Advanced Rhodium-Catalyzed Synthesis of Isoxazolidine Scaffolds for Commercial Pharmaceutical Applications
Introduction to Next-Generation Isoxazolidine Manufacturing
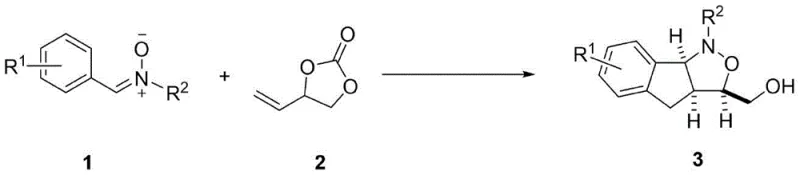
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for more efficient, sustainable, and scalable processes. A significant breakthrough in this domain is detailed in patent CN115215814A, which discloses a novel synthetic method for isoxazolidine compounds. This technology leverages advanced transition metal catalysis to construct complex heterocyclic scaffolds that are ubiquitous in bioactive natural products and therapeutic agents. For R&D directors and procurement specialists alike, understanding the nuances of this rhodium-catalyzed C-H activation strategy is crucial. The process utilizes nitrone compounds and ethylene vinyl carbonates as primary building blocks, reacting them under the influence of a rhodium catalyst system to yield highly functionalized isoxazolidine derivatives. This approach represents a paradigm shift from traditional multi-step syntheses, offering a streamlined pathway that aligns with modern green chemistry principles while maintaining the rigorous purity standards required for high-purity pharmaceutical intermediates.
The significance of the isoxazolidine skeleton cannot be overstated in medicinal chemistry. Molecules containing this unique structural motif, such as Pyrinodemin A-D and (-)-Flueggine A, have demonstrated potent biological activities ranging from antitumor properties to treatments for neurological disorders. Consequently, the ability to access these structures efficiently is a key competitive advantage. The patented method addresses historical challenges associated with constructing these rings, particularly regarding stereocontrol and substrate compatibility. By establishing a robust protocol that operates under relatively mild thermal conditions (40-80°C) and utilizes commercially accessible reagents, this technology paves the way for the commercial scale-up of complex heterocycles. It provides a reliable foundation for generating diverse libraries of compounds for drug screening programs, thereby accelerating the timeline from lead identification to clinical candidate selection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isoxazolidine frameworks has relied heavily on 1,3-dipolar cycloadditions between nitrones and electron-deficient alkenes. While effective in specific contexts, these classical approaches often suffer from significant limitations that hinder their utility in large-scale industrial applications. One major drawback is the requirement for highly activated dipolarophiles, which can be expensive, unstable, or difficult to source in bulk quantities. Furthermore, traditional methods frequently struggle with regioselectivity and stereoselectivity, leading to complex mixtures of isomers that require tedious and yield-eroding purification steps. In many cases, harsh reaction conditions involving strong acids, bases, or extreme temperatures are necessary to drive the reaction to completion, which poses safety risks and increases energy consumption. These factors collectively contribute to higher production costs and longer lead times, creating bottlenecks in the supply chain for reliable pharmaceutical intermediate suppliers who must meet strict delivery schedules.
The Novel Approach
In stark contrast, the methodology described in CN115215814A introduces a transformative catalytic cycle that overcomes these inherent inefficiencies. By employing a cationic rhodium(III) catalyst system, specifically [Cp*RhCl2]2 activated by AgSbF6, the process enables the direct coupling of nitrones with vinyl carbonates through a C-H activation mechanism. This novel approach eliminates the need for pre-functionalized alkene partners, significantly simplifying the starting material portfolio. The reaction proceeds with exceptional regio- and stereoselectivity, predominantly yielding the trans-fused isoxazolidine products with high fidelity. This precision reduces the burden on downstream purification, directly translating to improved overall yields and reduced waste generation. Moreover, the operational simplicity of the protocol—often requiring just mixing reagents in a solvent like chlorobenzene and heating—makes it highly amenable to automation and scale-up. This innovation effectively lowers the barrier to entry for producing complex isoxazolidine derivatives, offering substantial cost reduction in API manufacturing by minimizing raw material costs and processing time.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation
To fully appreciate the technical robustness of this synthesis, one must delve into the mechanistic underpinnings of the rhodium-catalyzed transformation. The reaction initiates with the generation of the active cationic rhodium(III) species through the abstraction of chloride ligands by the silver salt additive. This electrophilic metal center then coordinates with the nitrone substrate, facilitating the cleavage of a proximal C-H bond on the aromatic ring or alkene moiety, depending on the specific substrate design. This C-H activation step is the turnover-limiting event in many such catalytic cycles, but the optimized ligand environment provided by the pentamethylcyclopentadienyl (Cp*) group ensures high reactivity even at moderate temperatures. Following metallacycle formation, the vinyl carbonate inserts into the Rh-C bond, setting the stage for the subsequent cyclization.
The final steps involve the reductive elimination or nucleophilic attack that closes the isoxazolidine ring and regenerates the active catalyst. A critical aspect of this mechanism is the control of stereochemistry. The rigid geometry of the rhodacycle intermediate dictates the facial selectivity of the alkene insertion, ensuring that the newly formed stereocenters are established with high diastereomeric ratios. This intrinsic stereocontrol is vital for pharmaceutical applications where the biological activity is often dependent on a specific enantiomer or diastereomer. Furthermore, the presence of additives like Ag2CO3 plays a dual role: acting as a base to neutralize acidic byproducts and potentially assisting in the stabilization of the catalytic species. Understanding these mechanistic details allows process chemists to fine-tune reaction parameters, such as solvent polarity and temperature, to maximize efficiency and minimize impurity profiles, ensuring the production of high-purity isoxazolidine derivatives suitable for sensitive biological assays.
How to Synthesize Isoxazolidine Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational protocols to ensure reproducibility and safety. The general procedure involves charging a pressure-resistant vessel with the nitrone substrate and the vinyl carbonate coupling partner in a molar ratio typically ranging from 1:1 to 1:1.5. The catalytic system is then introduced, comprising the rhodium dimer and the silver salt activator, along with an appropriate additive such as silver carbonate. The choice of solvent is critical; chlorobenzene and trifluoroethanol have shown superior performance in optimizing yield and selectivity. The reaction mixture is sealed under an inert atmosphere, such as argon or nitrogen, to prevent catalyst deactivation by oxygen, and heated to temperatures between 60°C and 80°C for a duration of 12 to 24 hours. Upon completion, standard workup procedures involving solvent evaporation and silica gel chromatography are employed to isolate the target isoxazolidine. For detailed standardized synthesis steps, please refer to the guide below.
- Prepare the reaction mixture by combining the nitrone substrate and vinyl carbonate derivative in an organic solvent such as chlorobenzene or trifluoroethanol.
- Add the rhodium catalyst precursor [Cp*RhCl2]2 along with the silver salt additive AgSbF6 and a base like Ag2CO3 under an inert atmosphere.
- Heat the sealed reaction vessel to temperatures between 40-80°C for 12-24 hours, then isolate the product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this rhodium-catalyzed technology offers compelling advantages that extend beyond mere chemical elegance. The primary benefit lies in the simplification of the supply chain. By utilizing vinyl carbonates and nitrones, which are often commodity chemicals or easily synthesized from abundant precursors, manufacturers can reduce dependency on exotic or single-source reagents. This diversification of the raw material base enhances supply chain resilience, mitigating the risk of disruptions that can plague the pharmaceutical industry. Additionally, the mild reaction conditions translate directly into energy savings and reduced equipment wear and tear, contributing to lower overhead costs. The high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which simplifies quality control processes and reduces the volume of hazardous waste requiring disposal. These factors collectively drive significant cost reduction in API manufacturing, allowing companies to allocate resources more effectively towards R&D and market expansion.
- Cost Reduction in Manufacturing: The elimination of pre-functionalized alkene partners and the use of low catalyst loadings (typically around 4 mol%) drastically reduce the bill of materials. Furthermore, the high atom economy of the C-H activation process means less waste is generated per kilogram of product, lowering disposal costs and improving the overall environmental footprint of the manufacturing process. The simplified purification workflow also reduces solvent consumption and labor hours associated with chromatography or recrystallization, leading to substantial operational savings.
- Enhanced Supply Chain Reliability: The broad substrate scope of this methodology means that a single platform technology can be used to generate a wide array of derivatives. This flexibility allows supply chain managers to respond quickly to changing demand patterns or to pivot production towards different analogues without needing to retool entire production lines. The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is essential for maintaining long-term contracts with downstream pharmaceutical clients who require stringent consistency in their intermediate supplies.
- Scalability and Environmental Compliance: The protocol's compatibility with standard organic solvents and moderate temperatures makes it inherently scalable from gram to kilogram scales without significant re-optimization. This ease of scale-up accelerates the transition from discovery to commercial production. Moreover, the reduced generation of toxic byproducts and the potential for solvent recycling align with increasingly strict global environmental regulations, ensuring long-term compliance and sustainability for the manufacturing facility.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the implementation and capabilities of this synthesis technology, we have compiled the following answers based on the technical data provided in the patent literature. These insights are designed to clarify the practical aspects of adopting this method for your specific project needs. Whether you are concerned about catalyst recovery, substrate limitations, or downstream processing, understanding these fundamentals is key to successful project execution. The following responses reflect the current state of the art as described in the intellectual property documentation.
Q: What are the primary advantages of this Rh-catalyzed method over traditional isoxazolidine synthesis?
A: This method utilizes readily available nitrone and vinyl carbonate starting materials under mild conditions (40-80°C), avoiding harsh reagents and offering excellent regio- and stereoselectivity compared to classical cycloaddition routes.
Q: Can this synthesis protocol be applied to complex drug-like molecules?
A: Yes, the patent demonstrates successful application on substrates derived from natural products and pharmaceuticals such as Gemfibrozil, Ibuprofen, and Estrone, indicating high functional group tolerance.
Q: What downstream transformations are possible with the resulting isoxazolidine products?
A: The synthesized isoxazolidines serve as versatile intermediates that can be converted into iodides, chlorides, ketones, azides, and pyrrolidine derivatives, facilitating late-stage functionalization for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoxazolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN115215814A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative academic and patent-level chemistry into robust, commercial-scale manufacturing processes. Our team of experienced process chemists is adept at optimizing reaction parameters to ensure maximum yield and purity, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that the transition from benchtop to plant floor involves unique challenges, and our rigorous QC labs and stringent purity specifications guarantee that every batch of isoxazolidine intermediate meets the highest industry standards. We are committed to being a reliable pharmaceutical intermediate supplier that empowers your drug development pipeline with high-quality materials.
We invite you to collaborate with us to explore the full potential of this isoxazolidine synthesis technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project's volume and purity requirements. By partnering with us, you gain access to our deep reservoir of chemical knowledge and our commitment to excellence in service. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with confidence and efficiency.
