Advanced Biomimetic Transamination for Scalable Alpha-Amino Acid Ester Production
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for greener, more efficient, and cost-effective synthetic routes. A pivotal advancement in this domain is detailed in Chinese Patent CN102675135A, which discloses a highly efficient method for synthesizing alpha-amino acid esters. This technology represents a significant departure from traditional methodologies by employing a biomimetic transamination strategy that utilizes alpha-ketoesters and benzylamine as primary raw materials, with triethylamine serving as a robust organic catalyst. The process is designed as a one-pot reaction that simplifies operational complexity while achieving impressive yields of up to 95%. For R&D directors and procurement specialists alike, this patent offers a compelling alternative to metal-catalyzed hydrogenation, addressing critical pain points regarding impurity profiles, equipment safety, and overall process economics in the production of high-value amino acid derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of alpha-amino acids has heavily relied on the catalytic hydrogenation of imines. While effective in laboratory settings, this conventional approach introduces significant challenges when scaled for commercial manufacturing. The primary drawback is the necessity for transition metal catalysts and high-pressure hydrogen gas, which creates severe safety hazards and necessitates specialized, high-cost reactor infrastructure. Furthermore, the presence of residual heavy metals in the final product is a major regulatory concern for pharmaceutical applications, often requiring additional, costly purification steps such as scavenging or recrystallization to meet stringent purity specifications. These factors collectively inflate the cost of goods sold (COGS) and extend lead times, making traditional hydrogenation less attractive for large-scale supply chains seeking reliability and compliance.
The Novel Approach
In stark contrast, the novel methodology described in the patent leverages a biomimetic transamination pathway that operates under remarkably mild conditions. By utilizing alpha-ketoesters and highly active nitrogen sources like substituted 2-hydroxybenzylamine or pyridoxamine, the reaction proceeds efficiently with simple triethylamine catalysis. This approach eliminates the need for high-pressure equipment and toxic metal catalysts entirely. The process is characterized by its operational simplicity, proceeding in a one-pot fashion where the imine intermediate is generated in situ and subsequently hydrolyzed without isolation. This streamlined workflow not only enhances safety but also drastically reduces solvent consumption and waste generation. As illustrated in the specific synthesis route below, the conversion of tert-butyl phenylpyruvate to the corresponding amino ester demonstrates the practical viability of this superior synthetic strategy.
Mechanistic Insights into Triethylamine-Catalyzed Transamination
The core innovation of this technology lies in its mechanistic efficiency, which mimics biological transamination processes but utilizes simple organic reagents. The reaction initiates with the condensation of the alpha-ketoester carbonyl group with the amine nitrogen source to form an imine intermediate. Triethylamine acts as a base catalyst, facilitating the proton transfer steps essential for the transamination equilibrium. Unlike enzymatic systems that require complex cofactors, this chemical system achieves high conversion rates through the careful selection of reaction parameters, specifically maintaining a temperature range of 25 to 100 degrees Celsius, with 60 degrees Celsius being optimal. The use of methanol as the primary solvent further enhances the solubility of reactants and stabilizes the transition states, ensuring that the reaction proceeds to completion with minimal side products.
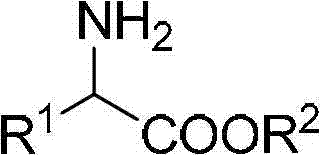
Following the formation of the imine, the second critical phase involves hydrolysis to release the free alpha-amino acid ester. This step is conducted in a biphasic or mixed solvent system comprising tetrahydrofuran and dilute hydrochloric acid. The acidic environment promotes the cleavage of the carbon-nitrogen double bond, regenerating the carbonyl functionality on the nitrogen source and releasing the desired amino ester into the aqueous phase as a salt. Subsequent basification allows for the extraction of the free amine product into an organic solvent. This two-stage mechanism ensures high selectivity and purity, as the mild hydrolysis conditions prevent racemization or degradation of sensitive functional groups often present in complex pharmaceutical intermediates. The general structural versatility of this mechanism is depicted below, highlighting the broad range of R groups compatible with the process.
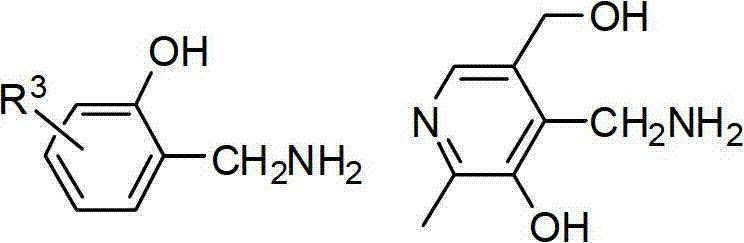
How to Synthesize Alpha-Amino Acid Esters Efficiently
Implementing this synthesis route requires precise control over stoichiometry and reaction conditions to maximize yield and purity. The patent outlines a standardized protocol that begins with the mixing of alpha-ketoester substrates with a twofold molar excess of the amine nitrogen source in methanol. The addition of catalytic triethylamine initiates the transamination, which is typically allowed to proceed for 5 to 60 hours depending on the specific substrate reactivity. Upon completion, the solvent is removed under reduced pressure to yield a crude transamination product that is sufficiently pure for direct progression to the hydrolysis step, eliminating the need for intermediate purification. For a detailed, step-by-step guide on executing this synthesis with specific reagent quantities and workup procedures, please refer to the standardized protocol section below.
- Perform transamination by reacting alpha-ketoesters with benzylamine or pyridoxamine using triethylamine as a catalyst in methanol at 60°C.
- Hydrolyze the resulting imine intermediate in a mixture of tetrahydrofuran and dilute hydrochloric acid at room temperature.
- Isolate the final product through extraction, pH adjustment to alkaline conditions, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this transamination technology offers transformative benefits for supply chain stability and cost management. By shifting away from metal-catalyzed hydrogenation, manufacturers can significantly reduce their dependency on volatile precious metal markets and eliminate the logistical complexities associated with handling high-pressure hydrogen gas. This transition translates directly into lower operational expenditures and a reduced risk profile for production facilities. Furthermore, the simplified workup procedure, which avoids complex metal scavenging steps, shortens the overall production cycle time, allowing for faster turnaround on customer orders and improved inventory turnover rates for key pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a major cost driver from the bill of materials. Additionally, the avoidance of high-pressure reactors reduces capital depreciation costs and maintenance expenses. The one-pot nature of the reaction minimizes solvent usage and labor hours required for intermediate isolation, leading to substantial overall cost savings in the manufacturing of complex amino acid derivatives without compromising on quality or yield.
- Enhanced Supply Chain Reliability: The raw materials required for this process, such as alpha-ketoesters and benzylamine derivatives, are commodity chemicals with stable global supply chains. This contrasts sharply with specialized chiral catalysts or enzymes that may have limited suppliers. By utilizing widely available reagents, manufacturers can mitigate the risk of supply disruptions and ensure consistent production schedules, thereby enhancing reliability for downstream pharmaceutical clients who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metals make this process inherently easier to scale from pilot plant to commercial tonnage. The reduced environmental footprint, characterized by lower energy consumption and simpler waste streams devoid of toxic metals, facilitates easier regulatory compliance. This aligns with modern green chemistry principles, making the process more sustainable and attractive for companies aiming to reduce their carbon footprint and meet increasingly strict environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the process capabilities and limitations for potential adopters and partners.
Q: How does this method eliminate heavy metal contamination risks?
A: Unlike conventional catalytic hydrogenation which requires transition metals, this patent utilizes an organic amine catalyst (triethylamine), completely removing the need for expensive and difficult-to-remove metal scavengers.
Q: What is the substrate scope for this transamination process?
A: The method demonstrates broad applicability, successfully synthesizing derivatives with aromatic, heteroaromatic, and aliphatic side chains, including phenyl, naphthyl, and cyclohexyl groups.
Q: Does this process require high-pressure equipment?
A: No, the reaction proceeds under atmospheric pressure and mild temperatures (25-100°C), significantly reducing capital expenditure requirements for high-pressure reactors compared to hydrogenation methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Amino Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN102675135A for producing high-quality pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha-amino acid ester meets the exacting standards required by global regulatory bodies, providing our partners with absolute confidence in product quality and consistency.
We invite pharmaceutical and agrochemical companies to collaborate with us to leverage this cost-effective and scalable technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data for our existing inventory or to discuss route feasibility assessments for your custom synthesis projects, ensuring a seamless integration of this superior technology into your manufacturing operations.
