Scalable Metal-Free Synthesis of Bioactive Trifluoromethyl Triazole Intermediates
Scalable Metal-Free Synthesis of Bioactive Trifluoromethyl Triazole Intermediates
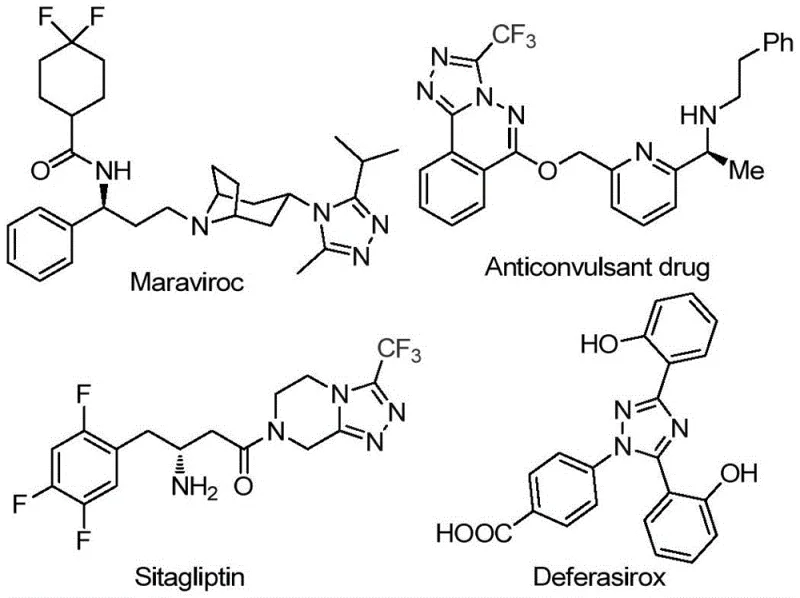
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are pivotal for enhancing metabolic stability and bioavailability. Patent CN113105402A discloses a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, a structural scaffold prevalent in numerous high-value therapeutic agents. As illustrated in the provided chemical landscape, molecules such as Maraviroc, Sitagliptin, and Deferasirox rely on similar heterocyclic cores to exert their biological functions, highlighting the critical demand for efficient synthetic routes to these intermediates. This novel approach leverages a metal-free, iodine-promoted strategy that circumvents the limitations of traditional transition-metal catalysis, offering a streamlined pathway for the production of complex pharmaceutical building blocks.
The significance of this technology extends beyond mere academic interest; it addresses fundamental supply chain vulnerabilities associated with the procurement of specialized catalysts and the management of heavy metal residues. By utilizing inexpensive and commercially available starting materials such as arylethanones and trifluoroacetimidoyl hydrazides, the process ensures a reliable supply of high-purity pharmaceutical intermediates. The reaction system is designed to be operationally simple, avoiding the stringent requirements for inert atmospheres or ultra-dry solvents that often plague large-scale manufacturing. This accessibility makes the technology particularly attractive for contract development and manufacturing organizations (CDMOs) aiming to reduce lead times for high-purity heterocyclic intermediates while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles has relied heavily on transition metal catalysts or harsh reaction conditions that pose significant challenges for industrial scale-up. Traditional routes often necessitate the use of expensive palladium, copper, or rhodium complexes, which not only inflate the raw material costs but also introduce complex purification burdens to meet stringent residual metal specifications required by regulatory bodies. Furthermore, many existing protocols demand strictly anhydrous and anaerobic environments, requiring specialized equipment such as gloveboxes or Schlenk lines, which drastically increases capital expenditure and operational overhead. The sensitivity of these conventional methods to moisture and oxygen often leads to inconsistent batch-to-batch reproducibility, creating bottlenecks in the supply chain for critical API intermediates. Additionally, the generation of toxic heavy metal waste streams necessitates elaborate treatment processes, conflicting with modern green chemistry principles and environmental compliance mandates.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113105402A introduces a paradigm shift by employing elemental iodine and dimethyl sulfoxide (DMSO) as the primary reaction drivers. This metal-free system effectively replaces costly transition metals with abundant and inexpensive iodine, fundamentally altering the cost structure of the synthesis. The reaction proceeds through a tandem sequence where arylethanones undergo iodine-mediated oxidation to form reactive alpha-dicarbonyl species, which subsequently condense with trifluoroacetimidoyl hydrazides to close the triazole ring. This approach eliminates the need for exotic ligands or sensitive organometallic reagents, thereby simplifying the logistical framework for procurement managers. The robustness of the system allows it to tolerate ambient atmospheric conditions, removing the necessity for expensive inert gas purging and drying protocols. Consequently, this novel route offers a direct path to cost reduction in pharmaceutical intermediate manufacturing by minimizing both reagent expenses and waste disposal liabilities.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The core of this synthetic innovation lies in the dual role of iodine within the dimethyl sulfoxide solvent system, facilitating a cascade of oxidative and cyclization events. Initially, the arylethanone substrate undergoes a Kornblum-type oxidation mediated by the iodine-DMSO complex, transforming the methyl ketone into an alpha-dicarbonyl intermediate, specifically an aryl glyoxal equivalent. This oxidation step is critical as it generates the electrophilic center necessary for the subsequent nucleophilic attack. Following this activation, the trifluoroacetimidoyl hydrazide acts as a bidentate nucleophile, condensing with the newly formed dicarbonyl species to establish the initial hydrazone linkage. The presence of base, specifically sodium dihydrogen phosphate and pyridine, facilitates the deprotonation steps required to drive the equilibrium towards the cyclized product. The final ring closure is promoted by the continued presence of iodine, which likely assists in the oxidative aromatization of the dihydro-triazole intermediate to yield the stable 3,4,5-trisubstituted 1,2,4-triazole core.
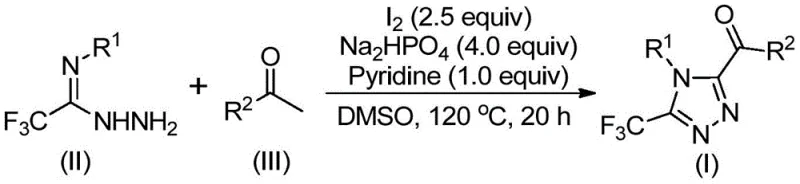
Understanding the mechanistic nuances is vital for R&D directors focused on impurity control and process optimization. The stepwise nature of the reaction, involving a distinct oxidation phase followed by a cyclization phase, allows for precise control over reaction kinetics. By maintaining the initial temperature at 90-110°C, the formation of the alpha-dicarbonyl species is maximized before the introduction of the hydrazide component. Subsequent heating to 110-130°C ensures complete cyclization and aromatization. This thermal staging minimizes the formation of side products such as unreacted hydrazones or over-oxidized byproducts. The use of pyridine as an additive further stabilizes the reaction environment, scavenging acidic byproducts like hydrogen iodide that could otherwise degrade the sensitive trifluoromethyl group. This mechanistic clarity ensures that the process can be reliably transferred from laboratory benchtop to commercial production scales with predictable impurity profiles.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for maximum efficiency and ease of handling, making it an ideal candidate for rapid process development. The protocol involves a straightforward one-pot procedure where reagents are added sequentially, eliminating the need for intermediate isolation of the reactive alpha-dicarbonyl species. This telescoped approach not only saves time but also reduces solvent consumption and material loss associated with multiple workup steps. The reaction utilizes standard laboratory glassware and does not require specialized high-pressure reactors or cryogenic cooling systems. For detailed operational parameters regarding stoichiometry, specific temperature ramps, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the optimal conditions derived from the patent data.
- Oxidation Phase: Mix arylethanone and iodine in DMSO, heating to 90-110°C for 4-6 hours to generate the reactive alpha-dicarbonyl intermediate via Kornblum oxidation.
- Cyclization Phase: Add trifluoroacetimidoyl hydrazide, sodium dihydrogen phosphate, pyridine, and additional iodine, then heat to 110-130°C for 12-20 hours to complete the heterocyclic ring formation.
- Purification: Filter the reaction mixture, adsorb onto silica gel, and purify via column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this iodine-promoted synthesis offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The elimination of precious metal catalysts represents a significant opportunity for cost reduction in API manufacturing, as it removes the volatility associated with the pricing of palladium and other rare earth elements. Furthermore, the absence of heavy metals simplifies the downstream purification process, potentially reducing the number of chromatography columns or crystallization steps required to meet pharmacopeial standards. This streamlining of the purification train translates to higher overall throughput and reduced cycle times, enhancing the agility of the supply chain. The reliance on commodity chemicals like iodine, DMSO, and acetophenones ensures a stable and diversified supply base, mitigating the risk of raw material shortages that can disrupt production schedules.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with elemental iodine drastically lowers the direct material costs associated with the synthesis. Since iodine is a bulk commodity chemical with a stable global supply, manufacturers are insulated from the price fluctuations typical of specialized organometallic catalysts. Additionally, the simplified workup procedure, which avoids complex metal scavenging resins, reduces the consumption of auxiliary materials and lowers waste disposal costs. The high atom economy of the tandem reaction further contributes to economic efficiency by maximizing the conversion of starting materials into the desired product, minimizing the financial loss associated with unreacted feedstock.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which do not require strict exclusion of air or moisture, significantly enhances operational reliability. This tolerance allows for the use of standard industrial reactors without the need for extensive inertization protocols, reducing the risk of batch failures due to equipment leaks or operator error. The wide availability of the starting materials, including various substituted arylethanones and hydrazides, ensures that production can be scaled up rapidly to meet surging demand without long lead times for custom reagent synthesis. This flexibility is crucial for maintaining continuity of supply in the fast-paced pharmaceutical market where time-to-market is a critical competitive factor.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to proceed efficiently from milligram to gram scales with consistent yields, indicating a clear path to kilogram and tonne-level production. The use of DMSO, a high-boiling polar aprotic solvent, facilitates heat transfer and mixing in large vessels, while the absence of toxic heavy metals aligns with increasingly stringent environmental regulations regarding effluent discharge. The simplified waste stream, primarily consisting of organic salts and iodine residues, is easier to treat and dispose of compared to heavy metal-contaminated waste, thereby reducing the environmental footprint and compliance burden for manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations. By addressing these key areas, we aim to facilitate informed decision-making for technical teams evaluating this route for their specific project needs.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the process described in patent CN113105402A utilizes elemental iodine as a non-metallic promoter, completely eliminating the need for costly palladium or copper catalysts and simplifying downstream purification.
Q: What is the substrate tolerance for this triazole synthesis?
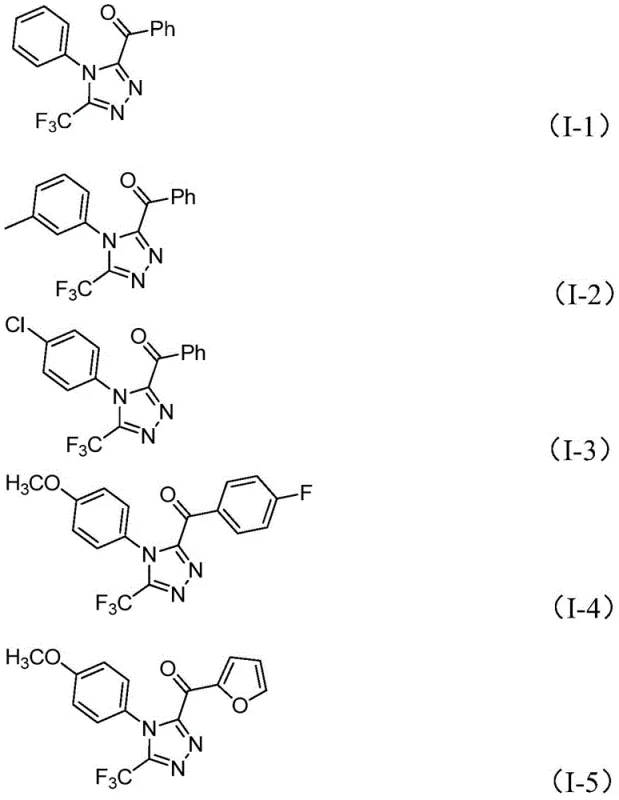
A: The method demonstrates excellent functional group tolerance, successfully accommodating various substituents such as methyl, methoxy, chloro, and trifluoromethyl groups on both the arylethanone and hydrazide components.
Q: Are strict anhydrous or anaerobic conditions required?
A: Unlike many traditional heterocyclic syntheses, this protocol operates efficiently without the need for rigorous anhydrous or oxygen-free environments, significantly reducing operational complexity and equipment costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the production of next-generation pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this iodine-promoted process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We are committed to leveraging this innovative chemistry to deliver high-quality trifluoromethyl triazole derivatives that accelerate your drug development timelines.
We invite you to collaborate with us to explore the full commercial viability of this synthetic route for your specific pipeline candidates. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this metal-free approach can optimize your budget. Please contact our technical procurement team today to request specific COA data for our library of triazole intermediates and to discuss detailed route feasibility assessments for your custom synthesis projects.
