Advanced Oxidation Strategy for Benzoylformic Acid Derivatives: A Scalable Solution for Pharmaceutical Manufacturing
Advanced Oxidation Strategy for Benzoylformic Acid Derivatives: A Scalable Solution for Pharmaceutical Manufacturing
The pharmaceutical industry continuously demands more efficient and safer synthetic routes for critical building blocks, particularly benzene ring polysubstituted compounds based on benzoylformic acid. Patent CN111116358B introduces a transformative synthesis method that addresses long-standing safety and efficiency challenges in producing these vital pharmaceutical intermediates. This novel approach utilizes a specific oxidant system comprising dichromate in the presence of an organic solvent and acidic substance to convert acetophenone derivatives into their corresponding benzoylformic acid analogues. By shifting away from traditional hazardous reagents, this technology offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates, ensuring both operational safety and high chemical fidelity. The method demonstrates exceptional versatility across a wide range of substituents, including halogens and alkyl groups, making it a cornerstone technology for modern API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenylglyoxylic acid derivatives has relied heavily on selenium dioxide (SeO2) as the primary oxidant, typically employed in pyridine solvents. While chemically effective, this legacy process presents severe drawbacks for modern manufacturing environments. The reduction of selenium dioxide generates elemental selenium as a byproduct, which poses significant environmental hazards and can sublime into extremely toxic gases upon exposure to high heat, creating dangerous working conditions. Furthermore, pyridine is a solvent known for its high toxicity, unpleasant odor, and difficulty in removal, often requiring rigorous purification steps that increase processing time and cost. These factors collectively result in lower overall safety profiles and operational inefficiencies, hindering the cost reduction in pharmaceutical intermediate manufacturing that procurement teams strive to achieve.
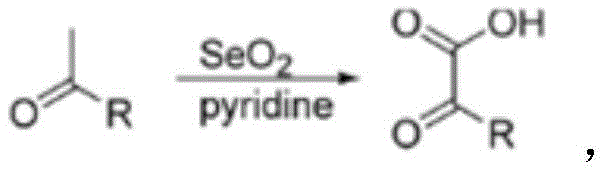
The Novel Approach
In stark contrast, the methodology disclosed in CN111116358B replaces the hazardous selenium-pyridine system with a dichromate-based oxidation protocol utilizing trifluorotoluene as the solvent. This strategic substitution fundamentally alters the reaction landscape, enabling oxidation to proceed under milder conditions, typically between 90°C and 100°C, with significantly enhanced safety margins. The use of trifluorotoluene not only provides excellent solubility for the substrates but also offers superior stability towards the oxidant, facilitating a smoother reaction profile. As demonstrated in the specific synthesis of 2-(2-chlorobenzene)-2-glyoxylic acid, this new route achieves high conversion rates with simplified workup procedures, effectively eliminating the risks associated with selenium sublimation. This innovation represents a critical step forward for any reliable pharmaceutical intermediate supplier aiming to deliver high-purity products without compromising on safety or environmental standards.
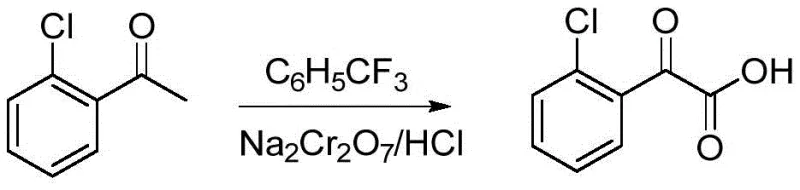
Mechanistic Insights into Dichromate-Catalyzed Oxidation
The core of this synthetic advancement lies in the precise control of the oxidation state of the benzylic carbon through the use of sodium dichromate in an acidic medium. Under these conditions, the dichromate ion acts as a potent oxidizing agent, facilitating the transformation of the methyl ketone group into an alpha-keto acid functionality. The acidic environment, typically maintained by hydrochloric acid, plays a dual role: it activates the oxidant species and stabilizes the reaction intermediates, preventing over-oxidation or degradation of the sensitive aromatic ring. This mechanistic precision ensures that the structural integrity of diverse substituents, such as fluorine, chlorine, bromine, and iodine, is preserved throughout the reaction, which is crucial for maintaining the biological activity of downstream API candidates. The reaction kinetics are optimized to proceed efficiently within a 5 to 10-hour window, balancing reaction speed with thermal management to prevent side reactions.
Impurity control is another critical aspect where this mechanism excels, particularly during the post-reaction workup phase. The process employs a sophisticated pH-swing purification strategy where the reaction mixture is first adjusted to an alkaline pH of approximately 9. This step effectively solubilizes the desired carboxylic acid product while allowing neutral organic impurities and inorganic salts to be separated or removed. Subsequently, re-acidification to a pH of around 3 precipitates the pure benzoylformic acid derivative, which can then be further purified via recrystallization using petroleum ether. This rigorous purification protocol ensures that the final product meets stringent purity specifications required for pharmaceutical applications, minimizing the burden on downstream processing and ensuring consistent quality for high-purity pharmaceutical intermediates.
How to Synthesize 2-(2-chlorobenzene)-2-glyoxylic acid Efficiently
The practical implementation of this synthesis route is designed for straightforward execution in standard chemical reactors, making it highly accessible for process chemists. The procedure begins with the dissolution of the acetophenone starting material in trifluorotoluene, followed by the careful addition of aqueous hydrochloric acid to establish the necessary acidic environment. Once the system is stabilized, sodium dichromate is introduced, and the mixture is heated to reflux temperatures between 90°C and 100°C. Maintaining these conditions for the prescribed duration ensures complete conversion of the starting material. Following the reaction, the solvent is removed, and the pH adjustment sequence is executed to isolate the product. For a comprehensive understanding of the specific operational parameters and stoichiometry, detailed standardized synthesis steps are provided in the guide below.
- Dissolve the acetophenone derivative in trifluorotoluene and add aqueous HCl.
- Add sodium dichromate under acidic conditions and reflux at 90-100°C for 5-10 hours.
- Remove solvent, adjust pH to 9 to remove impurities, then acidify to pH 3 to precipitate and recrystallize the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this dichromate-based oxidation method offers substantial benefits that resonate deeply with procurement and supply chain objectives. By eliminating the need for selenium dioxide, manufacturers can avoid the complex regulatory and disposal costs associated with selenium waste, leading to significant operational savings. The switch to trifluorotoluene, a solvent with favorable boiling point characteristics, allows for energy-efficient recovery and reuse, further driving down the cost of goods sold. Moreover, the robustness of the reaction conditions means that production schedules are less likely to be disrupted by safety incidents or difficult purification bottlenecks, ensuring a steady flow of materials for downstream synthesis. This reliability is paramount for maintaining continuous supply chains in the competitive pharmaceutical sector.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous selenium reagents directly reduces raw material costs, while the simplified workup procedure minimizes labor and utility consumption. The ability to recover and recycle the trifluorotoluene solvent adds another layer of economic efficiency, making the overall process financially attractive for large-scale production. Additionally, the high yields reported across various substrates mean less starting material is wasted, maximizing the value extracted from every batch. These factors combine to create a leaner, more cost-effective manufacturing model that enhances profit margins without sacrificing quality.
- Enhanced Supply Chain Reliability: The reagents required for this process, such as sodium dichromate and substituted acetophenones, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages. The mild reaction conditions reduce the wear and tear on reactor equipment, extending asset life and minimizing unplanned maintenance downtime. Furthermore, the reduced toxicity profile simplifies logistics and storage requirements, allowing for more flexible inventory management. This stability ensures that partners can rely on consistent delivery timelines, which is critical for meeting the demanding schedules of drug development programs.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from gram to multi-kilogram scales without loss of efficiency. The absence of toxic selenium byproducts simplifies wastewater treatment and aligns with increasingly strict environmental regulations, reducing the risk of compliance penalties. The use of standard reflux equipment means that existing manufacturing infrastructure can be utilized without the need for specialized high-pressure or cryogenic installations. This ease of scale-up facilitates rapid transition from pilot plant to commercial production, accelerating time-to-market for new therapeutic candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology, derived directly from the patent specifications and experimental data. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing workflows. The answers reflect the proven capabilities of the dichromate oxidation system in delivering high-quality intermediates safely and efficiently. For further technical clarification or custom synthesis requests, our team is available to provide in-depth consultations.
Q: Why is the dichromate method preferred over selenium dioxide for benzoylformic acid synthesis?
A: The dichromate method eliminates the risk of toxic elemental selenium sublimation and avoids the use of highly toxic pyridine solvent, offering a safer and more environmentally compliant process.
Q: What are the typical yields for this oxidation process?
A: The patent data indicates consistently high yields ranging from 85% to 91% across various substituted acetophenone substrates, demonstrating robust substrate tolerance.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of standard reflux conditions (90-100°C) and commercially available reagents like sodium dichromate and trifluorotoluene makes it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoylformic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development. Our expertise in scaling diverse synthetic pathways allows us to bridge the gap between laboratory innovation and industrial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of benzoylformic acid derivatives meets the highest industry standards. We are committed to being a partner that not only supplies chemicals but also provides technical solutions that enhance your overall process efficiency.
We invite you to explore how our advanced synthesis capabilities can support your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate the value of our optimized manufacturing processes. Let us help you secure a reliable supply chain for your critical intermediates, ensuring your projects move forward without interruption.
