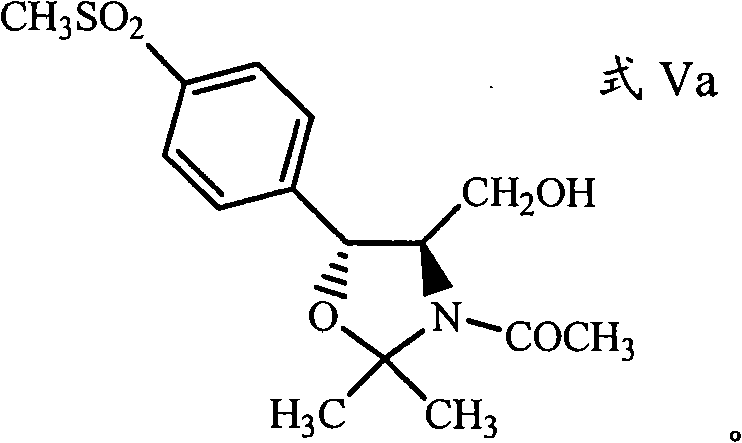
Following the reduction, the crude aminodiol solution is not isolated but is instead directly treated with an oxazolidine-forming reagent. Common reagents include acetone, 2,2-dimethoxypropane, or 2-methoxypropene, often in the presence of a base such as potassium carbonate or triethylamine. This step facilitates the formation of the oxazolidine ring (Formula VIII), which effectively protects the amino and hydroxyl groups. Subsequently, an N-acylating agent, such as acetyl chloride or propionyl chloride, is introduced to the same vessel to yield the final protected intermediate (Formula V). This seamless integration of steps minimizes exposure of the sensitive amine to potentially degrading conditions and significantly reduces the generation of waste solvents, aligning with modern principles of green chemistry and environmental compliance.
How to Synthesize Oxazolidine-Protected Aminodiols Efficiently
The synthesis of these critical intermediates requires careful control of reaction parameters to maximize yield and purity. The patented method provides a clear roadmap for executing this telescoped sequence, emphasizing the importance of solvent exchange and temperature control during the transition from reduction to protection. Detailed operational guidelines are essential for R&D teams looking to implement this technology, as slight variations in stoichiometry or thermal conditions can impact the formation of the oxazolidine ring. The following guide outlines the standardized procedure derived from the patent examples, ensuring reproducibility and safety during scale-up operations.
- Reduce the ester precursor (Formula VI) using a hydride reducing agent like KBH4 in an alcoholic solvent to generate the aminodiol in situ.
- React the crude aminodiol mixture directly with an oxazolidine-forming reagent such as acetone or 2,2-dimethoxypropane in the presence of a base.
- Perform N-acylation on the resulting oxazolidine intermediate using an acyl chloride to yield the protected aminodiol compound (Formula V).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this telescoped synthesis route offers compelling economic and logistical benefits. The primary advantage stems from the elimination of the isolation step for the aminodiol free base, which is traditionally a major source of yield loss and cost inflation. By processing the intermediate in situ, manufacturers can drastically reduce the volume of solvents required for extraction and washing, leading to direct savings in raw material costs and waste disposal fees. Furthermore, the use of stable ester starting materials simplifies inventory management, as these precursors have longer shelf lives and are less sensitive to storage conditions compared to their amine counterparts.
- Cost Reduction in Manufacturing: The telescoped nature of the reaction sequence significantly lowers the operational expenditure by reducing the number of unit operations. Eliminating the isolation and drying of the intermediate aminodiol saves considerable energy and labor hours. Additionally, the higher overall yield achieved by avoiding material loss during purification steps translates to a lower cost per kilogram of the final API. The process also utilizes common, inexpensive reagents like acetone and potassium borohydride, further enhancing the economic viability of the route compared to methods requiring exotic catalysts or complex protecting group strategies.
- Enhanced Supply Chain Reliability: Relying on stable ester precursors rather than unstable free amines mitigates the risk of supply disruptions caused by material degradation during transit or storage. The robustness of the starting materials ensures a consistent quality of input, which is critical for maintaining steady production schedules. Moreover, the simplified process flow reduces the dependency on specialized equipment for handling sensitive intermediates, allowing for greater flexibility in manufacturing site selection and capacity allocation. This resilience is vital for ensuring the continuous supply of veterinary antibiotics in a volatile global market.
- Scalability and Environmental Compliance: The reduction in solvent usage and waste generation inherent in this one-pot methodology aligns perfectly with increasingly stringent environmental regulations. Fewer work-up steps mean less aqueous and organic waste to treat, lowering the environmental footprint of the manufacturing process. From a scalability perspective, the straightforward addition of reagents in a single vessel simplifies the engineering controls required for large-scale production. This ease of scale-up facilitates the rapid transition from pilot plant to commercial manufacturing, ensuring that market demand for high-purity veterinary pharmaceutical intermediates can be met without prolonged lead times.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this route for their specific production needs.
Q: Why is the oxazolidine protection strategy superior to isolating the free aminodiol?
A: The free aminodiol (Formula III) is amphoteric and difficult to isolate and handle due to stability issues. The oxazolidine protection stabilizes the molecule, allowing for easier purification and higher overall yields in the subsequent fluorination steps.
Q: What are the preferred reducing agents for this synthesis?
A: The patent specifies potassium borohydride (KBH4) as a preferred reducing agent when using alcoholic solvents like methanol, typically at a molar ratio of 1.5:1. Lithium aluminum hydride (LiAlH4) is also suitable for anhydrous conditions in solvents like THF.
Q: How does this process impact the cost of Florfenicol manufacturing?
A: By telescoping the reduction, protection, and acylation steps into a single reaction vessel, the process eliminates the need to isolate the expensive and unstable aminodiol intermediate, significantly reducing solvent consumption, processing time, and yield losses associated with multiple work-ups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Florfenicol Intermediate Supplier
The technological advancements detailed in CN101300237A represent a significant opportunity for optimizing the production of Florfenicol intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage such innovative synthetic routes effectively. Our team is adept at translating complex patent methodologies into robust, commercial-scale processes. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our rigorous QC labs are equipped to monitor every stage of the synthesis, guaranteeing the high quality required for veterinary pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain. By collaborating with us, you can access a Customized Cost-Saving Analysis tailored to your production volumes and quality requirements. We encourage you to request specific COA data and route feasibility assessments to verify the compatibility of our capabilities with your project goals. Together, we can drive efficiency and reliability in the global supply of essential veterinary medicines.
