Optimizing Trilaciclib Production: A Novel 4-Step Synthetic Route for Commercial Scale-Up
Introduction to Advanced Trilaciclib Manufacturing
The pharmaceutical landscape for oncology treatments has been significantly transformed by the advent of CDK4/6 inhibitors, with Trilaciclib (also known as Traasiril) standing out as a first-in-class myeloprotective agent. As detailed in patent CN114014863B, a novel preparation method has been developed that addresses critical bottlenecks in the existing supply chain for this high-value active pharmaceutical ingredient. This innovative approach leverages readily available commercial intermediates to construct the complex molecular architecture of Trilaciclib through a concise four-step sequence. By optimizing reaction conditions and streamlining purification protocols, this technology offers a robust pathway for producing high-purity pharmaceutical intermediates. The strategic implementation of halogenation, protective group removal, amide condensation, and methylthio-amino substitution reactions ensures not only chemical efficiency but also operational safety. For global procurement teams and R&D directors, understanding the nuances of this patented route is essential for securing a stable supply of this critical bone marrow protective agent.
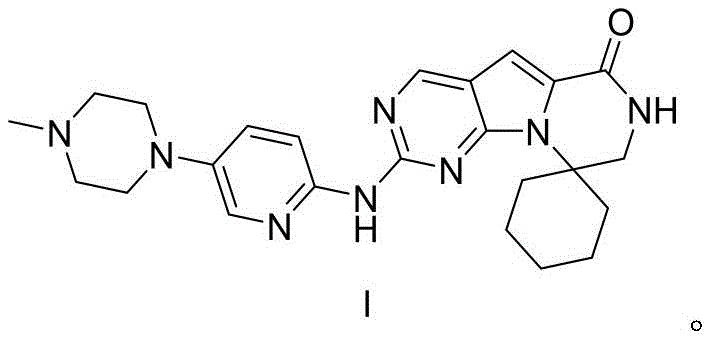
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for complex heterocyclic compounds like Trilaciclib often suffer from excessive step counts and reliance on harsh reaction conditions that compromise overall yield. Conventional methodologies frequently necessitate the use of expensive transition metal catalysts or hazardous reagents that require rigorous removal processes to meet stringent regulatory purity standards. Furthermore, legacy processes often involve multiple protection and deprotection cycles that generate substantial chemical waste and increase the environmental footprint of manufacturing. The reliance on column chromatography for purification in earlier stages of synthesis creates a significant bottleneck for commercial scale-up, driving up both time and operational costs. These inefficiencies result in a fragile supply chain where minor deviations in reaction parameters can lead to batch failures, jeopardizing the continuity of API production. Consequently, there is an urgent industry need for a more streamlined, cost-effective, and environmentally benign synthesis strategy.
The Novel Approach
The patented methodology introduces a paradigm shift by utilizing a convergent synthesis strategy that minimizes step count while maximizing atom economy. As illustrated in the reaction scheme below, the process begins with the alkylation of a commercially accessible pyrazine derivative, followed by a highly selective hydrolysis step that simultaneously removes protecting groups. This is succeeded by an efficient intramolecular amidation to close the central ring system, culminating in a mild oxidative substitution to install the final piperazine moiety. The brilliance of this approach lies in its ability to perform complex transformations under relatively mild thermal conditions, typically ranging from 40°C to 100°C, which reduces energy consumption and equipment stress. Moreover, the protocol emphasizes simple workup procedures such as filtration and solvent pulping, drastically reducing the dependency on resource-intensive chromatographic separations. This holistic optimization ensures that the final product meets high-purity specifications suitable for clinical applications while maintaining economic viability.
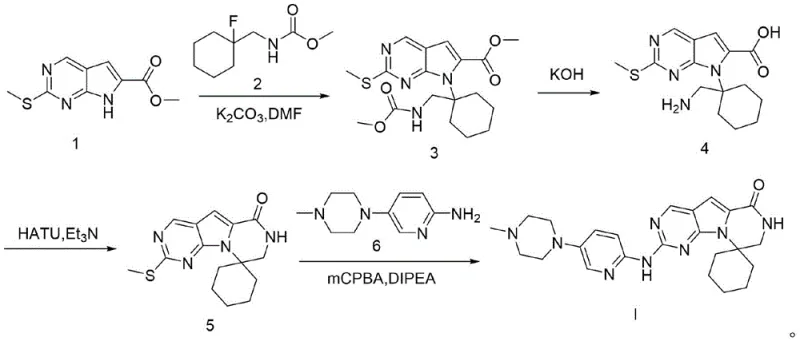
Mechanistic Insights into the 4-Step Cascade Synthesis
The core of this synthetic innovation relies on precise control over nucleophilic substitution and condensation mechanisms to ensure regioselectivity. In the initial step, the reaction between Formula 1 and Formula 2 is mediated by potassium carbonate in DMF at elevated temperatures, facilitating a clean SNAr-type displacement that establishes the critical carbon-nitrogen bond without affecting other sensitive functional groups. The subsequent hydrolysis step employs aqueous potassium hydroxide in ethanol, a condition carefully tuned to cleave ester and protecting groups simultaneously while preserving the integrity of the heterocyclic core. This dual-functionality of the base is crucial for reducing the total number of unit operations required. Following deprotection, the intramolecular cyclization is driven by HATU-mediated amide coupling, which activates the carboxylic acid for nucleophilic attack by the proximal amine, effectively locking the molecular conformation into the desired tricyclic framework. The final step involves the oxidation of a methylthio group to a reactive sulfone or sulfoxide intermediate using mCPBA, which then undergoes displacement by the piperazine derivative. This oxidative activation strategy avoids the need for pre-halogenated intermediates, thereby simplifying the precursor supply chain and reducing halogenated waste.
Impurity control is inherently built into the reaction design through the selection of specific reagents and stoichiometric ratios that favor the formation of the target molecule over potential byproducts. For instance, the use of a slight excess of inorganic base in the first step ensures complete conversion of the starting material, minimizing the carryover of unreacted halides that could complicate downstream purification. The choice of solvent systems, such as the petroleum ether and ethyl acetate mixtures used for pulping, is optimized to selectively precipitate the desired product while keeping soluble impurities in the mother liquor. This physical separation mechanism complements the chemical selectivity of the reactions, resulting in a final product with a superior impurity profile. By avoiding the use of heavy metal catalysts, the process eliminates the risk of metal contamination, a common concern in pharmaceutical manufacturing that often requires additional scavenging steps. The cumulative effect of these mechanistic refinements is a robust process capable of delivering consistent quality across multiple batches.
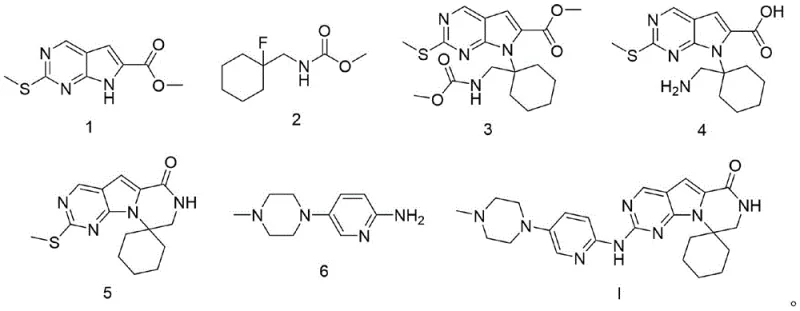
How to Synthesize Trilaciclib Efficiently
Implementing this synthesis route requires careful attention to reaction parameters such as temperature, stirring speed, and addition rates to maximize yield and purity. The process is designed to be scalable, utilizing standard reactor configurations and commonly available solvents that simplify technology transfer from laboratory to plant scale. Operators should focus on maintaining strict control over the exothermic nature of the base additions and ensuring adequate mixing during the precipitation phases to achieve uniform particle size distribution. The following guide outlines the critical operational milestones derived from the patent examples, providing a framework for establishing a reliable manufacturing protocol. Detailed standardized synthesis steps are provided in the section below to assist technical teams in replicating these results.
- Perform alkylation of Formula 1 with Formula 2 using K2CO3 in DMF at 100°C to obtain Formula 3.
- Execute deprotection and hydrolysis of Formula 3 using KOH aqueous solution in EtOH at 45°C to yield Formula 4.
- Conduct intramolecular amidation of Formula 4 using HATU and triethylamine in DMF at 50°C to form Formula 5.
- Finalize synthesis via oxidative substitution of Formula 5 with Formula 6 using mCPBA and DIPEA in toluene at room temperature.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this novel synthesis route offers transformative benefits that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. By shifting away from complex multi-step sequences that rely on custom-synthesized building blocks, the new method leverages commodity chemicals that are readily available in the global market. This strategic sourcing advantage mitigates the risk of raw material shortages and price volatility, ensuring a more predictable cost structure for long-term production planning. The elimination of expensive transition metal catalysts and the reduction in chromatographic purification steps translate into substantial operational savings, as these are typically the most cost-intensive components of fine chemical manufacturing. Furthermore, the simplified workflow reduces the overall cycle time per batch, allowing facilities to increase throughput without requiring significant capital investment in new infrastructure. These efficiencies collectively enhance the competitiveness of the supply chain, enabling faster response times to market demand fluctuations.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by replacing high-cost purification techniques with economical slurry washing methods. Traditional routes often depend on repeated silica gel column chromatography, which consumes vast quantities of solvents and adsorbents while generating hazardous waste; this new approach largely circumvents those expenses. Additionally, the use of inexpensive inorganic bases like potassium carbonate and potassium hydroxide instead of specialized organic bases or organometallic reagents further drives down the bill of materials. The high yield observed in each step means that less raw material is wasted, improving the overall mass balance and reducing the cost per kilogram of the final API. These factors combine to create a leaner, more cost-effective production model that maximizes margin potential.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved because the starting materials, specifically Formula 1 and Formula 2, are established commercial intermediates with mature supply chains. Unlike proprietary precursors that may be sourced from a single vendor, these commoditized inputs can be procured from multiple qualified suppliers, reducing the risk of supply disruption. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain inconsistencies. This reliability is critical for maintaining continuous production schedules and meeting strict delivery commitments to downstream pharmaceutical partners. Ultimately, a more resilient supply chain translates to greater trust and stronger partnerships with key stakeholders in the healthcare sector.
- Scalability and Environmental Compliance: The synthesis is inherently designed for scale-up, utilizing solvents and conditions that are compatible with large-scale industrial reactors. The avoidance of cryogenic temperatures and high-pressure hydrogenation simplifies the engineering requirements, making it easier to transfer the process from pilot plants to multi-ton production facilities. From an environmental standpoint, the reduction in solvent usage and the minimization of hazardous waste generation align with green chemistry principles and increasingly stringent regulatory standards. The simplified workup procedures reduce the load on wastewater treatment systems and lower the carbon footprint associated with solvent recovery and disposal. This alignment with sustainability goals not only ensures regulatory compliance but also enhances the corporate social responsibility profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Trilaciclib synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios. The answers reflect a commitment to transparency and technical accuracy, ensuring that decision-makers have the information needed to proceed with confidence.
Q: What are the key advantages of this novel Trilaciclib synthesis route?
A: The process utilizes commercially available starting materials and features mild reaction conditions (40-100°C). It significantly reduces purification costs by replacing complex column chromatography with simple slurry purification in most steps, leading to higher overall yields and industrial operability.
Q: How does this method improve impurity control compared to conventional routes?
A: The invention ensures single-site reactivity at each step, minimizing side reactions. The use of specific inorganic bases like K2CO3 and KOH allows for selective deprotection and cyclization, effectively controlling the impurity profile without requiring extensive downstream processing.
Q: Is this process suitable for large-scale manufacturing of CDK4/6 inhibitors?
A: Yes, the route is designed for scalability. It avoids hazardous reagents and extreme conditions, utilizing standard solvents like DMF, EtOH, and Toluene. The simplified workup procedures, such as filtration and pulping, facilitate easy scale-up from pilot to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trilaciclib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the production of complex oncology intermediates like Trilaciclib. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch meets the highest industry standards. Our commitment to quality is backed by a deep understanding of the chemical nuances involved in synthesizing CDK4/6 inhibitors, allowing us to troubleshoot and optimize processes effectively. By choosing us as your partner, you gain access to a supply chain that is both robust and responsive to the dynamic needs of the pharmaceutical market.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project requirements. We are prepared to provide a Customized Cost-Saving Analysis that quantifies the potential economic advantages of switching to this more efficient manufacturing method. Please contact us to request specific COA data and route feasibility assessments tailored to your development timeline. Our goal is to collaborate closely with you to accelerate your drug development programs while optimizing your overall production costs. Let us help you secure a sustainable and high-quality supply of this vital therapeutic intermediate.
