Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Drug Discovery
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Drug Discovery
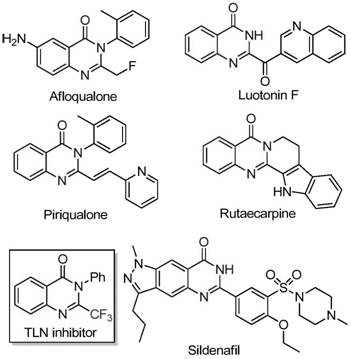
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles, particularly quinazolinones, due to their pervasive presence in bioactive molecules ranging from anti-cancer agents to antifungal drugs. A significant technological breakthrough in this domain is detailed in patent CN111675662B, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone compounds. This innovation addresses critical bottlenecks in modern medicinal chemistry by utilizing a cheap iron catalyst to facilitate the cyclization of readily available starting materials. The introduction of the trifluoromethyl group is strategically vital, as it enhances the electronegativity, metabolic stability, and lipophilicity of the target scaffold, thereby improving overall bioavailability. By leveraging this novel methodology, manufacturers can access complex molecular architectures that were previously difficult or prohibitively expensive to synthesize using conventional transition metal catalysis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functional groups has relied heavily on the cyclization of synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate with substrates like anthranilamide or isatoic anhydride. While these methods are chemically valid, they suffer from severe practical limitations that hinder large-scale adoption in commercial manufacturing. The reaction conditions are often excessively harsh, requiring stringent temperature controls or hazardous reagents that pose safety risks in an industrial setting. Furthermore, the substrate scope is frequently narrow, limiting the structural diversity accessible to medicinal chemists during lead optimization phases. Perhaps most critically for procurement teams, the reliance on specialized fluorinated synthons drives up the raw material costs significantly, making the final active pharmaceutical ingredients (APIs) less competitive in the global market.
The Novel Approach
In stark contrast to legacy techniques, the method described in patent CN111675662B utilizes trifluoroethylimidoyl chloride and isatin as the primary building blocks, catalyzed by inexpensive ferric chloride. This approach represents a paradigm shift towards green and sustainable chemistry by replacing precious metals with earth-abundant iron. The reaction proceeds through a tandem sequence involving alkali-promoted carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization. This mechanistic pathway not only simplifies the operational procedure but also dramatically improves functional group tolerance, allowing for the incorporation of diverse substituents such as halogens, alkyls, and nitro groups without protecting group strategies. The result is a versatile platform technology capable of generating high-purity intermediates suitable for downstream drug development.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this synthetic innovation lies in the unique reactivity of the iron catalyst in promoting decarbonylative cyclization. As illustrated in the reaction scheme below, the process initiates with the interaction between trifluoroethylimidoyl chloride and isatin in the presence of sodium hydride. The base facilitates the initial nucleophilic attack, forming a transient trifluoroacetamidine intermediate. Subsequently, the ferric chloride catalyst activates the system for a crucial decarbonylation step, effectively removing the carbonyl oxygen from the isatin moiety while simultaneously closing the quinazolinone ring. This dual functionality of the iron species ensures high atom economy and minimizes the formation of unwanted byproducts that typically plague multi-step heterocycle syntheses.
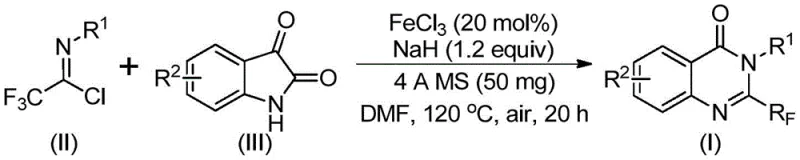
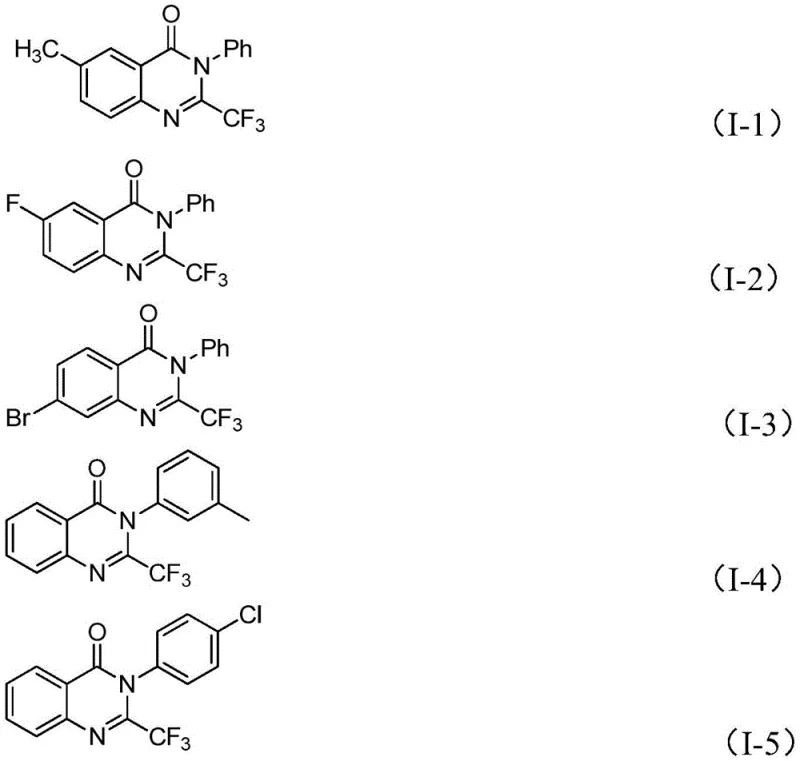
From an impurity control perspective, this mechanism offers distinct advantages for R&D directors focused on purity profiles. The use of 4A molecular sieves in the reaction mixture plays a pivotal role in scavenging moisture, which is critical for maintaining the activity of the sodium hydride and preventing hydrolysis of the sensitive imidoyl chloride starting material. By strictly controlling the water content and utilizing a mild oxidative environment (air), the reaction selectively favors the formation of the desired 2-trifluoromethyl quinazolinone over potential hydrolysis products or polymerization side-reactions. The broad substrate scope demonstrated in the patent, covering various substituted aryl groups at both the R1 and R2 positions, confirms the robustness of this catalytic cycle against electronic and steric variations, ensuring consistent quality across different analogues.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and safety. The protocol involves a two-stage temperature profile, starting with a lower temperature phase to establish the initial intermediates, followed by a higher temperature phase to drive the cyclization to completion. Detailed standard operating procedures regarding reagent addition rates, stirring speeds, and work-up protocols are essential for reproducibility. For a comprehensive breakdown of the exact molar ratios, solvent volumes, and purification steps validated in the patent examples, please refer to the standardized synthesis guide provided below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C for 18-20 hours under air.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iron-catalyzed methodology presents a compelling value proposition centered on cost efficiency and supply security. The replacement of expensive palladium or rhodium catalysts with ferric chloride results in a drastic reduction in catalyst costs, which directly impacts the bottom line of the manufacturing budget. Moreover, the starting materials—isatin and trifluoroethylimidoyl chloride—are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. This abundance ensures that production schedules remain uninterrupted even during periods of raw material volatility, providing a stable foundation for long-term supply agreements with pharmaceutical partners.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes the need for costly metal scavenging steps and complex purification processes required to meet strict residual metal limits in APIs. This simplification of the downstream processing workflow leads to substantial cost savings in both labor and consumables. Additionally, the high conversion rates observed in DMF solvent reduce the volume of waste generated per kilogram of product, further lowering disposal costs and enhancing the overall economic viability of the process for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By utilizing widely available feedstocks like isatin and simple aromatic amines, the supply chain becomes inherently more resilient. Unlike specialized fluorinated reagents that may have limited production capacity, the precursors for this reaction are produced on a massive scale for various industries. This accessibility allows for flexible sourcing strategies and reduces lead times for high-purity pharmaceutical intermediates. Manufacturers can maintain healthy inventory levels of raw materials without tying up excessive capital, ensuring rapid response to sudden increases in demand from clinical trial phases.
- Scalability and Environmental Compliance: The reaction conditions described, operating at moderate temperatures and under air, are inherently safer and easier to scale than high-pressure or inert atmosphere reactions. The use of DMF, while requiring careful handling, is a well-understood solvent in the industry with established recovery and recycling protocols. The simplicity of the work-up, involving filtration and standard column chromatography, facilitates a smooth transition from gram-scale discovery to multi-kilogram production. This ease of scale-up supports the rapid progression of drug candidates through the pipeline while adhering to increasingly stringent environmental regulations regarding heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of the method for industrial partners.
Q: What are the advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: FeCl3 is significantly cheaper and more abundant than precious metal catalysts, offering a cost-effective solution without compromising reaction efficiency or yield.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent explicitly states the method is expandable to gram levels and provides possibilities for industrial scale application due to simple operation and cheap raw materials.
Q: What is the substrate scope for R1 and R2 groups in this reaction?
A: The method tolerates a wide range of substituents including alkyl, halogen, methoxy, nitro, and various aryl groups at ortho-, meta-, or para-positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes in accelerating drug discovery timelines. Our team of expert chemists has extensively evaluated the iron-catalyzed cyclization method described in CN111675662B and is fully prepared to support your development programs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from benchtop to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cost-effective technology for your next-generation therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall project costs.
