Advanced Gas-Phase Oxidation Catalyst for High-Purity Benzoylformic Acid Production
The chemical industry is currently witnessing a paradigm shift in the synthesis of high-value alpha-keto acids, driven by the urgent need for greener and more economically viable manufacturing processes. A pivotal development in this sector is documented in patent CN109499618B, which discloses a novel heterogeneous catalyst system designed specifically for the one-step preparation of benzoylformic acid and its ester derivatives. This technology represents a significant departure from traditional batch-wise liquid-phase oxidations, introducing a continuous gas-phase oxidation methodology that utilizes styrene—a widely available petrochemical feedstock—as the primary starting material. By leveraging a sophisticated composite catalyst comprising ferrous oxide, manganese dioxide, tungsten oxide, and rare earth metal complexes, this innovation achieves exceptional selectivity and yield while virtually eliminating the generation of hazardous heavy metal waste. For global procurement leaders and R&D directors seeking a reliable benzoylformic acid supplier, understanding the mechanistic advantages and supply chain implications of this patent is critical for securing long-term competitive advantages in the pharmaceutical and agrochemical markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of benzoylformic acid has been plagued by severe economic and environmental bottlenecks inherent to legacy synthetic routes. One prevalent method involves the hydrolysis of benzoyl nitrile under acidic conditions, a process depicted in the reaction scheme below. While chemically straightforward, this route relies on benzoyl nitrile, a precursor that is not only expensive to source but also introduces significant safety hazards due to the presence of cyanide groups, leading to complex wastewater treatment requirements and elevated operational costs.
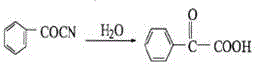
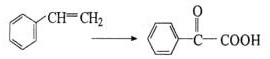
Furthermore, alternative oxidation pathways, such as the liquid-phase oxidation of styrene using potassium permanganate, suffer from catastrophic atom economy issues. As illustrated in the following reaction diagram, this method consumes stoichiometric amounts of expensive oxidants and generates massive quantities of manganese dioxide sludge as a byproduct. This manganese mud constitutes a hazardous solid waste that requires costly disposal procedures, rendering the process environmentally unsustainable and economically unviable for modern large-scale pharmaceutical intermediates manufacturing. Additionally, methods utilizing chromium-based oxidants pose severe toxicity risks, creating regulatory hurdles for companies aiming to maintain green supply chains.
The Novel Approach
In stark contrast to these inefficient batch processes, the technology disclosed in CN109499618B introduces a revolutionary gas-phase catalytic oxidation strategy that fundamentally redefines the production landscape. By employing a fixed-bed reactor system, this method enables the continuous conversion of styrene and air directly into benzoylformic acid in a single step. The elimination of stoichiometric liquid oxidants in favor of molecular oxygen from air drastically reduces raw material costs and removes the burden of heavy metal waste disposal. This continuous flow approach not only enhances process safety by avoiding the accumulation of reactive intermediates but also ensures a consistent product quality profile that is essential for downstream applications in drug synthesis. The ability to utilize cheap, bulk commodity chemicals like styrene and ethylbenzene as feedstocks positions this technology as a cornerstone for cost reduction in fine chemical intermediates manufacturing.
Mechanistic Insights into Fe-Mn-W Composite Catalytic Oxidation
The core innovation of this patent lies in the precise engineering of a multi-component heterogeneous catalyst that balances oxidative activity with selectivity. The main catalytic body consists of ferrous oxide (Fe2O3) acting as the primary active site for oxygen activation, synergistically promoted by manganese dioxide (MnO2) and tungsten oxide (WO3). This specific tri-metallic oxide framework is crucial for facilitating the selective oxidation of the vinyl group in styrene to the alpha-keto functionality without causing over-oxidation to benzoic acid or complete combustion to carbon dioxide. The inclusion of tungsten oxide is particularly strategic, as it modifies the surface acidity and electronic properties of the catalyst, thereby stabilizing the intermediate species and preventing the cleavage of the carbon-carbon bond which would otherwise lead to yield loss.
Further enhancing this system is the incorporation of auxiliary promoters in the form of lanthanum and strontium complexes, specifically L-homophenylalanine lanthanum and D-strontium homomalate. These chiral organic-metallic complexes serve a dual purpose: they act as structural promoters that disperse the active metal oxides on the carrier surface (such as ZSM-5 molecular sieves or gamma-alumina), and they potentially influence the stereoelectronic environment of the active sites to favor the formation of the desired keto-acid. This sophisticated formulation ensures that the reaction proceeds with high molar selectivity, typically exceeding 90% under optimized conditions, while maintaining catalyst stability over extended operation periods. For R&D teams, this implies a robust process capable of delivering high-purity benzoylformic acid with minimal byproduct formation, simplifying downstream purification.
How to Synthesize Benzoylformic Acid Efficiently
The implementation of this catalytic system requires a systematic approach to reactor engineering and process control to maximize the benefits of the gas-phase oxidation pathway. The synthesis protocol outlined in the patent emphasizes the importance of catalyst preparation, specifically the impregnation and calcination steps, which determine the final dispersion of active sites. Once the catalyst is prepared and loaded into a fixed-bed reactor, the process operates under continuous flow conditions where vaporized styrene is mixed with air and passed over the catalyst bed at controlled temperatures ranging from 200°C to 290°C. Detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- Prepare the composite catalyst by impregnating a carrier (e.g., ZSM-5) with ferrous oxide, manganese dioxide, tungsten oxide, and lanthanum/strontium complexes, followed by calcination.
- Load the catalyst into a fixed-bed reactor and control the temperature between 200-290°C and pressure at 1-5 MPa.
- Introduce vaporized styrene mixed with air (ratio 1: 9 to 1:20) at a weight space velocity of 1-5 h⁻¹ to produce benzoylformic acid continuously.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this gas-phase catalytic technology offers transformative benefits that extend far beyond simple yield improvements. The shift from batch liquid-phase oxidation to continuous gas-phase processing fundamentally alters the cost structure of benzoylformic acid production. By replacing expensive, specialized oxidants like potassium permanganate or sodium dichromate with freely available atmospheric air, the variable costs associated with reagents are significantly reduced. Moreover, the use of styrene, a commodity chemical produced in millions of tons annually for the polymer industry, ensures a stable and low-cost raw material supply base, insulating manufacturers from the price volatility often seen with niche fine chemical precursors.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the elimination of stoichiometric oxidants and the reduction of waste treatment expenses. Traditional methods generate substantial volumes of heavy metal-containing wastewater and solid sludge, the disposal of which incurs high regulatory and operational costs. By achieving a "zero three-waste" discharge status through the use of a clean gas-phase reaction, manufacturers can bypass expensive effluent treatment protocols. Additionally, the continuous nature of the fixed-bed reactor allows for higher throughput and better energy efficiency compared to batch reactors, leading to substantial overall cost savings in the production of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for global buyers, and this technology directly addresses potential bottlenecks. Since the process relies on styrene and air, both of which are available from a vast global network of petrochemical suppliers, the risk of raw material shortage is minimized. Unlike processes dependent on mandelic acid or benzoyl nitrile, which may have limited production capacity and longer lead times, the feedstock for this method is ubiquitous. Furthermore, the continuous operation mode of the fixed-bed reactor ensures a steady stream of product output, enabling suppliers to meet large-volume orders with consistent lead times and reducing the risk of production delays associated with batch turnover.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but the fixed-bed gas-phase design is inherently scalable from pilot plants to industrial megatons. The simplicity of the reaction engineering—essentially mixing gas and passing it over a solid bed—allows for straightforward capacity expansion without the complex heat transfer and mixing issues found in large liquid-phase oxidation tanks. From an environmental perspective, the process aligns perfectly with increasingly stringent global regulations regarding heavy metal emissions. The absence of chromium, manganese sludge, or aluminum chloride waste means that facilities utilizing this technology face fewer regulatory hurdles and lower compliance risks, ensuring long-term operational viability in eco-conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of adopting this route for commercial production. Understanding these details is essential for stakeholders assessing the transition from traditional synthesis methods to this advanced catalytic system.
Q: How does this new catalyst improve environmental compliance compared to traditional methods?
A: Unlike traditional liquid-phase oxidation using potassium permanganate or sodium dichromate which generates heavy metal sludge and toxic wastewater, this gas-phase process uses air as the oxidant and produces zero three-waste discharge, significantly simplifying environmental treatment.
Q: What are the primary cost advantages of using styrene as the raw material?
A: Styrene is a bulk petrochemical commodity available at a fraction of the cost of specialized precursors like benzoyl nitrile or mandelic acid. Coupled with the use of air instead of expensive stoichiometric oxidants, the raw material costs are drastically reduced.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent describes a continuous fixed-bed reactor setup rather than a batch process. This design inherently supports scalable industrial manufacturing, allowing for consistent product quality and higher throughput compared to batch oxidation methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoylformic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic technologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO specializing in complex organic synthesis, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art fixed-bed reactor systems and rigorous QC labs capable of meeting stringent purity specifications required by the global pharmaceutical industry. We are committed to leveraging innovations like the Fe-Mn-W composite catalyst to deliver high-quality intermediates that drive efficiency for our clients.
We invite you to collaborate with us to explore how this green synthesis route can optimize your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation translates into tangible value for your business.
