Advanced Palladium-Catalyzed Bis-Carbonylation for Scalable 4-Acyl-2(5H)-Furanone Production
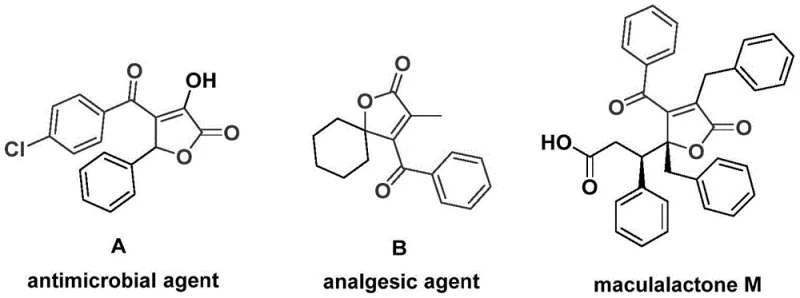
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex heterocyclic scaffolds efficiently. A significant breakthrough in this domain is documented in patent CN112239436A, which discloses a highly efficient preparation method for 4-acyl-2(5H)-furanone compounds. These structural motifs are not merely academic curiosities; they serve as critical backbones in numerous bioactive natural products and therapeutic agents. For instance, derivatives of this class have demonstrated significant antimicrobial and analgesic activities, making them invaluable targets for drug discovery programs. The disclosed technology leverages a sophisticated palladium-catalyzed bis-carbonylation strategy, transforming readily available propiolic alcohols and aryl trifluoromethanesulfonates into high-value furanone derivatives in a single operational step. This innovation addresses long-standing challenges in heterocycle synthesis, offering a pathway that is both atom-economical and operationally simple, thereby positioning it as a cornerstone technology for modern pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 4-acyl-2(5H)-furanone skeleton has been fraught with synthetic inefficiencies that hinder large-scale production. Traditional routes often rely on multi-step sequences involving harsh reaction conditions, expensive reagents, or unstable intermediates that complicate purification protocols. Many classical approaches require the use of gaseous carbon monoxide under high pressure, which introduces significant safety hazards and necessitates specialized equipment that is not universally available in standard laboratory or pilot plant settings. Furthermore, conventional methods frequently suffer from poor regioselectivity and limited substrate scope, failing to accommodate sensitive functional groups that are often present in advanced drug candidates. These limitations result in lower overall yields, increased waste generation, and prolonged lead times, creating bottlenecks for cost reduction in API manufacturing and delaying the availability of critical medicinal chemistry building blocks.
The Novel Approach
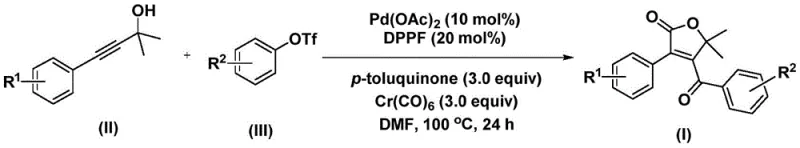
In stark contrast to these legacy methods, the technology described in CN112239436A introduces a streamlined, one-pot catalytic cycle that dramatically simplifies the synthetic landscape. By utilizing chromium hexacarbonyl as a solid carbon monoxide surrogate, the process eliminates the need for handling hazardous CO gas, significantly enhancing operational safety and ease of execution. The reaction proceeds under relatively mild thermal conditions (100-120 °C) in polar aprotic solvents like DMF, ensuring high conversion rates without degrading sensitive substrates. This novel approach enables the direct assembly of the furanone core through a tandem carbonylation-cyclization sequence, effectively merging two distinct bond-forming events into a single transformative step. The result is a methodology that not only accelerates the synthesis timeline but also expands the chemical space accessible to medicinal chemists, allowing for the rapid generation of diverse libraries for biological screening.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
The elegance of this synthesis lies in its intricate yet efficient catalytic mechanism, which orchestrates the assembly of the furanone ring through a series of well-defined organometallic steps. The cycle initiates with the oxidative addition of the aryl trifluoromethanesulfonate to the active palladium(0) species, generating a key aryl-palladium intermediate. Subsequently, carbon monoxide, liberated in situ from the thermal decomposition of chromium hexacarbonyl, inserts into the palladium-carbon bond to form an acyl-palladium complex. This acyl species then undergoes nucleophilic attack by the propiolic alcohol substrate, triggering a cyclization event that constructs the five-membered lactone ring. A second insertion of carbon monoxide follows, expanding the coordination sphere before a final reductive elimination releases the desired 4-acyl-2(5H)-furanone product and regenerates the active catalyst. This mechanistic pathway highlights the dual role of the palladium catalyst in facilitating both C-C bond formation and carbonyl incorporation, ensuring high fidelity in the construction of the target scaffold.
From an impurity control perspective, the choice of ligands and additives plays a pivotal role in maintaining product purity. The use of 1,1'-bis(diphenylphosphino)ferrocene (DPPF) as a bidentate ligand stabilizes the palladium center, preventing the formation of inactive palladium black and minimizing side reactions such as homocoupling of the aryl halide. Additionally, the inclusion of p-toluquinone as an additive serves to re-oxidize any reduced palladium species or scavenge hydrides that could lead to reduced byproducts, thereby sharpening the selectivity profile of the reaction. The compatibility of this system with a wide range of substituents—including electron-rich methoxy groups and electron-deficient halogens—demonstrates the robustness of the catalytic cycle against electronic perturbations. This tolerance ensures that the high-purity pharmaceutical intermediates produced meet stringent quality specifications required for downstream processing, reducing the burden on purification teams.
How to Synthesize 4-Acyl-2(5H)-Furanone Efficiently
The practical implementation of this synthesis is designed for scalability and reproducibility, making it an ideal candidate for technology transfer from bench to plant. The protocol involves charging a reaction vessel with the propiolic alcohol, aryl triflate, palladium catalyst, ligand, and the solid CO source in a suitable solvent like DMF. The mixture is then heated to the specified temperature range, allowing the cascade reaction to proceed to completion over a defined period. Post-reaction processing is remarkably straightforward, typically involving simple filtration to remove metal residues followed by standard chromatographic purification.
- Combine palladium acetate, DPPF ligand, chromium hexacarbonyl, p-toluquinone, propiolic alcohol, and aryl triflate in DMF solvent.
- Heat the reaction mixture to 100-120°C and stir for 24-48 hours to facilitate bis-carbonylation and cyclization.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 4-acyl-2(5H)-furanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical novelty. The shift towards this catalytic process represents a fundamental optimization of the supply chain for heterocyclic building blocks, addressing key pain points related to cost, safety, and reliability. By replacing hazardous gaseous reagents with stable solids and consolidating multiple synthetic steps into one, the process inherently reduces the logistical complexity and risk profile associated with manufacturing these intermediates.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven primarily by the simplification of the operational workflow and the utilization of commodity-grade starting materials. Eliminating the need for high-pressure CO equipment and multi-step isolation procedures drastically lowers capital expenditure and operational overheads. Furthermore, the high yields reported (often exceeding 80-90% for optimized substrates) mean that less raw material is wasted, directly improving the cost-of-goods sold (COGS). The use of chromium hexacarbonyl as a convenient CO source avoids the infrastructure costs associated with gas handling, while the recovery of palladium catalysts can further enhance the economic viability of the process on a multi-ton scale.
- Enhanced Supply Chain Reliability: Sourcing reliable pharmaceutical intermediates is often complicated by the fragility of complex synthetic routes. This method enhances supply security by relying on widely available precursors such as substituted phenols (for triflates) and commercially accessible alkynols. The robustness of the reaction conditions ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed runs or difficult purifications. Additionally, the shortened synthetic timeline allows for faster response to market demand fluctuations, enabling suppliers to maintain healthier inventory levels and reduce lead times for critical drug development projects.
- Scalability and Environmental Compliance: As regulatory pressures regarding waste disposal and solvent usage intensify, the environmental footprint of chemical processes becomes a decisive factor. This one-pot synthesis generates significantly less waste compared to traditional multi-step sequences, aligning with green chemistry principles. The ability to run the reaction in standard solvents like DMF, coupled with a simple workup procedure, facilitates easier scale-up from kilogram to metric ton quantities without requiring exotic engineering solutions. This scalability ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved smoothly, supporting the continuous supply needs of global healthcare markets.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its potential integration into their supply chains. The following questions address common inquiries regarding the practical application and scope of this patented technology, providing clarity on its operational parameters and versatility.
Q: What is the source of carbon monoxide in this synthesis?
A: The process utilizes chromium hexacarbonyl (Cr(CO)6) as a solid, safe substitute for gaseous carbon monoxide, releasing CO in situ under heating conditions.
Q: What represents the optimal catalyst system for this transformation?
A: The patent specifies a system comprising Palladium Acetate (Pd(OAc)2) as the catalyst and 1,1'-Bis(diphenylphosphino)ferrocene (DPPF) as the ligand, achieving high turnover efficiency.
Q: Can this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the method demonstrates excellent substrate compatibility, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens and trifluoromethyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acyl-2(5H)-Furanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN112239436A for accelerating drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the highest standards of quality and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 4-acyl-2(5H)-furanone delivered is ready for immediate use in sensitive biological assays or further synthetic elaboration.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this efficient synthetic route for their specific project needs. By engaging with our technical team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this methodology can optimize your budget. We encourage you to contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments, ensuring your supply chain is built on a foundation of scientific excellence and commercial reliability.
