Advanced One-Pot Synthesis of Nitrogen-Containing Heterocycles for Commercial Scale Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to streamline the production of complex heterocyclic scaffolds, which serve as the backbone for numerous active pharmaceutical ingredients. Patent CN1022920C presents a groundbreaking advancement in this domain by disclosing a highly efficient, one-pot synthesis process for preparing aromatic substances containing a 5-member heterocyclic ring with nitrogen. This technology specifically targets the production of imidazole and pyrrole derivatives, which are critical precursors in the synthesis of alkaloids such as Pilocarpine. Unlike traditional multi-step sequences that suffer from cumulative yield losses and operational complexity, this novel approach consolidates the reaction into a single container, thereby maximizing atomic economy and process throughput. For R&D directors and procurement specialists, understanding the mechanistic elegance of this patent is crucial, as it offers a pathway to significantly reduce manufacturing costs while enhancing the reliability of the supply chain for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing these valuable heterocyclic compounds, such as those described in EP-OS 0207563, typically involve cumbersome multi-step sequences that are inherently inefficient for large-scale manufacturing. For instance, the preparation of specific imidazole carboxylic acid esters often requires four distinct synthetic steps starting from N-alkyl glycine ester hydrochloride, each step necessitating separate isolation, purification, and quality control procedures. Alternative routes involving diaminomaleonitrile and triethyl orthoformate extend the sequence to five steps, further compounding the issues of material loss and solvent consumption. These conventional pathways are characterized by low overall yields due to the multiplicative effect of individual step efficiencies, and they often generate substantial quantities of chemical waste. Furthermore, the reliance on multiple intermediate isolations increases the risk of contamination and extends the total production lead time, creating bottlenecks that are unacceptable for modern just-in-time supply chains demanding rapid turnaround for clinical and commercial batches.
The Novel Approach
In stark contrast to the fragmented nature of legacy processes, the methodology disclosed in CN1022920C achieves the construction of the target heterocyclic core through a convergent, one-pot reaction strategy. This innovative route involves the direct reaction of a methylene compound, represented by Formula II or its acid salt, with an amidine salt of Formula III in the presence of a base. 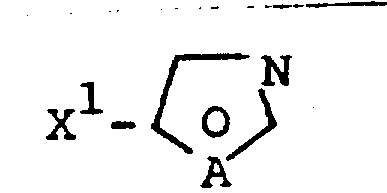 The ability to form the complex Formula I structure directly from simple, commercially available starting materials eliminates the need for isolating unstable or difficult-to-handle intermediates. This consolidation of steps not only drastically simplifies the operational workflow but also enhances the safety profile of the manufacturing process by reducing the number of unit operations. For a reliable pharmaceutical intermediates supplier, adopting this technology means transitioning from a linear, loss-prone production model to a streamlined, high-yield system that is inherently more cost-effective and scalable. The versatility of this approach allows for the facile adjustment of substituents R1 and X1, enabling the rapid generation of diverse compound libraries for drug discovery without compromising on process efficiency or purity standards.
The ability to form the complex Formula I structure directly from simple, commercially available starting materials eliminates the need for isolating unstable or difficult-to-handle intermediates. This consolidation of steps not only drastically simplifies the operational workflow but also enhances the safety profile of the manufacturing process by reducing the number of unit operations. For a reliable pharmaceutical intermediates supplier, adopting this technology means transitioning from a linear, loss-prone production model to a streamlined, high-yield system that is inherently more cost-effective and scalable. The versatility of this approach allows for the facile adjustment of substituents R1 and X1, enabling the rapid generation of diverse compound libraries for drug discovery without compromising on process efficiency or purity standards.
Mechanistic Insights into One-Pot Heterocyclic Cyclization
The success of this synthesis relies on the precise orchestration of nucleophilic attack and cyclization events within a single reaction vessel. The process initiates with the deprotonation of the methylene compound (Formula II) by a suitable base, generating a reactive nucleophilic species that attacks the electrophilic center of the amidine salt (Formula III). 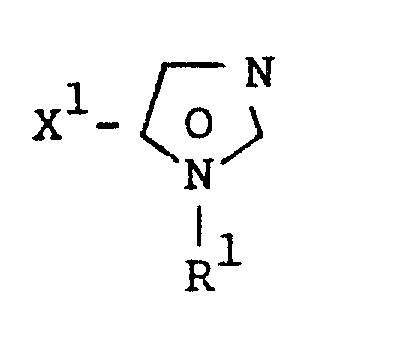 The choice of base is critical and can range from alkali metal hydroxides like sodium hydroxide to alkoxides such as sodium methylate or organic bases like triethylamine, depending on the specific acidity of the methylene precursor. The reaction is typically conducted in inert solvents such as toluene, dioxane, or tetrahydrofuran at temperatures ranging from 20°C to 100°C, ensuring optimal kinetic energy for cyclization without promoting decomposition. A key mechanistic feature is the ability to control the product distribution between esters and amides by manipulating the reaction conditions; for example, the presence of non-enolizable carboxylic esters helps suppress the formation of unwanted acid amide byproducts. This level of control over the reaction trajectory ensures a clean impurity profile, which is paramount for meeting the stringent purity specifications required for API intermediates in regulated markets.
The choice of base is critical and can range from alkali metal hydroxides like sodium hydroxide to alkoxides such as sodium methylate or organic bases like triethylamine, depending on the specific acidity of the methylene precursor. The reaction is typically conducted in inert solvents such as toluene, dioxane, or tetrahydrofuran at temperatures ranging from 20°C to 100°C, ensuring optimal kinetic energy for cyclization without promoting decomposition. A key mechanistic feature is the ability to control the product distribution between esters and amides by manipulating the reaction conditions; for example, the presence of non-enolizable carboxylic esters helps suppress the formation of unwanted acid amide byproducts. This level of control over the reaction trajectory ensures a clean impurity profile, which is paramount for meeting the stringent purity specifications required for API intermediates in regulated markets.
Furthermore, the structural integrity of the final product is maintained through the careful selection of counter-ions and reaction parameters that minimize side reactions. The patent specifies that the anion Y in the amidine salt can be chloride, bromide, or other stable inorganic radicals, with chloride being particularly preferred for its availability and reactivity profile. The reaction tolerates a wide variety of substituents on the nitrogen atom, including alkyl, aryl, and aralkyl groups, demonstrating the robustness of the catalytic system. By avoiding the use of transition metal catalysts, this method sidesteps the complex and costly downstream processing steps associated with heavy metal removal, which is a significant advantage for cost reduction in pharmaceutical manufacturing. The resulting heterocyclic products exhibit high thermal stability and defined melting points, facilitating straightforward purification via crystallization or column chromatography, thus ensuring consistent quality across different production batches.
How to Synthesize 1-Methyl-1H-Imidazole-5-Carboxylic Acid Methyl Ester Efficiently
To implement this technology for the specific production of key intermediates like 1-methyl-1H-imidazole-5-carboxylic acid methyl ester, operators must adhere to strict protocols regarding reagent addition and temperature control. The process begins with the preparation of a suspension of sodium methylate in an inert solvent like toluene under a nitrogen atmosphere to prevent moisture ingress. Subsequently, sarcosine methyl ester hydrochloride and the amidine salt are added sequentially, and the mixture is heated to facilitate the cyclization reaction. The detailed standardized synthesis steps, including exact molar ratios, stirring speeds, and workup procedures, are critical for reproducibility and are outlined below for technical reference.
- Prepare a suspension of alkali metal alcoholate in an inert solvent such as toluene or dioxane under nitrogen protection.
- Add glycine ester hydrochloride and 3-dimethylamino-2-azepine-2-alkene-1-propylidene dimethyl ammonium chloride sequentially to the reaction mixture.
- Stir the mixture at temperatures between 60°C and 90°C for 12 to 24 hours, then isolate the product via filtration and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this one-pot synthesis technology offers profound benefits for procurement managers and supply chain heads tasked with optimizing operational expenditures and ensuring material availability. The elimination of multiple synthetic steps translates directly into a significant reduction in labor costs, solvent consumption, and energy usage, as fewer unit operations are required to reach the final product. Moreover, the use of readily available starting materials such as glycine esters and common amidine salts mitigates the risk of raw material shortages, thereby enhancing supply chain resilience against market volatility. This process efficiency allows manufacturers to offer more competitive pricing structures without compromising on quality, making it an attractive option for long-term supply agreements in the competitive landscape of fine chemical intermediates.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot process fundamentally alters the cost structure of producing nitrogen-containing heterocycles by removing the need for intermediate isolation and purification stages. By consolidating the synthesis into a single reactor run, manufacturers can achieve substantial cost savings through reduced solvent volumes, lower waste disposal fees, and decreased equipment occupancy time. Additionally, the absence of expensive transition metal catalysts eliminates the capital and operational expenses associated with metal scavenging and validation, further driving down the cost of goods sold. These efficiencies enable a more aggressive pricing strategy while maintaining healthy margins, providing a distinct competitive advantage in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and simple reaction conditions ensures a robust and dependable supply chain that is less susceptible to disruptions caused by specialized reagent shortages. Since the starting materials are widely produced and the reaction does not require exotic catalysts or extreme conditions, sourcing risks are minimized, guaranteeing consistent delivery schedules for downstream customers. This reliability is crucial for pharmaceutical companies managing tight production timelines for clinical trials and commercial launches, as it reduces the likelihood of delays caused by raw material bottlenecks. Consequently, partners can plan their inventory levels with greater confidence, knowing that the supply of critical intermediates will remain stable and continuous throughout the contract period.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, which often involves basic filtration and concentration steps, makes this process exceptionally easy to scale from laboratory grams to multi-ton commercial production without significant re-engineering. The reduced generation of chemical waste and the use of recoverable solvents align with increasingly stringent environmental regulations, lowering the compliance burden and potential liability for manufacturing sites. This scalability ensures that the technology can meet growing market demand efficiently, while the greener profile of the process supports corporate sustainability goals. For supply chain leaders, this means securing a future-proof source of materials that can grow with their business needs while adhering to global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this one-pot synthesis method?
A: The primary advantage is the significant reduction in synthetic steps compared to conventional multi-step routes, which leads to higher overall yields and simplified purification processes.
Q: Can this process be scaled for industrial production of Pilocarpine intermediates?
A: Yes, the use of common solvents like toluene and dioxane, along with readily available starting materials, makes this process highly suitable for commercial scale-up.
Q: How does this method impact impurity profiles?
A: By avoiding multiple isolation steps and utilizing specific non-enolizable carboxylic esters, the method effectively suppresses the formation of unwanted acid amide byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthesis technologies like CN1022920C in delivering high-quality intermediates to the global pharmaceutical market. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale manufacturing. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards, guaranteeing the consistency and reliability required for critical drug substance applications.
We invite you to collaborate with our technical procurement team to explore how this efficient synthesis route can optimize your supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this one-pot method for your specific requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in delivering complex heterocyclic building blocks for the next generation of therapeutic agents.
