Scalable Synthesis of 2-Trifluoromethyl Quinazolinones via Palladium-Catalyzed Carbonylation for Pharma Intermediates
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones via Palladium-Catalyzed Carbonylation for Pharma Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Among these, quinazolinone derivatives stand out due to their profound biological activities, ranging from antifungal and antiviral properties to potent anticancer effects. A recent technological breakthrough, documented in patent CN112480015B, introduces a highly efficient multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. This innovation addresses long-standing challenges in heterocyclic chemistry by leveraging a palladium-catalyzed carbonylation cascade. For R&D directors and procurement specialists, this methodology represents a significant leap forward, offering a pathway to high-purity pharmaceutical intermediates with improved cost-efficiency and operational simplicity. The integration of the trifluoromethyl group is particularly strategic, as it enhances the metabolic stability and lipophilicity of the final drug candidates, making this synthesis route highly relevant for modern medicinal chemistry programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied on methodologies that impose severe constraints on both laboratory research and industrial manufacturing. Traditional approaches often necessitate the use of high-pressure carbon monoxide gas, which requires specialized autoclaves and rigorous safety protocols, thereby inflating capital expenditure and operational risks. Furthermore, many established routes depend on expensive and less accessible starting materials, such as 2-bromoformylaniline or pre-activated acid anhydrides, which drive up the cost of goods sold (COGS). Other methods involving ruthenium or platinum catalysts frequently suffer from narrow substrate scope and moderate yields, limiting their utility in diverse drug discovery campaigns. The need for harsh reaction conditions and complex multi-step sequences not only prolongs the development timeline but also generates significant chemical waste, posing challenges for environmental compliance and sustainability goals in fine chemical manufacturing.
The Novel Approach
In stark contrast, the method disclosed in patent CN112480015B utilizes a streamlined one-pot strategy that dramatically simplifies the synthetic workflow. By employing cheap and readily available nitro compounds alongside trifluoroethylimidoyl chloride, this process bypasses the need for pre-activated substrates. The reaction operates under moderate thermal conditions (120°C) using solid molybdenum hexacarbonyl as a safe carbon monoxide source, effectively eliminating the hazards associated with high-pressure gas handling. As illustrated in the general reaction scheme below, the transformation is catalyzed by a palladium system that demonstrates exceptional functional group tolerance.
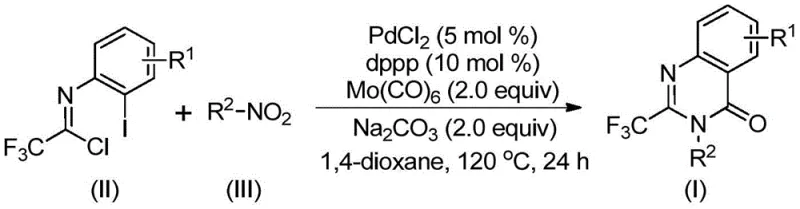
This novel approach not only achieves high conversion rates but also accommodates a wide variety of substituents on the aromatic rings, enabling the rapid generation of diverse chemical libraries. The operational simplicity of mixing reagents in a common organic solvent like dioxane makes this protocol highly attractive for reliable pharmaceutical intermediate supplier operations seeking to optimize their production pipelines.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to replicate or modify the process for specific analogs. The reaction is believed to initiate with the reduction of the nitro group to an amine by molybdenum hexacarbonyl, which simultaneously serves as the carbonyl source. This in situ generated amine then undergoes a base-promoted intermolecular coupling with the trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the imidoyl chloride derivative, forming a key divalent palladium species. As heating proceeds, carbon monoxide is released from the molybdenum complex and inserts into the carbon-palladium bond, generating an acyl-palladium intermediate.
The cyclization is driven by the formation of a palladium-nitrogen bond, creating a seven-membered cyclic palladium intermediate that eventually undergoes reductive elimination to yield the target 2-trifluoromethyl quinazolinone. This intricate cascade is meticulously balanced to minimize side reactions, ensuring a clean impurity profile. The choice of ligands, specifically 1,3-bis(diphenylphosphino)propane (dppp), plays a pivotal role in stabilizing the active catalytic species and facilitating the reductive elimination step. For quality control teams, this mechanistic clarity allows for better prediction of potential byproducts and the implementation of targeted purification strategies, ensuring that the final high-purity OLED material or API intermediate meets stringent regulatory specifications.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize yield and purity. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, and carbonyl source before introducing the organic substrates. The specific stoichiometry and solvent choice are critical; for instance, using dioxane as the solvent has been shown to provide superior conversion rates compared to other aprotic solvents. The reaction mixture is then heated to maintain a steady temperature of 120°C for a duration of 16 to 30 hours, allowing the multi-component cascade to reach completion. Detailed standardized operating procedures for scaling this reaction are essential for maintaining batch-to-batch consistency.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours to allow the carbonylation cascade to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical elegance. The shift from high-pressure gas systems to solid reagents fundamentally alters the risk profile and infrastructure requirements of the manufacturing facility. By utilizing commodity chemicals like nitro compounds and avoiding exotic catalysts, companies can secure a more resilient supply chain that is less susceptible to market volatility. The simplified workup procedure, involving basic filtration and chromatography, reduces the demand for specialized downstream processing equipment, thereby lowering the overall barrier to entry for commercial production.
- Cost Reduction in Manufacturing: The economic impact of this method is driven primarily by the substitution of expensive, pre-activated starting materials with inexpensive nitro compounds. Additionally, the elimination of high-pressure carbon monoxide equipment removes a significant capital expenditure burden, allowing for production in standard glass-lined reactors. The high atom economy and reduced need for complex purification steps further contribute to substantial cost savings in API manufacturing, making the final product more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Sourcing reliability is paramount for continuous manufacturing operations. Since the key reagents, including the nitro compounds and the palladium catalyst system, are widely available from multiple global vendors, the risk of supply disruption is minimized. The robustness of the reaction conditions means that slight variations in raw material quality are less likely to cause batch failures, ensuring a consistent flow of materials to downstream formulation teams and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The transition from bench-scale to commercial scale is facilitated by the use of standard organic solvents and moderate temperatures. The process generates less hazardous waste compared to traditional methods involving heavy metal catalysts or toxic gases, aligning with modern green chemistry principles. This environmental compatibility simplifies regulatory approval processes and waste disposal logistics, supporting the commercial scale-up of complex polymer additives or drug substances without compromising on sustainability metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what partners can expect when integrating this chemistry into their portfolio. Understanding these nuances helps stakeholders make informed decisions about process adoption and resource allocation.
Q: What are the advantages of using nitro compounds over traditional aniline derivatives in this synthesis?
A: Nitro compounds are significantly cheaper and more readily available than pre-activated substrates like 2-bromoformylaniline. Furthermore, this method avoids the need for harsh pre-activation steps, streamlining the supply chain and reducing raw material costs.
Q: Does this process require high-pressure carbon monoxide equipment?
A: No, the process utilizes solid molybdenum hexacarbonyl (Mo(CO)6) as a safe and convenient carbon monoxide surrogate. This eliminates the safety hazards and capital expenditure associated with high-pressure gas cylinders and specialized autoclaves.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability to the gram level with high reaction efficiency and broad substrate compatibility. The use of standard organic solvents and moderate temperatures (120°C) facilitates easy translation to kilogram and ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in accelerating drug development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-trifluoromethyl quinazolinone performs consistently in your downstream applications. Our infrastructure is designed to handle complex heterocyclic syntheses with the utmost precision and safety.
We invite you to collaborate with us to leverage this advanced palladium-catalyzed technology for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive pharmaceutical landscape.
