Advanced Synthesis of Benzo-dioxane Thiazole Derivatives for Commercial Pharmaceutical Applications
Advanced Synthesis of Benzo-dioxane Thiazole Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical industry continuously seeks novel scaffolds that offer enhanced bioavailability and targeted therapeutic effects, and patent CN103724340A presents a significant advancement in this domain by disclosing a class of thiazole derivatives containing benzo-oxygen heterocyclic structures. These compounds are characterized by a unique fusion of a 1,4-benzodioxane or 1,4-benzodioxepine moiety linked via a hydrazone bridge to a substituted thiazole ring, creating a robust molecular architecture with promising pharmacological potential. The structural versatility allows for various substituents on the phenyl ring, enabling fine-tuning of physicochemical properties such as lipophilicity and metabolic stability, which are critical parameters for drug development. This innovation addresses the growing demand for high-purity intermediates that can serve as foundational building blocks for next-generation anti-inflammatory and antiviral agents.
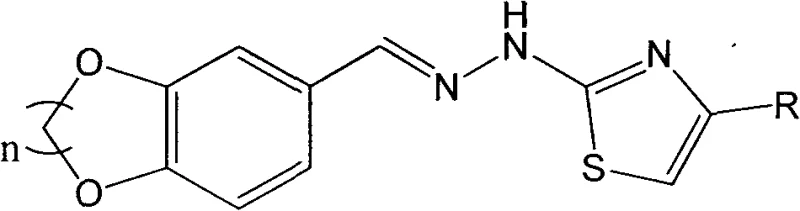
From a strategic sourcing perspective, the availability of such specialized intermediates is vital for R&D teams aiming to explore new chemical space without the burden of developing complex synthetic routes from scratch. The patent outlines a method that not only ensures the structural integrity of the sensitive benzodioxane ring but also facilitates the efficient introduction of the thiazole functionality under relatively mild conditions. For procurement managers and supply chain directors, understanding the underlying chemistry is essential to evaluate the reliability of the supply chain and the potential for cost-effective scale-up. By leveraging this patented technology, manufacturers can secure a consistent supply of high-value intermediates that meet stringent quality specifications required for clinical and commercial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing complex heterocyclic systems often suffer from harsh reaction conditions, the use of toxic heavy metal catalysts, and multi-step sequences that result in poor overall yields and difficult purification processes. Conventional approaches to linking benzodioxane cores with nitrogen-containing heterocycles frequently require protecting group strategies that add unnecessary steps, increase waste generation, and escalate production costs significantly. Furthermore, many existing protocols rely on expensive reagents or anhydrous conditions that are challenging to maintain on a large industrial scale, leading to batch-to-batch variability and supply chain disruptions. The accumulation of impurities from side reactions in these older methods often necessitates extensive chromatographic purification, which is neither economically nor environmentally sustainable for commercial manufacturing.
The Novel Approach
In contrast, the methodology described in CN103724340A offers a streamlined three-step synthesis that avoids the pitfalls of conventional routes by utilizing readily available starting materials and benign reaction conditions. The process employs simple bases like potassium carbonate or sodium hydroxide and common organic solvents such as acetone and ethanol, eliminating the need for exotic catalysts or cryogenic temperatures. This novel approach capitalizes on the nucleophilicity of the catechol hydroxyl groups to form the benzodioxane ring efficiently, followed by a direct condensation and cyclization sequence that builds the thiazole moiety in high yield. The simplicity of the workup procedures, which primarily involve hot filtration and recrystallization, ensures that the final products are obtained with high purity, drastically reducing the downstream processing burden and enhancing the overall economic viability of the manufacturing process.
Mechanistic Insights into the Three-Step Cascade Synthesis
The synthesis begins with a nucleophilic substitution reaction where protocatechualdehyde reacts with a dibromoalkane, such as 1,2-dibromoethane or 1,3-dibromopropane, in the presence of a base catalyst. This step is critical as it constructs the benzo-oxygen heterocyclic core, specifically forming the 1,4-benzodioxane or 1,4-benzodioxepine ring system through an intramolecular etherification mechanism. The reaction is typically conducted at temperatures between 75°C and 85°C, which provides sufficient thermal energy to drive the substitution to completion while minimizing degradation of the aldehyde functionality. The choice of solvent, whether acetonitrile or acetone, plays a pivotal role in solubilizing the reactants and facilitating the interaction between the phenoxide anion and the alkyl halide, ensuring a clean conversion to the aldehyde intermediate.
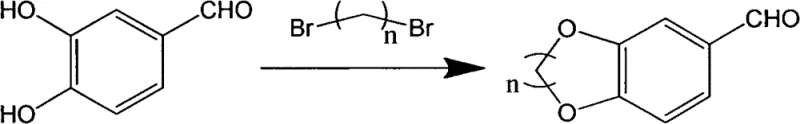
Following the formation of the heterocyclic aldehyde, the second step involves a condensation reaction with thiosemicarbazide to generate a hydrazone intermediate, which serves as the precursor for the final thiazole ring closure. The third and final step is a cyclocondensation reaction between this hydrazone intermediate and a substituted bromoacetophenone, which proceeds through a nucleophilic attack of the sulfur atom on the carbonyl carbon followed by dehydration and aromatization to form the thiazole ring. This cascade effectively links the lipophilic benzodioxane tail with the pharmacologically active thiazole head, creating a molecule with optimized logP values for better membrane permeability. The entire sequence is designed to be telescoped or performed in sequential batches with minimal isolation steps, maximizing throughput and minimizing material loss.
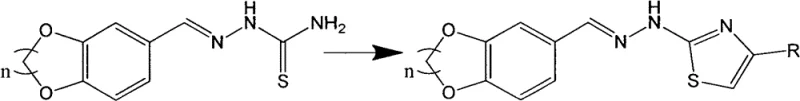
How to Synthesize Benzo-dioxane Thiazole Derivatives Efficiently
To achieve optimal results in the production of these valuable intermediates, strict adherence to the molar ratios and temperature profiles specified in the patent is essential to ensure high purity and yield. The process requires precise control over the stoichiometry, particularly in the first step where a slight excess of the dibromoalkane (molar ratio 1:1.05) is used to drive the etherification to completion without excessive waste. Operators must monitor the reaction progress using thin-layer chromatography (TLC) at each stage to determine the exact endpoint, preventing over-reaction or the formation of byproducts that could complicate purification. While the general procedure is robust, scaling up requires careful attention to heat transfer and mixing efficiency to maintain the uniform reaction conditions described in the laboratory examples.
- React protocatechualdehyde with dibromoalkane (1,2-dibromoethane or 1,3-dibromopropane) in acetone or acetonitrile with a base catalyst at 75-85°C to form the benzo-heterocyclic aldehyde.
- Condense the resulting aldehyde intermediate with thiosemicarbazide in ethanol or isopropanol at 75-85°C to generate the hydrazone precursor.
- Cyclize the hydrazone with substituted bromoacetophenone in ethanol or isopropanol at 75-85°C, followed by recrystallization to obtain the pure thiazole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers substantial strategic benefits by simplifying the raw material portfolio and reducing dependency on scarce or regulated reagents. The reliance on commodity chemicals like protocatechualdehyde, dibromoalkanes, and thiosemicarbazide ensures a stable and resilient supply chain that is less susceptible to market volatility compared to routes requiring specialized organometallic catalysts. Additionally, the use of green solvents such as ethanol and acetone aligns with increasingly stringent environmental regulations, reducing the costs associated with solvent recovery and hazardous waste disposal. This operational efficiency translates directly into a more predictable cost structure and shorter lead times for delivering high-quality intermediates to downstream pharmaceutical customers.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and complex protecting group manipulations significantly lowers the direct material costs associated with production. By utilizing simple inorganic bases and avoiding cryogenic conditions, the process reduces energy consumption and equipment wear, contributing to a lower overall cost of goods sold. The high efficiency of the recrystallization steps means that less material is lost during purification, further enhancing the economic yield of the process and allowing for more competitive pricing strategies in the global market.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this synthesis is straightforward as the key starting materials are bulk chemicals produced by multiple suppliers worldwide, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is not sensitive to minor fluctuations in ambient conditions that might halt more delicate synthetic operations. This reliability ensures that production timelines can be met consistently, providing downstream partners with the confidence needed to plan their own clinical and commercial manufacturing schedules without fear of interruption.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton commercial production without the need for fundamental process redesign. The absence of heavy metals and the use of recyclable solvents simplify the environmental compliance landscape, reducing the regulatory burden and permitting timelines for new manufacturing facilities. This scalability ensures that as demand for these thiazole derivatives grows in the pharmaceutical sector, the supply can be ramped up quickly to meet market needs without compromising on quality or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these benzo-oxygen heterocyclic thiazole derivatives, based on the detailed disclosures within the patent literature. Understanding these aspects helps stakeholders make informed decisions about integrating these intermediates into their drug discovery pipelines or manufacturing workflows. The answers provided reflect the specific experimental conditions and observed benefits documented in the intellectual property, ensuring accuracy and relevance for technical audiences.
Q: What are the primary advantages of the benzo-dioxane thiazole scaffold?
A: This scaffold combines the high lipophilicity and low toxicity of 1,4-benzodioxane with the biological activity of thiazoles, enhancing cell penetration and potential anti-inflammatory effects.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process utilizes common solvents like ethanol and acetone, operates at moderate temperatures (75-85°C), and involves simple filtration and recrystallization steps, making it highly scalable.
Q: What represents the critical quality control point in this synthesis?
A: Monitoring the reaction progress via TLC at each of the three steps is crucial, particularly ensuring complete conversion during the final cyclization to minimize impurities before recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in accelerating drug development timelines and ensuring the success of commercial pharmaceutical launches. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of thiazole derivatives meets the exacting standards required by global regulatory bodies. Our commitment to excellence ensures that our partners receive materials that are not only chemically pure but also consistent in their physical properties, facilitating reproducible results in downstream formulation and testing.
We invite potential partners to engage with our technical procurement team to discuss how our capabilities align with your specific project requirements and to request a Customized Cost-Saving Analysis tailored to your volume needs. By collaborating with us, you gain access to specific COA data and comprehensive route feasibility assessments that can help optimize your supply chain strategy and reduce overall development costs. Whether you require small quantities for preclinical studies or large volumes for commercial API production, NINGBO INNO PHARMCHEM is positioned to be your trusted partner in delivering innovative chemical solutions with speed, reliability, and technical expertise.
