Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Manufacturing
Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which confer superior metabolic stability and lipophilicity to drug candidates. Patent CN110467579B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds that addresses many of the historical pain points associated with trifluoromethylation chemistry. This technology leverages a non-metallic iodine-promoted cyclization strategy, utilizing inexpensive starting materials like trifluoroethylimidoyl chloride and hydrazones to generate high-value scaffolds found in numerous bioactive molecules. As illustrated in the structural diversity of known triazole-based drugs, the ability to efficiently access these cores is critical for modern drug discovery pipelines.
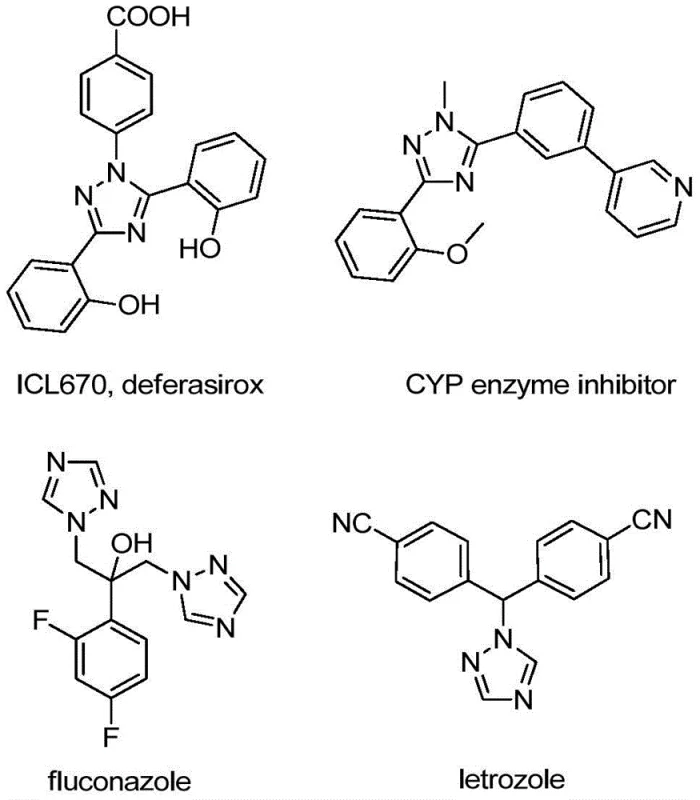
The significance of this patent lies not just in the chemical transformation itself, but in its alignment with green chemistry principles and industrial practicality. By eliminating the need for stringent anhydrous and anaerobic conditions, the process drastically reduces the infrastructure requirements for manufacturing, making it an attractive option for both laboratory-scale optimization and large-scale commercial production. For R&D directors and procurement managers alike, this represents a shift towards more resilient supply chains where complex heterocyclic intermediates can be produced reliably without dependence on exotic or hazardous reagents. The method's versatility allows for the synthesis of variously substituted 1,2,4-triazole compounds, providing a powerful tool for medicinal chemists to explore structure-activity relationships (SAR) around this privileged scaffold.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a trifluoromethyl group into a nitrogen-containing heterocycle has been a formidable challenge in organic synthesis, often plagued by harsh reaction conditions and limited substrate scope. Conventional literature reports typically describe two primary strategies: the direct trifluoromethylation of pre-synthesized nitrogen heterocycles or the cycloaddition of trifluoromethyl-bearing synthons with coupling substrates. The former approach frequently relies on expensive and sometimes unstable trifluoromethylating reagents, which can drive up the cost of goods significantly and introduce safety hazards during handling. Furthermore, these reactions often require transition metal catalysts that necessitate rigorous purification steps to meet the stringent residual metal limits imposed by regulatory bodies for pharmaceutical ingredients. The latter approach, while more direct, has traditionally relied on synthons like trifluorodiazoethane, which is highly explosive and difficult to handle safely on a large scale, thereby limiting its applicability in industrial settings.
The Novel Approach
In stark contrast to these conventional limitations, the methodology described in patent CN110467579B offers a streamlined and safe alternative that utilizes trifluoroethylimidoyl chloride as a stable and accessible trifluoromethyl synthon. This novel approach combines trifluoroethylimidoyl chloride with hydrazones in the presence of sodium acetate and elemental iodine to directly construct the 1,2,4-triazole ring with the trifluoromethyl group already in place. The reaction proceeds smoothly in common organic solvents like dichloroethane (DCE) at moderate temperatures around 80°C, completely bypassing the need for cryogenic conditions or inert atmosphere gloveboxes. This simplicity translates directly into operational efficiency, as the process tolerates a wide range of functional groups and does not require the specialized equipment often mandated for handling sensitive organometallic reagents or hazardous diazo compounds.
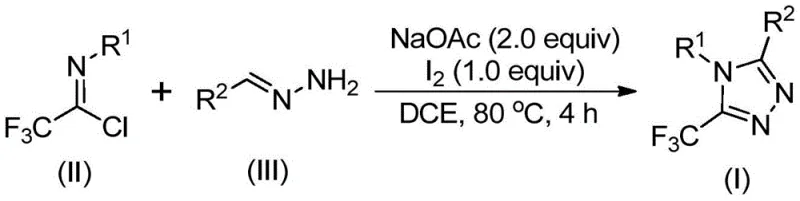
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is a fascinating example of how simple reagents can orchestrate complex bond formations through a cascade of elementary steps. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond formation between the hydrazone and the trifluoroethylimidoyl chloride, generating a trifluoroacetamidine intermediate. This intermediate subsequently undergoes isomerization, setting the stage for the critical oxidative step mediated by elemental iodine. The iodine acts as a mild oxidant, facilitating an oxidative iodination that generates an iodinated intermediate, which is primed for the final ring-closing event. This sequence culminates in an intramolecular electrophilic substitution reaction followed by aromatization, yielding the thermodynamically stable 5-trifluoromethyl substituted 1,2,4-triazole core. Understanding this mechanism is vital for process chemists, as it highlights the dual role of the reagents: sodium acetate serves as a proton scavenger to drive the condensation, while iodine drives the oxidative aromatization without the need for harsh oxidants.
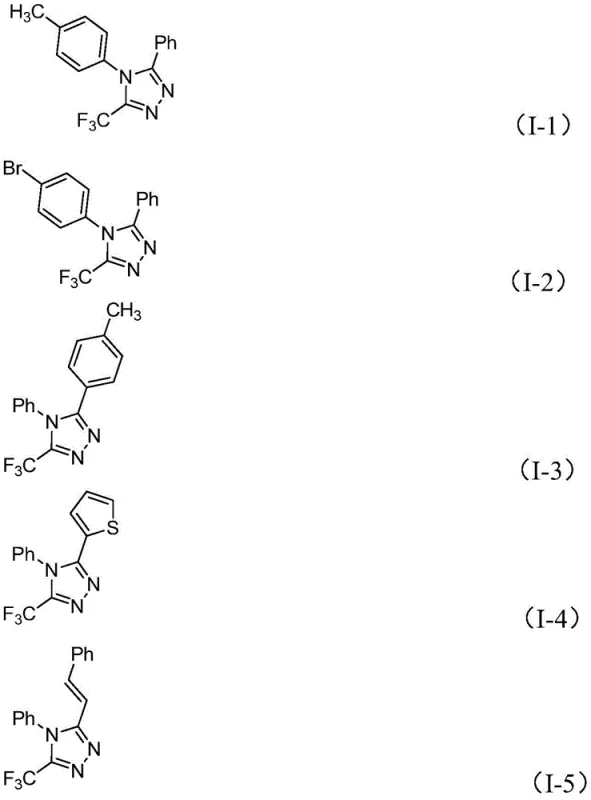
From an impurity control perspective, this mechanism offers distinct advantages over radical-based trifluoromethylation methods which often suffer from poor regioselectivity and the formation of difficult-to-remove byproducts. The stepwise nature of the iodine-promoted cyclization ensures that the trifluoromethyl group is incorporated at the specific 5-position of the triazole ring with high fidelity. The mild reaction conditions further minimize the risk of thermal decomposition or side reactions involving sensitive functional groups on the aryl or heteroaryl substituents. For quality control teams, this means a cleaner crude reaction profile, which simplifies downstream purification and ultimately leads to higher overall yields and purity levels. The ability to tolerate diverse substituents on both the R1 and R2 positions, as demonstrated by the successful synthesis of derivatives bearing electron-donating and electron-withdrawing groups, underscores the robustness of this catalytic system.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is straightforward and relies on standard unit operations familiar to any process chemistry team. The protocol involves charging a reactor with sodium acetate, trifluoroethylimidoyl chloride, and the appropriate hydrazone substrate in a suitable organic solvent such as dichloroethane. The mixture is then heated to approximately 80°C and stirred for a period of 2 to 4 hours to ensure complete conversion to the intermediate species. Following this initial phase, elemental iodine is introduced into the reaction system, and the mixture is maintained at temperature for an additional 1 to 2 hours to drive the oxidative cyclization to completion. The detailed standardized synthesis steps, including precise stoichiometric ratios and workup procedures, are outlined in the guide below to ensure reproducibility and safety.
- Mix sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane (DCE).
- Heat the reaction mixture to 80°C and stir for 2 to 4 hours to allow initial condensation and cyclization.
- Add elemental iodine to the system and continue reacting for 1 to 2 hours to promote oxidative aromatization, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology presents a compelling value proposition centered on cost reduction, supply security, and operational simplicity. By shifting away from expensive and hazardous reagents like trifluorodiazoethane or specialized trifluoromethylating agents, manufacturers can significantly lower their raw material costs and reduce the overhead associated with safety compliance and waste disposal. The use of commodity chemicals such as sodium acetate and elemental iodine, combined with readily available hydrazones and imidoyl chlorides, creates a supply chain that is less vulnerable to the volatility often seen in the market for exotic fine chemicals. Furthermore, the elimination of heavy metal catalysts removes a major bottleneck in the purification process, reducing the consumption of silica gel and solvents required for metal scavenging, which contributes to substantial cost savings in the overall manufacturing budget.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of high-cost reagents with inexpensive, bulk-available starting materials. Since the reaction does not require precious metal catalysts like palladium or copper, there is no need for expensive ligand systems or complex metal recovery processes, which drastically simplifies the bill of materials. Additionally, the mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for specialized Hastelloy equipment often required for corrosive fluorination chemistry, leading to lower capital expenditure and maintenance costs. The high atom economy of the cyclization also means less waste generation, further reducing the environmental fees and disposal costs associated with chemical production.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved because the key building blocks—hydrazones and trifluoroethylimidoyl chlorides—are derived from common aldehydes and amines that are produced on a massive global scale. This abundance ensures that production schedules are not held hostage by the limited availability of niche reagents, a common risk in complex heterocycle synthesis. Moreover, the robustness of the reaction against moisture and oxygen means that storage and handling requirements for intermediates are less stringent, reducing the risk of batch failures due to reagent degradation. This reliability allows for more accurate forecasting and inventory management, ensuring consistent delivery of high-purity intermediates to downstream customers.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram and eventually to tonnage levels is facilitated by the absence of exothermic hazards associated with diazo compounds and the use of standard organic solvents. The simplified workup procedure, which typically involves filtration and column chromatography or recrystallization, is easily adaptable to continuous processing or large-batch operations. From an environmental standpoint, avoiding heavy metals aligns perfectly with increasingly strict global regulations regarding residual impurities in pharmaceuticals, reducing the regulatory burden on the manufacturer. The use of iodine, which can potentially be recovered and recycled, further enhances the sustainability profile of the process, making it an attractive option for companies aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 5-trifluoromethyl-1,2,4-triazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when adopting this route. Understanding these nuances is essential for making informed decisions about process integration and resource allocation.
Q: What are the advantages of this iodine-promoted method over traditional trifluoromethylation?
A: Unlike traditional methods that often require expensive trifluoromethylating reagents or dangerous trifluorodiazoethane, this method utilizes cheap and readily available trifluoroethylimidoyl chloride and hydrazones. It operates under mild conditions without the need for strict anhydrous or anaerobic environments, significantly simplifying the operational complexity.
Q: Does this synthesis route require heavy metal catalysts?
A: No, this novel preparation method completely avoids the use of toxic heavy metal catalysts. Instead, it employs elemental iodine as a promoter and sodium acetate as a base, which reduces the cost of raw materials and eliminates the need for complex heavy metal removal steps during post-processing.
Q: What is the substrate scope for R1 and R2 groups in this triazole synthesis?
A: The method demonstrates excellent functional group tolerance. R1 can be various substituted or unsubstituted aryl groups (e.g., phenyl with methyl, methoxy, bromo, or trifluoromethyl substituents), while R2 can be alkenyl, aryl, or heteroaryl groups, allowing for the design of diverse 1,2,4-triazole derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient heterocycle synthesis plays in accelerating drug development timelines and optimizing production costs. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity 5-trifluoromethyl-1,2,4-triazole intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis for a novel drug candidate or a reliable supply of established intermediates, our infrastructure is designed to support your most demanding projects with speed and precision.
We invite you to collaborate with us to leverage this advanced iodine-promoted synthesis technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates how this specific route can optimize your current supply chain. We encourage you to contact our technical procurement team today to request specific COA data for our triazole portfolio and to discuss route feasibility assessments tailored to your unique molecular targets. Let us help you turn complex chemical challenges into commercial successes.
